Abstract
Arbuscular mycorrhizal (AM) fungi can improve nitrogen (N2) fixing potential of legumes under metalloid stress by enhancing nutrient uptake and modulating antioxidant defense mechanisms. Diversity among AM species in imparting metalloid tolerance highlights the importance of selecting best host-fungal combinations. Therefore, effectiveness of AM species: Rhizoglomus intraradices (Ri), Funneliformis mosseae (Fm), and Claroideoglomus claroideum (Cc) in improving N2 fixation was assessed in two differentially metalloid tolerant chickpea genotypes: HC 3 and C 235 under arsenate (AsV) and arsenite (AsIII) stress. Nodulation potential had a negative correlation with As, the impacts being more severe in C 235 than HC 3. AM species strengthened symbiotic process by enhancing availability of N, P and Fe to the nodules through increased soil enzymes activities. AM also accelerated the activities of sucrolytic enzymes in host nodules, for improved C accessibility to the bacterioids. Mycorrhization significantly increased the antioxidant enzyme activities as well as the concentrations of thiol derivatives. Ri was most efficient in improving functional efficiency of nodules, closely followed by Fm, with least positive impact of Cc, which could be related to their differential mycorrhizal colonizing abilities. Study highlighted importance of Ri inoculation for maximising the symbiotic efficiency of chickpea in As contaminated soils.





Similar content being viewed by others
References
Abd-Alla MH, Issa AA, Ohyama T (2014) Impact of harsh environmental conditions on nodule formation and dinitrogen fixation of legumes. Advances in biology and ecology of nitrogen fixation 9:1. https://doi.org/10.5772/56997
Abedi T, Mojiri A (2020) Arsenic Uptake and Accumulation Mechanisms in Rice Species. Plants 9(2):129. https://doi.org/10.3390/plants9020129
Aebi H (1984) Catalase in vitro. In: Packer L (ed) Methods in enzymology, 105th edn. Academic Press, Orlando, pp 121–126. https://doi.org/10.1016/s0076-6879(84)05016-3
Ahemad M, Kibret M (2014) Mechanisms and applications of plant growth promoting rhizobacteria: current perspective. J King Saud Univ Sci 26(1):1–20. https://doi.org/10.1016/j.jksus.2013.05.001
Arakawa N, Tsutsumi K, Sanceda NG, Kurata T, Inagaki C (1981) A rapid and sensitive method for the determination of ascorbic acid using 4,7-diphenyl-1,10-phenanthroline. Agric Biol Chem 45:1289–1290. https://doi.org/10.1080/00021369.1981.10864697
Asada K (1984) Chloroplasts: formation of active oxygen and its scavenging. Methods in Enzymology 105:422–429. https://doi.org/10.1016/S0076-6879(84)05059-X
ATSDR (Agency for Toxic Substances and Disease Registry) (2007) Toxicological profile for arsenic. Public Health Service, Atlanta, U.S. Department of Health and Human Services
Balestrini R, Gómez-Ariza J, Lanfranco L, Bonfante P (2007) Laser microdissection reveals that transcripts for five plant and one fungal phosphate transporter genes are contemporaneously present in arbusculated cells. Mol Plant Microbe Interact 20(9):1055–1062. https://doi.org/10.1094/mpmi-20-9-1055
Benabdellah K, Merlos M-A, Azcón-Aguilar C, Ferrol N (2009) GintGRX1, the first characterized glomeromycotan glutaredoxin, is a multifunctional enzyme that responds to oxidative stress. Fungal Genet Biol 46:94–103. https://doi.org/10.1094/mpmi-20-9-1055
Bhargava P, Srivastava AK, Urmil S, Rai LC (2005) Phytochelatin plays a role in UV-B tolerance in N2-fixing cyanobacterium Anabaena doliolum. J Plant Physiol 162(11):1220–1225. https://doi.org/10.1016/j.jplph.2004.12.006
Bharti A, Garg N (2019) SA and AM symbiosis modulate antioxidant defense mechanisms and asada pathway in chickpea genotypes under salt stress. Ecotoxicol Environ Saf 178:66–78. https://doi.org/10.1016/j.ecoenv.2019.04.025
Bhattacharyya P, Tripathy S, Kim K, Kim SH (2008) Arsenic fractions and enzyme activities in arsenic-contaminated soils by groundwater irrigation in West Bengal. Ecotoxicol Environ Saf 71(1):149–156. https://doi.org/10.1016/j.ecoenv.2007.08.015
Bowles TM, Barrios-Masias FH, Carlisle EA, Cavagnaro TR, Jackson LE (2016) Effects of arbuscular mycorrhizae on tomato yield, nutrient uptake, water relations, and soil carbon dynamics under deficit irrigation in field conditions. Sci Total Environ 566:1223–1234. https://doi.org/10.1016/j.scitotenv.2016.05.178
