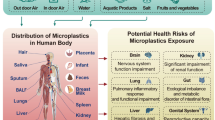
Abstract
Zinc oxide nanoparticles (ZnO NPs) is one of the most exploited nanoparticles in the biomedical field due to its enormous potential in disease diagnostics and monitoring. The upsurge in demand for ZnO NPs due to its peculiar properties and vast applications has led to an increase of inadvertent exposure of ZnO NPs to humans, resulting in higher risk for nanotoxicity. Previous literature is strongly suggestive of the notion that ZnO NPs may induce adverse health effects in humans and organisms in the environment. Hence, knowledge of the physiological interactions of ZnO NPs with the biological system as well as understanding the interplay between the physicochemical properties of ZnO NPs and its toxic effects are key factors in developing ZnO NPs that can be safely applied in the biomedical field. At present, many experiment models have been employed to unveil the possible unforeseen effect of ZnO NPs on the human, animals, and environment. Among these experiment models, animal models are of paramount importance as it allows the prediction of the possible toxic effects of ZnO NPs in individual organs or the body as a whole. The current review systemizes and summarizes the in vivo assessments of organ-specific nanotoxicity of ZnO NPs using animal models published between 2015 and 2020. Besides, this review attempts to outline the possible mechanisms of ZnO NPs–induced toxicity and factors influencing these toxic effects of ZnO NPs.





Similar content being viewed by others
References
Rauta, P. R., Mohanta, Y. K., & Nayak, D. (2019). Nanotechnology in biology and medicine. Nanotechnology in Biology and Medicine. https://doi.org/10.1201/9780429259333.
Sukhanova, A., Bozrova, S., Sokolov, P., Berestovoy, M., Karaulov, A., & Nabiev, I. (2018). Dependence of nanoparticle toxicity on their physical and chemical properties. Nanoscale Research Letters. https://doi.org/10.1186/s11671-018-2457-x.
Gornati, R., Papis, E., Di Gioacchino, M., Sabbioni, E., Dalle-Donne, I., Milzani, A., & Bernardini, G. (2009). In vivo and in vitro models for nanotoxicology testing. in nanotoxicity: From in vivo and in vitro models to health risks. doi:https://doi.org/10.1002/9780470747803.ch15
Boraschi, D., & Italiani, P. (2016). Model validity in nanoimmunosafety: Advantages and disadvantages of in vivo vs in vitro models, and human vs animal models. Current Bionanotechnology. https://doi.org/10.2174/2213529402666160601121721.
Akhtar, M. J., Ahamed, M., Kumar, S., Majeed Khan, M. A., Ahmad, J., & Alrokayan, S. A. (2012). Zinc oxide nanoparticles selectively induce apoptosis in human cancer cells through reactive oxygen species. International Journal of Nanomedicine, 7, 845–857. https://doi.org/10.2147/IJN.S29129.
Zangeneh, M., Nedaei, H. A., Mozdarani, H., Mahmoudzadeh, A., Kharrazi, S., & Salimi, M. (2019). The role and mechanisms of zinc oxide nanoparticles in the improvement of the radiosensitivity of lung cancer cells in clinically relevant megavoltage radiation energies in-vitro. Nanomedicine Journal, 6(4), 276–290. https://doi.org/10.22038/nmj.2019.06.000006.
Rasmussen, J. W., Martinez, E., Louka, P., & Wingett, D. G. (2010). Zinc oxide nanoparticles for selective destruction of tumor cells and potential for drug delivery applications. Expert Opinion on Drug Delivery. https://doi.org/10.1517/17425247.2010.502560.
Zhang, T., Du, E., Liu, Y., Cheng, J., Zhang, Z., Xu, Y., et al. (2020). Anticancer effects of zinc oxide nanoparticles through altering the methylation status of histone on bladder cancer cells. International Journal of Nanomedicine, 15, 1457–1468. https://doi.org/10.2147/IJN.S228839.
Abbasian, M., Mahmoodzadeh, F., Khalili, A., & Salehi, R. (2019). Chemotherapy of breast cancer cells using novel pH-responsive cellulose-based nanocomposites. Advanced Pharmaceutical Bulletin, 9(1), 122–131. https://doi.org/10.15171/apb.2019.015.
