Abstract
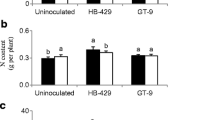
Organic farmers recognize the importance of using Rhizobium to meet crop N fertility needs and to reduce use of chemical fertilizers. Field experiments were conducted during the 2015 and 2016 seasons at different organic fields to assess the effect of indigenous Rhizobium leguminosarum bv. phaseoli (OrgK9) and commercial R. tropici (CIAT899) strains on symbiotic performance and grain yield of bean varieties, Eclipse and Redhawk. Averaged for both varieties, nodule numbers on plants inoculated with CIAT899 (35.2 ± 3.7 and 26.3 ± 2.6 /plant) and OrgK9 (29.6 ± 2.5 and 26.8 ± 2.1 /plant) were significantly greater (P < 0.0001) than the control (18.9 ± 2.2 and 10.5 ± 3.1) in 2015 and 2016, respectively. Bean genotype Eclipse cultivated at Lamberton and inoculated with OrgK9 had (P < 0.0001) greater number of nodules compared with CIAT899-inoculated or non-inoculated plants. The N-content (3.3 ± 0.04%) of plants inoculated with CIAT899 was greater (P < 0.0001) compared with plants inoculated with OrgK9. Strain CIAT899 increased nodule occupancy by 29 and 62% and 4 and 26% on Red Hawk cultivated in the Lamberton and Farmington fields compared with the control in season 2015 and 2016, respectively. The majority of nodules formed on Red Hawk (76%) and Eclipse (64%) at Farmington were occupied by CIAT899 and OrgK9 in 2015 and 2016, respectively. Eclipse inoculated with OrgK9 had a significantly greater seed yields (2683 ± 402 and 2546 ± 247 kg/ha) compared with control (2265 ± 996 and 2058 ± 51 kg/ha) in both years, respectively. These results clearly indicate that symbiotic performance and grain yield of dry bean can be significantly increased by using Rhizobium inoculation under organic farming systems.





Similar content being viewed by others
References
Abaidoo RC, George T, Bohlool BB, Singleton PW (1989) Influence of elevation and applied nitrogen on rhizosphere colonization and competition for nodule occupancy by different rhizobial strains on field-grown soybean and common bean. Can J Microbiol 36:92–96
Abou-Shanab RAI, Wongphatcharachai M, Sheaffer CC, Orf JC, Sadowsky MJ (2017) Competition between introduced Bradyrhizobium japonicum strains and indigenous bradyrhizobia in Minnesota organic farming systems. Symbiosis 73:155–163
Andrade DS, Hungria M (2002) Maximizing the contribution of biological nitrogen fixation in tropical legume crops. In: Finan TM et al (eds) Nitrogen fixation: global perspectives. Proc. 13th Int. Congr. On nitrogen fixation, Hamilton, ON, CAN, 2–7 July, vol 2001. CAB Publ, Wallingford, Oxon, pp 341–345
Asad S, Malik KA, Hafeez FY (1991) Competition between inoculated and indigenous Rhizobium/Bradyrhizobium spp. strains for nodulation of grain and fodder legumes in Pakistan. Biol Fertil Soils 12:107–111
Beringer JE (1974) R factor transfer in Rhizobiurn leguminosarum. J Gen Microbiol 84:188–198
Bohlool BB, Schmidt EL (1973) Persistence and competition aspects of Rhizobium japonicum observed in soil by immunofluorescence microscopy. Soil Sci Soc Am J 37:561–564
Brutti L, Piantanida N, Ljunggren H, Berggren I, Martensson A (1999) Competition between strains of Bradyrhizobium japonicum for nodulation of soybeans in argentine arable soils. Appl Soil Ecol 12:1–6
Buttery BR, Park SJ, van Berkum P (1997) Effects of common bean (Phaseolus vulgaris L.) cultivar and Rhizobium strain on plant growth, seed yield and nitrogen content. Can J Plant Sci 77:347–351