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1006/abio.1976.9999
Calabrese S, Pérez-Tienda J, Ellerbeck M, Arnould C, Chatagnier O, Boller T, Schüßler A, Brachmann A, Wipf D, Ferrol N, Courty PE (2016) GintAMT3–a low-affinity ammonium transporter of the arbuscular mycorrhizal Rhizophagus irregularis. Front Plant Sci 7:679. https://doi.org/10.3389/fpls.2016.00679
Casida LE Jr, Klein DA, Santoro T (1964) Soil dehydrogenase activity. Soil Sci 98(6):371–376
Castillo FI, Penel I, Greppin H (1984) Peroxidase release induced by ozone in Sedum album leaves. Plant Physiol 74:846–851. https://doi.org/10.1104/pp.74.4.846
Castillo FJ, Greppin H (1988) Extracellular ascorbic acid and enzyme activities related to ascorbic acid metabolism in Sedum album L. leaves after ozone exposure. Environ Exp Bot 28(3):231–238. https://doi.org/10.1016/0098-8472(88)90033-0
Chaturvedi I (2006) Effects of arsenic concentrations and forms on growth and arsenic uptake and accumulation by Indian mustard (Brassica juncea L.) genotypes. J Cent Eur Agric 7(1):31–40
Chen M, Yang G, Sheng Y, Li P, Qiu H, Zhou X, Chao Z (2017) Glomus mosseae inoculation improves the root system architecture, photosynthetic efficiency and flavonoids accumulation of liquorice under nutrient stress. Front in Plant Sci. 8:931
Corbin JD, Avis PG, Wilbur RB (2003) The role of phosphorus availability in the response of soil nitrogen cycling, understory vegetation and arbuscular mycorrhizal inoculum potential to elevated nitrogen inputs. Water Air Soil Pollut 147(1):141–162. https://doi.org/10.1023/A:1024569615325
Del Longo OT, González CA, Pastori GM, Trippi VS (1993) Antioxidant defences under hyperoxygenic and hyperosmotic conditions in leaves of two lines of maize with differential sensitivity to drought. Plant Cell Physiol 6(7):1023–1028. https://doi.org/10.1093/oxfordjournals.pcp.a078515
Dhindsa RS, Plumb-Dhindsa PAMELA, Thorpe TA (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot 32(1):93–101. https://doi.org/10.1093/jxb/32.1.93
Doke N (1983) Involvement of superoxide anion generation in the hypersensitive response of potato tuber tissues to infection with an incompatible race of Phytophthora infestans and to the hyphal wall components. Physiol Plant Pathol 23(3):345–357. https://doi.org/10.1016/0048-4059(83)90019-X
Duan GL, Zhu YG, Tong YP, Cai C, Kneer R (2005) Characterization of arsenate reductase in the extract of roots and fronds of Chinese brake fern, an arsenic hyperaccumulator. Plant Physiol 138(1):461–469. https://doi.org/10.1016/0048-4059(83)90019-X
Farzaneh M, Vierheilig H, Lössl A, Kaul HP (2011) Arbuscular mycorrhiza enhances nutrient uptake in chickpea. Plant Soil Environ 57(10):465–470. https://doi.org/10.17221/133/2011-PSE
Foyer CH, Lopez-Delgado H, Dat JF, Scott IM (1997) Hydrogen peroxide-and glutathione-associated mechanisms of acclimatory stress tolerance and signalling. Physiol Plant 100(2):241–254. https://doi.org/10.1111/j.1399-3054.1997.tb04780.x
Garg N, Chandel S (2015) Role of arbuscular mycorrhiza in arresting reactive oxygen species (ROS) and strengthening antioxidant defense in Cajanus cajan (L.) Millsp. nodules under salinity (NaCl) and cadmium (Cd) stress. Plant Growth Regul 75(2):521–534. https://doi.org/10.1007/s10725-014-0016-8
Garg N, Cheema A (2021) Relative roles of Arbuscular Mycorrhizae in establishing a correlation between soil properties, carbohydrate utilization and yield in Cicer arietinum L. under As stress. Ecotoxicol Environ Saf 207:111196. https://doi.org/10.1016/j.ecoenv.2020.111196
Garg N, Kashyap L (2019) Joint effects of Si and mycorrhiza on the antioxidant metabolism of two pigeonpea genotypes under As (III) and (V) stress. Environ Sci Pollut Res 26(8):7821–7839. https://doi.org/10.1007/s11356-019-04256-5
Georgiadis MM, Komiya H, Chakrabarti P, Woo D, Kornuc JJ, Rees DC (1992) Crystallographic structure of the nitrogenase iron protein from Azotobacter vinelandii. Science 257(5077):1653–1659 https://www.jstor.org/stable/2880123
Giovannetti M, Mosse B (1980) An evaluation of techniques for measuring vesicular arbuscular mycorrhizal infection in roots. New Phytol 84(3):489–500. https://doi.org/10.1111/j.1469-8137.1980.tb04556.x
Glover CR, Lindemann WC (2015) Nitrogen fixation by legumes revised. New Mexico State University and the US Department of Agriculture cooperating. Guide A-129. p, 4.