Hu, C., & Du, W. (2020). Zinc oxide nanoparticles (ZnO NPs) combined with cisplatin and gemcitabine inhibits tumor activity of NSCLC cells. Aging, 12. https://doi.org/10.18632/aging.104187.
Antoine, T. E., Hadigal, S. R., Yakoub, A. M., Mishra, Y. K., Bhattacharya, P., Haddad, C., et al. (2016). Intravaginal zinc oxide tetrapod nanoparticles as novel immunoprotective agents against genital herpes. The Journal of Immunology. https://doi.org/10.4049/jimmunol.1502373.
Agelidis, A., Koujah, L., Suryawanshi, R., Yadavalli, T., Mishra, Y. K., Adelung, R., & Shukla, D. (2019). An intra-vaginal zinc oxide tetrapod nanoparticles (zoten) and genital herpesvirus cocktail can provide a novel platform for live virus vaccine. Frontiers in Immunology. https://doi.org/10.3389/fimmu.2019.00500.
Siddiqui, S. A., Rashid, M. M. O., Uddin, M. G., Robel, F. N., Hossain, M. S., Haque, M. A., & Jakaria, M. (2020). Biological efficacy of zinc oxide nanoparticles against diabetes: A preliminary study conducted in mice. Bioscience Reports. https://doi.org/10.1042/BSR20193972.
Post-treatment flare-up incidence after using nano zinc oxide eugenol sealer in mandibular first molars with irreversible pulpitis. (2019). Iranian Registry of Clinical Trials. Retrieved from https://www.irct.ir/trial/36542
Mosavi, S. A., Gottasloo, R., Akbarzadeh, A., Sadighi, S., & Khoramdel, A. (2018). Evaluation of antibacterial and anti-fungal properties of adding zno nanoparticles to tissue conditioner used in complete denture. In Iranian Registry of Clinical Trials Retrieved from https://en.irct.ir/trial/38575.
Javidi, M., Javidi, M., Zarei, M., Ashrafpour, E., Gharechahi, M., & Bagheri, H. (2020). Post-treatment flare-up incidence after using nano zinc oxide eugenol sealer -IRCT20190103042224N1 in mandibular first molars with irreversible pulpitis. J Dent Shiraz Univ Med Scien, 4, 1–7.
Mohapatra, S. S., & Limayem, A. (2020). Chitosan oligomer and zinc oxide nanoparticle compositions for treating drug resistant bacteria and biofilm. Retrieved from https://worldwide.espacenet.com/patent/search/family/064953833/publication/US10576100B2?q=US10576100B2
Feng, L. B., Yong, L., Yu, L., & LiXia, Z. (2020). Drug-loaded metal nanoparticle/chitosan controlled-release hydrogel, and preparation method and application thereof. Retrieved from https://worldwide.espacenet.com/patent/search/family/072383495/publication/CN111658816A?q=CN111658816A
Kou, L., Lin, M. T., Lu, Cui Tao, Xu, H. L., Yang, Y., Yao, Q., … Zhao, Y. Z. (2020). Disulfiram nano-particle, and preparation method and application thereof. Retrieved from https://worldwide.espacenet.com/patent/search/family/069224594/publication/CN110721318A?q=CN110721318A
Liu, R., Shen, Q., Yang, J., & Zhang, X. (2020). Zinc oxide and antigen co-loading medicine nanometer vaccine, and preparation method and application thereof. Retrieved from https://worldwide.espacenet.com/patent/search/family/069338909/publication/CN110755607A?q = CN110755607A
Chen, H., Dong, Y., He, J., & Wu, S. (2020). Quantum dot light emitting devices (QLEDs) and method of manufacture. Retrieved from https://worldwide.espacenet.com/patent/search/family/065898143/publication/US10593902B2?q=US10593902B2
Sun, W., Sun, Y., & Yang, X. (2019). Preparation method of drug-loaded zinc oxide and silicon dioxide composite nanoparticles. Retrieved from https://worldwide.espacenet.com/patent/search/family/065799263/publication/CN109512800A?q=CN109512800A
Choi, H. A., Cho, N. H., Ham, W. S., Kim, Y. K., Lee, J A E Won, Park, B. C., & Sharma, P. (2019). Delivery of biomacromolecules employing clusters of nanowires. Retrieved from https://worldwide.espacenet.com/patent/search/family/065759731/publication/KR20190027346A?q = KR20190027346A
Eric, B., Wang, C., & Wu, M. (2019). Compositions and methods for treating striated muscle injury, treating striated muscle atrophy and/or for promoting striated muscle growth. Retrieved from https://worldwide.espacenet.com/patent/search/family/061902088/publication/US10398732B2?q=US10398732B2
Carlsson, J.-O., Johansson, A., & Rooth, M. (2019). Solid nanoparticle with inorganic coating.
