Abstract
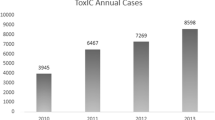
The Toxicology Investigators Consortium (ToxIC) Case Registry was established by the American College of Medical Toxicology in 2010. The Registry contains data from participating sites with the agreement that all bedside medical toxicology consultations will be entered. Currently, 83% of accredited medical toxicology fellowship programs in the USA participate. The Registry continues to grow each year, and as of 31 December 2016, a new milestone was reached, with more than 50,000 cases reported since its inception. The objective of this seventh annual report is to summarize the Registry’s 2016 data and activity with its additional 8529 cases. Cases were identified for inclusion in this report by a query of the ToxIC database for any case entered from 1 January to 31 December 2016. Detailed data was collected from these cases and aggregated to provide information which includes the following: demographics (age, gender, race, ethnicity, HIV status), reason for medical toxicology evaluation (intentional pharmaceutical exposure, envenomation, withdrawal from a substance), agent and agent class, clinical signs and symptoms (vital sign abnormalities, organ system dysfunction), treatments and antidotes administered, fatality and life support withdrawal data. Fifty percent of cases involved females, and adults aged 19–65 were the most commonly reported. There were 86 patients (1.0%) with HIV-positive status known. Non-opioid analgesics were the most commonly reported agent class, with acetaminophen the most common agent reported. There were 126 fatalities reported in 2016 (1.5% of cases). Major trends in demographics and exposure characteristics remained similar overall with past years’ reports. While treatment interventions were commonly required, fatalities were rare.

Similar content being viewed by others
References
Wax PM, Kleinschmidt KC, Brent J. ACMT ToxIC Case Registry Investigators. The Toxicology Investigators Consortium (ToxIC) Registry. J Med Toxicol. 2011 Dec;7(4):259–65.
Farrugia L, Rhyee SH, Campleman SL, et al. The Toxicology Investigators Consortium Case Registry—the 2015 experience. J Med Toxicol. 2016 Sep;12(3):224–47.
Beauchamp GA, Hendrickson RG, Hatten BW, Toxicology Investigators Consortium. Endotracheal intubation for toxicologic exposures: a retrospective review of Toxicology Investigators Consortium (ToxIC) cases. J Emerg Med. 2016;51(4):382–8.
Judge BS, Ouellette LM, VandenBerg M, Riley BD, Wax PM, On behalf of the ACMT Toxicology Investigators Consortium (ToxIC). Utilization of observation units for the care of poisoned patients: trends from the Toxicology Investigators Consortium Case Registry. J Med Toxicol. 2016;12:111–20.
Riederer AM, Campleman SI, Carlson RG, Toxicology Investigators Consortium (ToxIC), et al. Acute poisonings from synthetic cannabinoids—50 US Toxicology Investigators Consortium registry sites, 2010–2015. MMWR. Morb Mortal Wkly Rep. 2016;65(27):692–5.
Wang GS, Levitan R, Wiegand TJ, et al. Extracorporeal membrane oxygenation (ECMO) for severe toxicological exposures: review of the Toxicology Investigators Consortium (ToxIC). J Med Toxicol. 2016 Mar;12(1):95–9.
Rhyee SH, Farrugia L, Campleman SL, et al. The Toxicology Investigators Consortium Case Registry—the 2014 experience. J Med Toxicol. 2015 Dec;11(4):388–409.
Rhyee SH, Farrugia L, Wiegand T, et al. The Toxicology Investigators Consortium Case Registry—the 2013 experience. J Med Toxicol. 2014 Dec;10(4):342–59.
Wiegand T, Wax P, Smith E, et al. The Toxicology Investigators Consortium Case Registry—the 2012 experience. J Med Toxicol. 2013 Dec;9(4):380–404.
Wiegand TJ, Wax PM, Schwartz T, et al. The Toxicology Investigators Consortium Case Registry—the 2011 experience. J Med Toxicol. 2012 Dec;8(4):360–77.