Gopalakrishnan S, Sathya A, Vijayabharathi R, Varshney RK, Gowda CL, Krishnamurthy L (2015) Plant growth promoting rhizobia: challenges and opportunities. 3. Biotech 5(4):355–377. https://doi.org/10.1007/s13205-014-0241-x
Gordon AJ, Minchin FR, James CL, Komina O (1999) Sucrose synthase in legume nodules is essential for nitrogen fixation. Plant Physiol 120(3):867–878
Gordon AJ, Minchin FR, Skot L, James CL (1997) Stress-induced declines in soybean N2 fixation are related to nodule sucrose synthase activity. Plant Physiol 114(3):937–946
Gupta DK, Tohoyama H, Joho M, Inouhe M (2004) Changes in the levels of phytochelatins and related metal-binding peptides in chickpea seedlings exposed to arsenic and different heavy metal ions. J Plant Res 117:253–256
Gupta S, Thokchom SD, Kapoor R (2021) Arbuscular mycorrhiza improves photosynthesis and restores alteration in sugar metabolism in Triticum aestivum L. grown in arsenic contaminated soil. Front in Plant Sci, 12:334. https://doi.org/10.3389/fpls.2021.640379
Hartree EF (1957) Haematin compounds. In: Paech K, Tracey MV (eds) Modern methods of plant analysis. Springer-Verlag, Germany, Berlin, pp 197–245. https://doi.org/10.1007/978-3-642-64961-5_7
Hasanuzzaman M, Bhuyan MHM, Anee TI, Parvin K, Nahar K, Mahmud JA, Fujita M (2019) Regulation of ascorbate-glutathione pathway in mitigating oxidative damage in plants under abiotic stress. Antioxidants 8(9):384. https://doi.org/10.3390/antiox8090384
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys. 125(1):189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Huber SC (1983) Role of sucrose-phosphate synthase in partitioning of carbon in leaves. Plant Physiol 71(4):818–821. https://doi.org/10.1104/pp.71.4.818
Ibrahim M (2017) Effect of indigenous arbuscular mycorrhizal fungi combined with manure on the change in concentration some mineral elements in cotton (Gossypium hirsutum L.). J of Plant Nutr 40(20):2862–2871
Ighodaro OM, Akinloye OA (2018) First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria J Med 54(4):287–293. https://doi.org/10.1016/j.ajme.2017.09.001
Jones MG, Outlaw WH, Lowry OH (1977) Enzymic assay of 10− 7 to 10− 14 moles of sucrose in plant tissues. Plant Physiol 60(3):379–383
Kaczyńska G, Borowik A, Wyszkowska J (2015) Soil Dehydrogenases as an Indicator of Contamination of the Environment with Petroleum Products. Water Air Soil Pollut 226:372. https://doi.org/10.1007/s11270-015-2642-9
Kiers ET, Duhamel M, Beesetty Y, Mensah JA, Franken O, Verbruggen E, Fellbaum CR, Kowalchuk GA, Hart MM, Bago A, Palmer TM (2011) Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science. 333(6044):880−882. 880-882. https://doi.org/10.1126/science.1208473
Kleinert A, Venter M, Kossmann J, Valentine A (2014) The reallocation of carbon in P deficient lupins affects biological nitrogen fixation. J Plant Physiol 171:1619–1624. https://doi.org/10.1016/j.jplph.2014.07.017
Koegel S, Lahmidi NA, Arnould C, Chatagnier O, Walder F, Ineichen K, Boller T, Wipf D, Wiemken A, Courty PE (2013) The family of ammonium transporters (AMT) in Sorghum bicolor: two AMT members are induced locally, but not systemically in roots colonized by arbuscular mycorrhizal fungi. New Phytol 198(3):853−865. https://doi.org/10.1111/nph.12199
Krishnaveni S, Balasubramanian T, Sadasivam S (1984) Sugar distribution in sweet stalk sorghum. Food Chem 15(3):229–232