Dall’Agnol RF, Ribeiro RA, Ormeño-Orrillo E, Rogel MA, Delamuta JRM et al (2013) Rhizobium freirei sp. nov., a symbiont of Phaseolus vulgaris that is very effective at fixing nitrogen. Int J Sys Evol Microbiol 63:4167–4173
del Cerro P, Pérez-Montaño F, Gil-Serrano A, López-Baena FJ, Megías M et al (2017) The Rhizobium tropici CIAT 899 NodD2 protein regulates the production of nod factors under salt stress in a flavonoid independent manner. Sci Reports 7:46712
Dinesh DS, Lal Das M, Picado A, Roy L, Rijal S et al (2010) Insecticide susceptibility of Phlebotomus argentipes in visceral Leishmaniasis endemic districts in India and Nepal. Plos Neglect Trop D 4(10):e859
Dobermann A (2012) Getting back to the field. Nature 485:176–177
FAO (2013) FAO statistical yearbook 2013: world food and agriculture. http://www.fao.org/3/i3107e/i3107e00.htm. Accessed 30 June 2013
George MLC, Robert FM (1992) Competition among Rhizobium leguminosarum bv. phaseoli strains for nodulation of common bean. Can J Microbiol 38:I57–I160
Graham PH (1981) Some problems of nodulation and symbiotic nitrogen fixation in Phaseolus vulgaris L.: a review. Field Crop Res 4:93–112
Graham PH, Vance CP (2003) Legumes: importance and constraints to greater use. Plant Physiol 131:872–877
Han OK, Kaga A, Isemura T, Wang XW, Tomooka N, Vaughan DA (2005) A genetic linkage map for azuki bean. [Vigna angularis (Willd.) Ohwi & Ohashi] Theor. Appl Genet 111:1278–1287
Hardarson G (1993) Methods for enhancing symbiotic nitrogen fixation. Plant Soil 152:1–17
Herridge DF, Peoples M, Boddey RM (2008) Global inputs of biological nitrogen fixation in agricultural systems. Plant Soil 311:1–18
Hungria M, Mendes IC (2015) Nitrogen fixation with soybean: the perfect symbiosis? In: de Bruijn F (ed) Biological nitrogen fixation. JohnWiley & Sons, New Jersey, pp 1005–1019
Hungria M, Vargas MAT (2000) Environmental factors affecting N2 fixation in grain legumes in the tropics with an emphasis on Brazil. Field Crop Res 65:151–164
Hungria M, Andrade DS, Chueire LMO, Probanza A, Guttierrez-Man˜ ero FJ et al (2000) Isolation and characterization of new efficient and competitive bean (Phaseolus vulgaris L.) rhizobia from Brazil. Soil Biol Biochem 32:1515–1528
Kaschuk G, Nogueira MA, de Luca MJ, Hungria M (2016) Response of determinate and indeterminate soybean cultivars to basal and topdressing N fertilization compared to sole inoculation with Bradyrhizobium. Field Crop Res 195:21–27
Kelly JD (1988) Effect of row-spacing on dry bean yields in Michigan. Michigan Dry Bean Digest 12:6–8
Koskey G, Mburu SW, Njeru EM, Kimiti JM, Ombori O et al (2017) Potential of native rhizobia in enhancing nitrogen fixation and yields of climbing beans (Phaseolus vulgaris L.) in contrasting environments of eastern Kenya. Front. Plant Sci 8:443
López-López A, Rogel-Hernández MA, Barois I, Ortiz Ceballos AI, Martínez J et al (2012) Rhizobium grahamii sp. nov., from nodules of Dalea leporina, Leucaena leucocephala and Clitoria ternatea, and Rhizobium mesoamericanum sp. nov., from nodules of Phaseolus vulgaris, siratro, cowpea and Mimosa pudica. Int J Syst Evol Microbiol 62:2264–2271
Martennsson AM, Gustafsson JG (1985) Competition between Rhizobium trifolii strains for nodulation during growth in a fermenter, and in soil-based inoculants, studied by ELISA. J Gen Microbiol 131:3077–3082
Martínez-Romero E, Segovia L, Mercante FM, Franco AA, Graham P et al (1991) Rhizobium tropici, a novel species nodulating Phaseolus vulgaris L. beans and Leucaena sp. trees. Int J Syst Bacteriol 41:417–426
Martiny JB, Bohannan BJ, Brown JH, Colwell RK, Fuhrman JA (2006) Microbial biogeography: putting microorganisms on the map. Nat Rev Microbiol 4:102–112