Brent J, Wax PM, Schwartz T, et al. The toxicology investigators consortium case registry—The 2010 experience. J Med Toxicol. 2011 Dec;7(4):266–76.
Hedegaard H, Warner M, Minino AM. Drug overdose deaths in the United States, 1999-2015. NCHS Data Brief. 2017 Feb;273:1–8.
Rudd RA, Seth P, David F, Scholl L. Increases in drug and opioid-involved overdose deaths - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016 Dec 30;65(5051):1445–52.
Sjogren PP, Skarda DE, Park AH. Upper aerodigestive injuries from detergent ingestion in children. Laryngoscope. 2017 Feb;127(2):509–12.
Swain TA, McGwin G Jr, Griffin R. Laundry pod and non-pod detergent related emergency department visits occurring in children in the USA. Inj Prev. 2016 Dec;22(6):396–9.
Stromberg PE, Burt MH, Rose SR, Cumpston KL, Emswiler MP, Wills BK. Airway compromise in children exposed to single-use laundry detergent pods: a poison center observational case series. Am J Emerg Med. 2015 Mar;33(3):349–51.
Gray ME, West CE. Corneal injuries from liquid detergent pods. J AAPOS. 2014 Oct;18(5):494–5.
Huntington S, Heppner J, Vohra R, Mallios R, Geller RJ. Serious adverse effects from single-use detergent sacs: report from a U.S. statewide poison control system. Clin Toxicol (Phila). 2014 Mar;52(3):220–5.
Valdez AL, Casavant MJ, Spiller HA, Chounthirath T, Xiang H, Smith GA. Pediatric exposure to laundry detergent pods. Pediatrics. 2014 Dec;134(6):1127–35.
Beuhler MC, Gala PK, Wolfe HA, Meaney PA, Henretig FM. Laundry detergent "pod" ingestions: a case series and discussion of recent literature. Pediatr Emerg Care. 2013 Jun;29(6):743–7.
Centers for Disease Control and Prevention (CDC). Health hazards associated with laundry detergent pods - United States, May–June 2012. MMWR Morb Mortal Wkly Rep. 2012 Oct 19;61(41):825–9.
Settimi L, Giordano F, Lauria L, Celentano A, Sesana F, Davanzo F. Surveillance of paediatric exposures to liquid laundry detergent pods in Italy. Inj Prev. 2017 Feb 10. pii: injuryprev-2016-042263.
Villa A, Médernach C, Arropetian N, Lagrange F, Langrand J, Garnier R. Exposure to liquid detergent capsules: a study of the cases reported to the Paris Poison Center, 2011–2012. Arch Pediatr. 2014;21(6):608–13. French
Williams H, Jones S, Wood K, Scott RA, Eddleston M, Thomas SH, et al. Reported toxicity in 1486 liquid detergent capsule exposures to the UK National Poisons Information Service 2009-2012, including their ophthalmic and CNS effects. Clin Toxicol (Phila). 2014 Feb;52(2):136–40.
CPSC. 2013. CPSC and ACCC warn of poison dangers with liquid laundry packets. Release date: March 21, 2013; Available: https://www.cpsc.gov/content/cpsc-and-accc-warn-of-poison-dangers-with-liquid-laundry-packets, Accessed 18 June 2017.
AAPCC. 2017. Laundry detergent packets (unit dose liquid) Data, May 31, 2017. Available: http://www.aapcc.org/alerts/laundry-detergent-packets/, Accessed 18 June 2017.
ASTM International. New releases: new ASTM International standard will help improve safety of liquid detergent laundry packets. Release #9900, September 4, 2015. Available: http://www.astmnewsroom.org/default.aspx?pageid=3813, Accessed 18 June 2017.