Lafuente A, Pajuelo E, Caviedes MA, Rodríguez-Llorente ID (2010) Reduced nodulation in alfalfa induced by arsenic correlates with altered expression of early nodulins. J Plant Physiol 167(4):286−291. https://doi.org/10.1016/j.jplph.2009.09.014
Lokhandwala A, Hoeksema JD (2019) Priming by arbuscular mycorrhizal fungi of plant antioxidant enzyme production: a meta-analysis. Annu Plant Rev online 2(4):1069−1084. https://doi.org/10.1002/9781119312994.apr0680
López M, Herrera-Cervera JA, Iribarne C, Tejera NA, Lluch C (2008) Growth and nitrogen fixation in Lotus japonicus and Medicago truncatula under NaCl stress: nodule carbon metabolism. J Plant Physiol 165:641–650. https://doi.org/10.1016/j.jplph.2007.05.009
López-Pedrosa A, González-Guerrero M, Valderas A, Azcón-Aguilar C, Ferrol N (2006) GintAMT1 encodes a functional high-affinity ammonium transporter that is expressed in the extraradical mycelium of Glomus intraradices. Fungal Genet Biol 43(2):102−110. https://doi.org/10.1016/j.fgb.2005.10.005.
Lwalaba JLW, Louis LT, Zvobg G, Richmond MEA, Fu L, Naz S, Mwamba M, Mundende RPM, Zhang G (2020) Physiological and molecular mechanisms of cobalt and copper interaction in causing phyto-toxicity to two barley genotypes differing in Co tolerance. Ecotoxicol Environ Saf 187:109866. https://doi.org/10.1016/j.ecoenv.2019.109866
Małachowska-Jutsz A, Matyja K (2019) Discussion on methods of soil dehydrogenase determination. Int J Environ Sci Technol 16(12):77777790. https://doi.org/10.1007/s13762-019-02375-7
Maldonado-Mendoza IE, Dewbre GR, Harrison MJ (2001) A phosphate transporter gene from the extra-radical mycelium of an arbuscular mycorrhizal fungus Glomus intraradices is regulated in response to phosphate in the environment. Mol Plant Microbe Interact. 14(10):1140−1148. https://doi.org/10.1094/MPMI.2001.14.10.1140
Malik JA, Goel S, Sandhir R, Nayyar H (2011) Uptake and distribution of arsenic in chickpea: Effects on seed yield and seed composition. Commun Soil Sci Plant Anal 42(14):1728–1738
Matamoros MA, Baird LM, Escuredo PR, Dalton DA, Minchin FR, Iturbe-Ormaetxe I, Rubio MC, Moran JF, Gordon AJ, Becana M (1999) Stress-induced legume root nodule senescence. Physiol, Biochem, and structural alterations. Plant Physiol 121(1):97−112. https://doi.org/10.1104/pp.121.1.97
Merga B, Haji J (2019) Economic importance of chickpea: Production, value, and world trade. Cogent Food Agric 5(1):615718. https://doi.org/10.1080/23311932.2019.1615718
Morell M, Copeland L (1985) Sucrose synthase of soybean nodules. Plant Physiol 78(1):149–154
Muradoglu F, Gundogdu M, Ercisli S, Enuc T, Balta F, Jaafar HZE (2015) Cadmium toxicity affects chlorophyll a and b content, antioxidant enzyme activities and mineral nutrient accumulation in strawberry. Biol Res 48:11. https://doi.org/10.1186/S40659-015-0001-3
Nagarajan VK, Ebbs SD (2010) Arsenate reductase activity in roots from the arsenic hyperaccumulator Pteris vittata utilizes both glutathione and dithiothreitol as reductants. Plant Biosys 144(4):857–859. https://doi.org/10.1080/11263504.2010.522770
Nakagawara S, Sagisaka S (1984) Increase in enzyme activities related to ascorbate metabolism during cold acclimation of poplar (Populus gelrica) twigs. Plant Cell Physiol 25(6):899–906. https://doi.org/10.1093/oxfordjournals.pcp.a076804
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22(5):867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Namdjoyan S, Kermanian H, Soorki AA, Tabatabaei SM, Elyasi N (2018) Effects of exogenous salicylic acid and sodium nitroprusside on α-tocopherol and phytochelatin biosynthesis in zinc-stressed safflower plants. Turk J Botany 42(3):271–279. https://doi.org/10.3906/bot-1707-48