Ai, F., Cao, C., Li, W., Liu, D., Wang, X., Xin, Y., & Zhou, K. (2019). High molecular composite material for antibacterial medical catheter and preparation method of material. Retrieved from https://worldwide.espacenet.com/patent/search/family/067013778/publication/CN109939267A?q=CN109939267A
Vanepps, J. S., & Kotov, N. A. (2019). Antimicrobial and enzyme inhibitory zinc oxide nanoparticles.
XianFeng, G., Li, Q., & Zhao, X. (2018). Preparation method for anticancer drug nanoparticles.
Yoen, K. Do. (2018). The composition for catheter the central venous catheter and the intravascular tube catheter prepared by using the same.
PeiYang, Y. (2018). Medical high-efficient sterilizing mask.
Xiong, H., & Zhao, W. (2017). Self-assembled nano zinc oxide drug-loading capsule, and preparation method and application thereof.
Li, Y., Liu, X., Tan, L., Wu, S., & Xu, Z. (2017). Preparation method for hybridized biological functional coating based on halogen-ammonia compound and zinc oxide nanoparticles.
Bian, J., Chen, L., Guo, S., Liu, J., Song, C., Zhang, R., & Zheng, Y. (2017). Flexible electroluminescent X-ray image memory display panel.
Mao, C., Liu, X., Zhu, Y., Wu, S., & Xu, Z. (2017). Method for preparing nano-silver/zinc oxide composite hydrogel dressing with surface antibacterial properties.
Andrea, G., Gloria, H. A., Daniel, J., Vit, M., Jiri, M., Kristina, N., … Vladimir, V. (2017). Antitumor composition based on hyaluronic acid and inorganic nanoparticles method of preparation thereof and use thereof.
Liang, X., Liu, J., Ma, X., & Zhong, L. (2016). Zinc oxide-based nano-drug composition, and preparation method and application thereof.
Gao, J., Jiang, L., Li, C., Liu, Z., Mou, J., Pei, D., … Zhang, Y. (2016). Slow release gel loaded with sanqi and nanometer zinc oxide, preparing method thereof and applications of the slow-release gel.
Matouk, M. M. (2016). Silver-copper-zinc oxide wound care system.
Jiang, T., Sun, G., Wang, H., Xu, X., Yang, S., & Zhu, L. (2016). Three-dimension gene-tree biological chip and manufacturing method thereof.
Li, B., Li, L., Shan, C., & Song, D. (2015). Preparing method of antibiosis antiultraviolet contact lenses.
Sahu, Y. S. (2016). Nano zinc oxide market by application (Paints & Coatings, Cosmetics, and Others) - global opportunity analysis and industry forecast, 2014-2022. Allied Market Research.
Mohd Yusof, H., Mohamad, R., Zaidan, U. H., & Abdul Rahman, N. A. (2019). Microbial synthesis of zinc oxide nanoparticles and their potential application as an antimicrobial agent and a feed supplement in animal industry: A review. Journal of Animal Science and Biotechnology. https://doi.org/10.1186/s40104-019-0368-z.
Morris, A. S., & Salem, A. K. (2017). Nanotoxicity. In Nanomedicine for Inflammatory Diseases (pp. 67–79). https://doi.org/10.1201/978131515235.
Hao, L., Chen, L., Hao, J., & Zhong, N. (2013). Bioaccumulation and sub-acute toxicity of zinc oxide nanoparticles in juvenile carp (Cyprinus carpio): A comparative study with its bulk counterparts. Ecotoxicology and Environmental Safety, 91, 52–60. https://doi.org/10.1016/j.ecoenv.2013.01.007.
Srivastav, A. K., Kumar, M., Ansari, N. G., Jain, A. K., Shankar, J., Arjaria, N., et al. (2016). A comprehensive toxicity study of zinc oxide nanoparticles versus their bulk in Wistar rats. Human and Experimental Toxicology, 35(12), 1286–1304. https://doi.org/10.1177/0960327116629530.