P&G (Proctor & Gamble). P&G announces change to laundry pacs and taps safe kids worldwide to help promote safety among parents. June 30, 2015; Available: http://news.pg.com/press-release/pg-corporate-announcements/pg-announces-change-laundry-pacs-and-taps-safe-kids-worldwi, Accessed 18 June 2017.
Acknowledgments
Toxicology Investigators Consortium (ToxIC) Study Group Collaborators:
Aks SE, Algren DA, Alwasiyah D, Beauchamp G, Bentur Y, Beuhler MC, Boyle KL, Bruccoleri RE, Burns, MM, Cahana A, Cannon RD, Caravati EM, Carey JL, Chhabra N, Chomin J, Christian MR, Conner K, Cook MD, Cumpston KL, Dissanayake V, Dribben WH, Eisenga BH, Engebretsen KM, Falkowitz D, Farkas A, Fenton A, Froberg BA, Furmaga JF, Ganetsky M, Garlich F, Geib AJ, Gorodetsky R, Greene S, Greller HA, Gummin DD, Hart K, Hendrickson RG, Hernandez S, Hoyte CO, Judge BS, Kazzi Z, Kerns W, Kessler BD, King J, Kirschner R, Kleinschmidt KC, Kostic MA, Kusin S, Leikin, JB,LeRoy JM, Levine M, Lo CY, Lowry JA, Lung D, Lurie Y, Maddry J, Majlesi N, Manini AF, Marlin MB, McKay C, McKeever RG, McKeown NJ, Meggs WJ, Miller SN, Minns A, Moore E, Morgan BW, Mullins ME, Nappe TM, Nogar JN, Oakley E, Olmedo R, Othong R, Parker-Cote J,Regina A, Riley BD, Rowden A, Ruha AM, Rusyniak DE, Schult, R, Schwarz ES, Seifert SA, Sessions D, Shulman J, Smolinske SC, Smollin C, Spyres MB, Steck A, Stellpflug SJ, Sullivan R, Toce MS, Troendle MM, Vearrier D, Warrick BJ, Watts DC, Wills BK, Wolk BJ, Zosel AE.
We also wish to thank study coordinators AB Adefeso, Mary Connie Aubin, Anita Kurt, Julie Licata, Misti Marshall, Maureen Morgan, Tammy Phan, Andrea Ramirez, and Melissa VandenBerg.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Funding
ToxIC was supported by a continuation of a grant from the US National Institutes of Health on cardiovascular drug toxicity, a new contract with the US Food and Drug Administration, and the continuation of unrestricted grant support from BTG International, which was used to support the North American Snakebite Registry.
Conflicts of Interest
None
Previous Presentation of Data
None
Funding Sources
This study received funding from the NIH National Institute on Drug Abuse 1R56DA38366, and 1R01DA037317-01, a data sharing contract with the U.S. Food and Drug Administration, and BTG International Inc.
Electronic supplementary material
Table S1
(DOCX 28 kb)
Table S2
(DOCX 53 kb)
Table S3
(DOCX 42 kb)
Table S4
(DOCX 45 kb)
Table S5
(DOCX 44 kb)
Table S6
(DOCX 40 kb)
Table S7
(DOCX 45 kb)
Table S8
(DOCX 31 kb)
Table S9
(DOCX 34 kb)
Table S10
(DOCX 50 kb)
Table S11
(DOCX 34 kb)
Table S12
(DOCX 34 kb)
Table S13
(DOCX 30 kb)
Table S14
(DOCX 30 kb)
Table S15
(DOCX 28 kb)
Table S16
(DOCX 30 kb)
Table S17
(DOCX 28 kb)
Table S18
(DOCX 77 kb)
Rights and permissions
About this article
Cite this article
Farrugia, L.A., Rhyee, S.H., Calello, D.P. et al. The Toxicology Investigators Consortium Case Registry—the 2016 Experience. J. Med. Toxicol. 13, 203–226 (2017). https://doi.org/10.1007/s13181-017-0627-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13181-017-0627-3