Neidhardt H (2020) Arbuscular mycorrhizal fungi alleviate negative effects of arsenic-induced stress on crop plants: A meta-analysis. Plants People Planet. https://doi.org/10.1002/ppp3.10122
Paterson E, Sim A, Davidson J, Daniell TJ (2016) Arbuscular mycorrhizal hyphae promote priming of native soil organic matter mineralization. Plant Soil. 408:243–C254. https://doi.org/10.1007/s11104-016-2928-8
Peleg-Grossman S, Volpin H, Levine A (2007) Root hair curling and Rhizobium infection in Medicago truncatula are mediated by phosphatidylinositide-regulated endocytosis and reactive oxygen species. J Exp Bot 58(7):1637–1649. https://doi.org/10.1093/jxb/erm013
Pereira APDA, Andrade PAMD, Bini D, Durrer A, Robin A, Bouillet JP, Andreote FD, Cardoso EJBN (2017) Shifts in the bacterial community composition along deep soil profiles in monospecific and mixed stands of Eucalyptus grandis and Acacia mangium. PloS one 12(7):e0180371. https://doi.org/10.1371/journal.pone.0180371
Pérez-Tienda J, Testillano PS, Balestrini R, Fiorilli V, Azcón-Aguilar C, Ferrol N (2011) GintAMT2, a new member of the ammonium transporter family in the arbuscular mycorrhizal fungus Glomus intraradices. Fungal Genet Biol 48(11):1044–1055. https://doi.org/10.1016/j.fgb.2011.08.003
Phillips JM, Hayman DS (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Brit Mycol Soc 55(1):158–161. https://doi.org/10.1016/S0007-1536(70)80110-3
Püschel D, Janoušková M, Voříšková A, Gryndlerová H, Vosátka M, Jansa J (2017) Arbuscular mycorrhiza stimulates biological nitrogen fixation in two Medicago spp. through improved phosphorus acquisition. Front Plant Sci 8:390. https://doi.org/10.3389/fpls.2017.00390
Rachid CTCC, Balieiro FC, Peixoto RS, Pinheiro YAS, Piccolo MC, Chaer GM, Rosado AS (2013) Mixed plantations can promote microbial integration and soil nitrate increases with changes in the N cycling genes. Soil Biol Biochem 66:146–153. https://doi.org/10.1016/j.soilbio.2013.07.005
Rusin M, Gospodarek J, Barczyk G, Nadgórska-Socha A (2018) Antioxidant responses of Triticum aestivum plants to petroleum-derived substances. Ecotoxicology 27(10):1353–1367. https://doi.org/10.1007/s10646-018-1988-3
Sardar KHAN, Qing CAO, Hesham AEL, Yue X, He JZ (2007) Soil enzymatic activities and microbial community structure with different application rates of Cd and Pb. J Environ Sci 19(7):834–840. https://doi.org/10.1016/S1001-0742(07)60139-9
Sbarato VM, Sanchez HJ (2001) Analysis of arsenic pollution in groundwater aquifers by X-ray fluorescence. Appl Radiat Isot 54(5):737–740. https://doi.org/10.1016/S0969-8043(00)00349-3
Schüβler A, Walker C (2010) The Glomeromycota: a species list with new families and genera. Edinburgh & Kew, UK, The Royal Botanic Garden; Munich, Germany: Botanische Staatssammlung Munich and Oregon, USA: Oregon State University. Available from h t t p ://schuessler.userweb. mwn.de/amphylo/Schuessler&Walker2010_Glomeromycota.pdf
Seregin IV, Ivanov VB (2001) Physiological aspects of cadmium and lead toxic effects on higher plants. Russ J Plant Physiol 48(4):523–544. https://doi.org/10.1023/A:1016719901147
Shachar-Hill Y, Pfeffer PE, Douds D, Osman SF, Doner LW, Ratcliffe RG (1995) Partitioning of intermediary carbon metabolism in vesicular-arbuscular mycorrhizal leek. Plant Physiol 108(1):7–15