Qiao, Y., Liang, X., Yan, Y., Lu, Y., Zhang, D., Yao, W., et al. (2018). Identification of exosomal miRNAs in rats with pulmonary neutrophilic inflammation induced by zinc oxide nanoparticles. Frontiers in Physiology, 9(MAR). https://doi.org/10.3389/fphys.2018.00217.
Yoo, J., Seo, G. B., Yoon, B. I., Lim, Y. M., Kim, P., Kim, H. M., & Kwon, J. T. (2018). Evaluation of recovery from acute lung injury induced by intratracheal instillation of zinc oxide nanoparticles. Applied Ecology and Environmental Research, 16(3), 3145–3157. https://doi.org/10.15666/aeer/1603_31453157.
Pan, C. H., Chuang, K. J., Chen, J. K., Hsiao, T. C., Lai, C. H., Jones, T. P., et al. (2015). Characterization of pulmonary protein profiles in response to zinc oxide nanoparticles in mice: A 24-hour and 28-day follow-up study. International Journal of Nanomedicine, 10, 4705–4716. https://doi.org/10.2147/IJN.S82979.
Gosens, I., Kermanizadeh, A., Jacobsen, N. R., Lenz, A. G., Bokkers, B., De Jong, W. H., et al. (2015). Comparative hazard identification by a single dose lung exposure of zinc oxide and silver nanomaterials in mice. PLoS ONE, 10(5). https://doi.org/10.1371/journal.pone.0126934.
Wang, D., Li, H., Liu, Z., Zhou, J., & Zhang, T. (2017). Acute toxicological effects of zinc oxide nanoparticles in mice after intratracheal instillation. International Journal of Occupational and Environmental Health, 23(1), 11–19. https://doi.org/10.1080/10773525.2016.1278510.
Larsen, S. T., Jackson, P., Poulsen, S. S., Levin, M., Jensen, K. A., Wallin, H., et al. (2016). Airway irritation, inflammation, and toxicity in mice following inhalation of metal oxide nanoparticles. Nanotoxicology, 10(9), 1254–1262. https://doi.org/10.1080/17435390.2016.1202350.
Chen, J. K., Ho, C. C., Chang, H., Lin, J. F., Yang, C. S., Tsai, M. H., et al. (2015). Particulate nature of inhaled zinc oxide nanoparticles determines systemic effects and mechanisms of pulmonary inflammation in mice. Nanotoxicology, 9(1), 43–53. https://doi.org/10.3109/17435390.2014.886740.
Saptarshi, S. R., Feltis, B. N., Wright, P. F. A., & Lopata, A. L. (2015). Investigating the immunomodulatory nature of zinc oxide nanoparticles at sub-cytotoxic levels in vitro and after intranasal instillation in vivo. Journal of Nanobiotechnology, 13(1). https://doi.org/10.1186/s12951-015-0067-7.
Huang, K. L., Lee, Y. H., Chen, H. I., Liao, H. S., Chiang, B. L., & Cheng, T. J. (2015). Zinc oxide nanoparticles induce eosinophilic airway inflammation in mice. Journal of Hazardous Materials, 297, 304–312. https://doi.org/10.1016/j.jhazmat.2015.05.023.
Harkema, J. R., Nikula, K. J., & Haschek, W. M. (2013). Respiratory system. In Haschek and Rousseaux’s Handbook of Toxicologic Pathology. https://doi.org/10.1016/B978-0-12-415759-0.00051-0.
Hendrickson, C. M., Abbott, J., Zhuo, H., Liu, K. D., Calfee, C. S., & Matthay, M. A. (2017). Higher mini-BAL total protein concentration in early ARDS predicts faster resolution of lung injury measured by more ventilator-free days. American Journal of Physiology - Lung Cellular and Molecular Physiology. https://doi.org/10.1152/ajplung.00381.2016.
Drent, M., Cobben, N. A. M., Henderson, R. F., Wouters, E. F. M., & Van Dieijen-Visser, M. (1996). Usefulness of lactate dehydrogenase and its isoenzymes as indicators of lung damage or inflammation. European Respiratory Journal. https://doi.org/10.1183/09031936.96.09081736.