Mesquita FR, Corrêa AD, Abreu CMP, Lima RAZ, Abreu AFB (2007) Bean (Phaseolus vulgaris L.) lines: chemical composition and protein digestibility. Cienc Agrotec 31:1114–1121
Mhamdi R, Jebara M, Aouani ME, Ghrir R, Mars M (1999) Genotypic diversity and symbiotic effectiveness of rhizobia isolated from root nodules of Phaseolus vulgaris L., grown in Tunisian soils. Biol Fertil Soil 28:313–320
Mhamdi R, Laguerre G, Aouani ME, Mars M, Amarger N (2002) Different species and symbiotic genotypes of field rhizobia can nodulate Phaseolus vulgaris in Tunisian soils. FEMS Microbiol Ecol 41:77–84
Mnasri B, Saı ̈di S, Chihaoui SA, Mhamdi R (2012) Sinorhizobium americanum symbiovar mediterranense is a predominant symbiont that nodulates and fixes nitrogen with common bean (Phaseolus vulgaris L.) in a northern Tunisian field. Syst Appl Microbiol 35:263–269
Mnasri B, Liu TY, Saidi S, Chen WF, Chen WX, Zhang XX, Mhamdi R (2014) Rhizobium azibense sp nov., a nitrogen fixing bacterium isolated from root-nodules of Phaseolus vulgaris. Int J Syst Evol Microbiol 64:1501–1506
Moreira A, Moraes LAC (2017) Yield, nutritional status and soil fertility cultivated with common bean in response to amino acids foliar application. J. Plant Nutrition 40:344–351
Mostasso L, Mostasso FL, Dias BG, Vargas MAT, Hungria M (2002) Selection of bean (Phaseolus vulgaris L.) rhizobial strains for the Brazilian Cerrados. Field Crops Res 73:121–132
Naamala J, Jaiswal SK, Dakora FD (2016) Antibiotics resistance in Rhizobium: type, process, mechanism and benefit for agriculture. Curr Microbiol 72:804–816
Osorno JM, Grafton KF, Rojas-Cifuentes GA, Gelin R, Vander Wal AJ (2010) Registration of ‘lariat’ and ‘stampede’ pinto beans. J Plant Registrat 4:5–11
Park S, Cao C, Gardener BBM (2010) Inoculants and soil amendments for organic growers. Ohio State University extension. http://ohioline.osu.edu/sag-fact/pdf/0017. Accessed 25 June 2010
Pelegrin R, Mercante FM, Otsubo IMN, Otsubo AA (2009) Response of common bean crop to nitrogen fertilization and Rhizobium inoculation. Rev Bras Cienc Solo 33:219–226
Pimentel D, Hepperly P, Hanson J, Douds D, Seidel R (2005) Environmental, energetic, and economic comparisons of organic and conventional farming systems. Bioscience 55:573–582
Pinto FGS, Hungria M, Mercante FM (2007) Polyphasic characterization of Brazilian Rhizobium tropici strains effective in fixing N2 with common bean (Phaseolus vulgaris L.). Soil Biol Biochem 39:1851–1864
Popescu A (1998) Contributions and limitations of symbiotic nitrogen fixation in Phaseolus vulgaris in Romania. Plant Soil 204:117–125
Raposeiras R, Marriel IE, Muzzi MRS, Paiva E, Filho IAP et al (2006) Rhizobium strains competitiveness on bean nodulation in Cerrado soils. Brasília 41:439–447
Reeve JR, Hoagland LA, Villalba JJ, Carr PM, Atucha A et al (2016) Organic farming, soil health, and food quality: considering possible links. Adv Agron 37:319–367
Sadowsky MJ, Cregan PB, Keyser HH (1988) Nodulation and nitrogen fixation efficacy of Rhizobium fredii with Phaseolus vulgaris genotypes. Appl Environ Microbiol 54:1907–1910
Samago TY, Anniye EW, Dakora FD (2018) Grain yield of common bean (Phaseolus vulgaris L.) varieties is markedly increased by rhizobial inoculation and phosphorus application in Ethiopia. Symbiosis 75:245–255
Singleton PW, Tavares JW (1986) Inoculation response of legumes in relation to the number and effectiveness of indigenous Rhizobium populations. Appl Environ Microbiol 51:1013–1018