Silsbury JH (1990) Estimating nitrogenase activity of faba bean (Vicia faba) by acetylene reduction (AR) assay. Funct Plant Biol 17(5):489−502. https://doi.org/10.1023/A:1016719901147
Smedley PL, Kinniburgh DG (2002) A review of the source, behaviour and distribution of arsenic in natural waters. Appl Geochem 17(5):517−568. https://doi.org/10.1016/S0883-2927(02)00018-5
Smith IK, Vierheller TL, Thorne CA (1988) Assay of glutathione reductase in crude tissue homogenates using 5, 5′-dithiobis (2-nitrobenzoic acid). Anal Biochem 175(2):408–413. https://doi.org/10.1016/0003-2697(88)90564-7
Somogyi M (1952) Notes on sugar determination. J Biol Chem 195(1):19–23
Song WY, Mendoza-Cózatl DG, Lee Y, Schroeder JI, Ahn SN, Lee HS, Wicker T, Martinoia E (2014) Phytochelatin–metal (loid) transport into vacuoles shows different substrate preferences in barley and Arabidopsis. Plant Cell Environ 37(5):1192–1201. https://doi.org/10.1111/pce.12227
Sujkowska-Rybkowska M, Borucki W, Znojek E (2012) Structural changes in Medicago truncatula root nodules caused by short-term aluminum stress. Symbiosis (Philadelphia, Pa.), 58(1-3):161–170. https://doi.org/10.1007/s13199-012-0211-1
Tajini F, Drevon, J (2012) Effect of arbuscular mycorrhizas on P use efficiency for growth and N2 fixation in common bean (Phaseolus vulgaris L.). Sci Res Essays 7, 1681e1689. https://doi.org/10.5897/SRE12.093.
Talukdar D (2013) Arsenic-induced oxidative stress in the common bean legume, Phaseolus vulgaris L. seedlings and its amelioration by exogenous nitric oxide. Physiol Mol Biol Plants 19(1):69–79. https://doi.org/10.1007/s12298-012-0140-8
Tamayo E, Gómez-Gallego T, Azcón-Aguilar C, Ferrol N (2014) Genome-wide analysis of copper, iron and zinc transporters in the arbuscular mycorrhizal fungus Rhizophagus irregularis. Front Plant Sci 5:547. https://doi.org/10.3389/fpls.2014.00547
Tawfik DS, Viola RE (2011) Arsenate replacing phosphate: alternative life chemistries and ion promiscuity. Biochemistry 50(7):1128–1134. https://doi.org/10.1021/bi200002a
Thummler F, Verma DP (1987) Nodulin-100 of soybean is the subunit of sucrose synthase regulated by the availability of free heme in nodules. J Biol Chem 262(30):14730–14736
Tian P, Razavi BS, Zhang X, Wang Q, Blagodatskaya E (2020) Microbial growth and enzyme kinetics in rhizosphere hotspots are modulated by soil organics and nutrient availability. Soil Biol Biochem 141:107662. https://doi.org/10.1016/j.soilbio.2019.107662
Tisserant E, Malbreil M, Kuo A, Kohler A, Symeonidi A, Balestrini R, Charron P, Duensing N, dit Frey, NF, Gianinazzi-Pearson V, Gilbert LB (2013) Genome of an arbuscular mycorrhizal fungus provides insight into the oldest plant symbiosis. Proc Natl Acad Sci 110(50):20117−20122. https://doi.org/10.1073/pnas.1313452110
Tripathi P, Tripathi RD, Singh RP, Dwivedi S, Chakrabarty D, Trivedi PK, Adhikari B (2013) Arsenite tolerance in rice (Oryza sativa L.) involves coordinated role of metabolic pathways of thiols and amino acids. Env Sci Poll Res 20(2):884−896. https://doi.org/10.1007/s11356-012-1205-5
Udvardi M, Poole PS (2013) Transport and metabolism in legume-rhizobia symbioses. Annu Rev Plant Biol. 64:781–805. https://doi.org/10.1146/annurev-arplant-050312-120235
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines. Plant Sci 151(1):59−66. https://doi.org/10.1016/S0168-9452(99)00197-1
Wallace TC, Murray R, Zelman KM (2016) The Nutritional Value and Health Benefits of Chickpeas and Hummus. Nutrients 8(12):766. https://doi.org/10.3390/nu8120766