Larsen, S. T., Da Silva, E., Hansen, J. S., Jensen, A. C. Ø., Koponen, I. K., & Sørli, J. B. (2020). Acute inhalation toxicity after inhalation of ZnO nanoparticles: Lung surfactant function inhibition in vitro correlates with reduced tidal volume in mice. International Journal of Toxicology. https://doi.org/10.1177/1091581820933146.
Lin, Y. F., Chiu, I. J., Cheng, F. Y., Lee, Y. H., Wang, Y. J., Hsu, Y. H., & Chiu, H. W. (2016). The role of hypoxia-inducible factor-1α in zinc oxide nanoparticle-induced nephrotoxicity in vitro and in vivo. Particle and Fibre Toxicology, 13(1). https://doi.org/10.1186/s12989-016-0163-3.
Chien, C. C., Yan, Y. H., Juan, H. T., Cheng, T. J., Liao, J. B., Lee, H. P., & Wang, J. S. (2017). Sustained renal inflammation following 2 weeks of inhalation of occupationally relevant levels of zinc oxide nanoparticles in Sprague Dawley rats. Journal of Toxicologic Pathology, 30(4), 307–314. https://doi.org/10.1293/tox.2017-0025.
Khorsandi, L., Heidari-Moghadam, A., & Jozi, Z. (2018). Nephrotoxic effects of low-dose zinc oxide nanoparticles in rats. Journal of Nephropathology, 7(3), 158–165. https://doi.org/10.15171/jnp.2018.35.
Kausar, S., Jabeen, F., Sultana, S., & Rasul, A. (2018). Zinc oxide nanoparticles (ZnO NPs) induced nephrotoxicity in male sprague dawley rats. International Journal of Biosciences (IJB), 13(05), 457–463. https://doi.org/10.12692/ijb/13.5.457-463.
Alferah, M. A. Z. (2018). Renal toxicity of zinc oxide nanoparticles (ZnONPs) of male westar rats. International Journal of Science and Research (IJSR), 7(2), 1092–1097. https://doi.org/10.21275/16021801.
Salman, R. A. (2018). Histopathological effect of zinc oxide nanoparticles on kidney and liver tissues in albino male mice. Ibn AL- Haitham Journal For Pure and Applied Science, 31(1), 9. https://doi.org/10.30526/31.1.1844.
Abdel-Aziz, H., Hamdan, H., & Ragab, E. (2018). The histological effects of zinc oxide nanoparticles on the kidney of adult male rabbits. Sohag Medical Journal, 22(2), 297–301. https://doi.org/10.21608/smj.2018.40959.
Takemoto, Y., & Naganuma, T. (2012). Kidney function tests. In Japanese Journal of Clinical Urology (Vol. 66, pp. 274–278). https://doi.org/10.5005/jp/books/13014_26.
Hegazy, A. A., Ahmed, A., Shehata, M. A., & Abdelfattah, M. M. (2018). Changes in rats’ liver structure induced by zinc oxide nanoparticles and the possible protective role of vitamin E. International Journal of Human Anatomy, 1(3), 1–16. https://doi.org/10.14302/issn.2577-2279.ijha-18-2384.
Mansouri, E., Khorsandi, L., Orazizadeh, M., & Jozi, Z. (2015). Dose-dependent hepatotoxicity effects of Zinc oxide nanoparticles. Nanomed. J.
Almansour, M. I., Alferah, M. A., Shraideh, Z. A., & Jarrar, B. M. (2017). Zinc oxide nanoparticles hepatotoxicity: Histological and histochemical study. Environmental Toxicology and Pharmacology, 51, 124–130. https://doi.org/10.1016/j.etap.2017.02.015.
Alferah, M. A. Z. (2018). Histological Changes of malewestarrats liver following the ingestion of zinc oxide nanoparticles with special emphasis on the histochemical alterations. Journal of Histology and Histopathology, 5(1), 4. https://doi.org/10.7243/2055-091x-5-4.
Tang, H. Q., Xu, M., Rong, Q., Jin, R. W., Liu, Q. J., & Li, Y. L. (2016). The effect of ZnO nanoparticles on liver function in rats. International Journal of Nanomedicine, 11, 4275–4285. https://doi.org/10.2147/IJN.S109031.
Shahzad, K., Khan, M. N., Jabeen, F., Kosour, N., Chaudhry, A. S., Sohail, M., & Ahmad, N. (2019). Toxicity of zinc oxide nanoparticles (ZnO-NPs) in tilapia (Oreochromis mossambicus): tissue accumulation, oxidative stress, histopathology and genotoxicity. International Journal of Environmental Science and Technology, 16(4), 1973–1984. https://doi.org/10.1007/s13762-018-1807-7.