Siqueira-Neto M, Galdos MV, Feigl BJ, Cerri CEP, Cerri CC (2016) Direct N2O emission factors for synthetic N-fertilizer and organic residues applied on sugarcane for bioethanol production in central-southern Brazil. GCB Bioenergy 8:269–280
Smith GB, Wollum AG (1989) Nodulation of Glycine max by six Bradyrhizobium japonicum strains with different competitive abilities. Appl Environ Microbiol 55:1957–1962
Somasegaran P, Hoben HJ (1985) Methods in legume-Rhizobium technology. University of Hawaii NifTAL Project and MIRCEN, Dept. of Agronomy and Soil Science, Hawaii Institute of Tropical Agriculture and Human Resources, College of Tropical Agriculture and Human Resources, Paia, Maui, HI
Souza HAD, Natale W, Rozane DE, Hernandes A, Romualdo LM (2011) Liming and fertilization with boron in production of bean. Rev Ciênc Agron 42:249–257
Tajini F, Drevon JJ, Lamouchi L, Aouani ME, Trabelsi M (2008) Response of common bean lines to inoculation: comparison between the Rhizobium tropici CIAT899 and the native Rhizobium etli 12a3 and their persistence in Tunisian soils. World J Microbiol Biotechnol 24:407–417
Thies JE, Singleton PW, Bohlool BB (1991) Influence of the size of indigenous rhizobial populations on establishment and symbiotic performance of introduced rhizobia on field-grown legumes. Appl Environ Microbiol 57:19–28
Trabelsi M, Sifi B (1997) Screening for more performant common bean varieties. In: Drevon JJ (ed) FYSAME annual report. INRA Montpellier, pp 23–33
Willer H, Lernoud J (2017) The world of organic agriculture statistics and emerging trends. CH-5070 Frick, Switzerland
Williams PRD, Hammitt JK (2001) Perceived risks of conventional and organic produce: pesticides, pathogens, and natural toxins. Risk Anal 21:319–330
Wolff AB, Singleton PW, Siderelli M, Bohlool BB (1993) Influence of acid soil on nodulation and inter strain competitiveness in relation to tannin concentration in seeds and roots of Phaseolus vulgaris. Soil Biol Biochem 25:715–721
Wongphatcharachai M, Wang P, Staley C, Chun CL, Ferguson JA, Moncada KM, Sheaffer CC, Sadowsky MJ (2015) Site-specific distribution and competitive ability of indigenous bean-nodulating rhizobia isolated from organic fields in Minnesota. J Biotechnol 214:158–168
Zaman-Allah M, Sifi B, L’taief M, El Aouni H, Drevon JJ (2007) Rhizobial inoculation and P fertilization response in common bean (Phaseolus vulgaris) under glasshouse and field conditions. Exp Agric 43:67–77
Zurdo-Piñeiro JL, García-Fraile P, Rivas R, Peix A, León-Barrios M et al (2009) Rhizobia from Lanzarote, the Canary Islands, that nodulate Phaseolus vulgaris have characteristics in common with Sinorhizobium meliloti isolates from mainland Spain. Appl Environ Microbiol 75:2354–2359
Acknowledgements
This work was supported, in part, from grant 201151300-30743 from USDA-NIFA, and by the Minnesota Agricultural Experiment Station.
The authors thank Patrick Elia from USDA-ARS, Beltsville, MD for providing the Rhizobium tropici CIAT 899 (USDA 2744) reference strain. We also would like to especially thank Doug Kremer and Elizabeth Fleege of Terramax, Inc., Minneapolis, MN for preparing Rhizobium inoculants and the organic farmers who participated in our study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abou-Shanab, R.A.I., Wongphatcharachai, M., Sheaffer, C.C. et al. Response of dry bean (Phaseolus vulgaris L.) to inoculation with indigenous and commercial Rhizobium strains under organic farming systems in Minnesota. Symbiosis 78, 125–134 (2019). https://doi.org/10.1007/s13199-019-00609-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-019-00609-3