Yarnes SC, Sengupta-Gopalan C (2009) Sucrose synthase levels do not limit or regulate carbon transfer in the arbuscular mycorrhizal symbiosis. J Plant Interact 4(2):113–117. https://doi.org/10.1080/17429140902898429
Yizhu L, Imtiaz M, Ditta A, Rizwan MS, Ashraf M, Mehmood S, Aziz O, Mubeen F, Ali M, Elahi NN, Ijaz R (2020) Response of growth, antioxidant enzymes and root exudates production towards As stress in Pteris vittata and in Astragalus sinicus colonized by arbuscular mycorrhizal fungi. Environ Sci Pollut Res 27(2):2340–2352. https://doi.org/10.1007/s11356-019-06785-5
Zantua MI, Bremner JM (1975) Comparison of methods of assaying urease activity in soils. Soil Biol Biochem 7(4−5): 291−295. https://doi.org/10.1016/0038-0717(75)90069-3
Zhang XF, Hu ZH, Yan TX, Lu RR, Peng CL, Li SS, Jing YX (2019) Arbuscular mycorrhizal fungi alleviate Cd phytotoxicity by altering Cd subcellular distribution and chemical forms in Zea mays. Ecotoxicol Environ Saf. 171:352–360. https://doi.org/10.1016/j.ecoenv.2018.12.097
Zou YN, Wu QS, Kuča K (2020) Unravelling the role of arbuscular mycorrhizal fungi in mitigating the oxidative burst of plants under drought stress. Plant Biol 23:50−57. https://doi.org/10.1111/plb.13161
Zwiazek JJ, Blake TJ (1991) Early detection of membrane injury in black spruce (Picea mariana). Can J Forest Res 21(3):401−404. https://doi.org/10.1139/x91-050
Acknowledgements
Authors are thankful to Haryana Agriculture University (HAU), Hisar, Indian Agriculture Research Institute (IARI), The Energy and Resource Institute (TERI), New Delhi, for providing biological materials. We pay our sincere regards to Sophisticated Analytical Instrumentation Facility (SAIF), Panjab University, Chandigarh for technical assistance.
Availability of data and materials
The data supporting the results of present study are included in this article and supplementary materials.
Funding
The research work was financed by The Department of Biotechnology (DBT), Government of India (BT/PR13409/BPA/118/122/2015).
Author information
Authors and Affiliations
Contributions
Both the authors contributed equally in the preparation of manuscript. NG designed the work plan, AC conducted research experiments. Both NG and AC finalized the research article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information

ESM 1
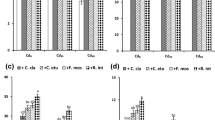
Fig. 1 Effect of mycorrhizal inoculations on enzymatic antioxidant activities: a. Ascorbate Peroxidase (APOX, nkat mg-1 protein), b. Monodehydroascorbate reductase (MDHAR, nkat mg-1 protein) and c. Dehydroascorbate reductase (DHAR, nkat mg-1 protein) in nodules of two Cicer arietinum L. genotypes (HC 3-As tolerant, and C 235-As sensitive) under AsV60 and AsIII10 stress. Values are means of six replicates ± standard error (SE). Points topped by the same letter do not differ significantly at p ≤ 0.05 as assessed by Duncan’s multiple range test (DMRT). ––NM (non-mycorrhizal plants), –Cc (Claroideoglomus claroideum), –Fm- (Funneliformis mosseae), –Ri- (Rhizoglomus intraradices) (PNG 4322 kb)
ESM 2
(DOCX 27 kb)
ESM 3
(DOCX 30 kb)
ESM 4
(DOCX 18 kb)
Rights and permissions
About this article
Cite this article
Cheema, A., Garg, N. Differential effectiveness of Arbuscular Mycorrhizae in improving Rhizobial symbiosis by modulating Sucrose metabolism and Antioxidant defense in Chickpea under As stress. Symbiosis 86, 49–69 (2022). https://doi.org/10.1007/s13199-021-00815-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-021-00815-y