Palani, S., Murugesan, M., Bukhari, A. S., & Mohamed, H. E. S. (2015). Morphological and liver histological effects of ZnO. Suganthi P et.al JOAASR.
Murali, A. R., & W. D. C. (2017). Liver test interpretation - approach to the patient with liver disease: A guide to commonly used liver tests. Disease Management.
Goessling, W., & Stainier, D. Y. (2016). Endoderm specification and liver development. Methods in Cell Biology. https://doi.org/10.1016/bs.mcb.2016.03.042.
Luster, M. I., Simeonova, P. P., Gallucci, R. M., Bruccoleri, A., Blazka, M. E., & Yucesoy, B. (2001). Role of inflammation in chemical-induced hepatotoxicity. In Toxicology Letters. https://doi.org/10.1016/S0378-4274(01)00284-3.
de Souza, J. M., de Mendes, B. O., Guimarães, A. T. B., de Rodrigues, A. S. L., Chagas, T. Q., Rocha, T. L., & Malafaia, G. (2018). Zinc oxide nanoparticles in predicted environmentally relevant concentrations leading to behavioral impairments in male swiss mice. Science of the Total Environment, 613–614, 653–662. https://doi.org/10.1016/j.scitotenv.2017.09.051.
Attia, H., Nounou, H., & Shalaby, M. (2018). Zinc oxide nanoparticles induced oxidative DNA damage, inflammation and apoptosis in rat’s brain after oral exposure. Toxics, 6(2). https://doi.org/10.3390/toxics6020029.
Aijie, C., Huimin, L., Jia, L., Lingling, O., Limin, W., Junrong, W., et al. (2017). Central neurotoxicity induced by the instillation of ZnO and TiO nanoparticles through the taste nerve pathway. Nanomedicine, 12(20), 2453–2470. https://doi.org/10.2217/nnm-2017-0171.
Liang, H., Chen, A., Lai, X., Liu, J., Wu, J., Kang, Y., et al. (2018). Neuroinflammation is induced by tongue-instilled ZnO nanoparticles via the Ca2+-dependent NF-κB and MAPK pathways. Particle and Fibre Toxicology, 15(1). https://doi.org/10.1186/s12989-018-0274-0.
Salim, S. (2017). Oxidative stress and the central nervous system. Journal of Pharmacology and Experimental Therapeutics. https://doi.org/10.1124/jpet.116.237503.
Sayre, L. M., Perry, G., & Smith, M. A. (2008). Oxidative stress and neurotoxicity. Chemical Research in Toxicology. https://doi.org/10.1021/tx700210j.
Ortiz, G. G., González-Usigli, H., Pacheco-Moisés, F. P., Mireles-Ramírez, M. A., Sánchez-López, A. L., Torres-Sánchez, E. D., et al. (2017). Physiology and pathology of neuroimmunology: role of inflammation in Parkinson’s disease. In Physiology and Pathology of Immunology. https://doi.org/10.5772/intechopen.70377.
Liu, H., Yang, H., Fang, Y., Li, K., Tian, L., Liu, X., et al. (2020). Neurotoxicity and biomarkers of zinc oxide nanoparticles in main functional brain regions and dopaminergic neurons. Science of the Total Environment, 705. https://doi.org/10.1016/j.scitotenv.2019.135809.
Fogal, B., & Hewett, S. J. (2008). Interleukin-1β: A bridge between inflammation and excitotoxicity? Journal of Neurochemistry. https://doi.org/10.1111/j.1471-4159.2008.05315.x.
Giovannoni, G. (2014). Cerebrospinal fluid analysis. In Handbook of Clinical Neurology. https://doi.org/10.1016/B978-0-444-52001-2.00029-7.
Gallo, V., & Deneen, B. (2014). Glial development: The crossroads of regeneration and repair in the CNS. Neuron. https://doi.org/10.1016/j.neuron.2014.06.010.
Chuang, H. C., Yang, Y. T., Chen, H. C., Hwang, Y. H., Wu, K. Y., Chen, T. F., et al. (2020). Acute effects of pulmonary exposure to zinc oxide nanoparticles on the brain in vivo. Aerosol and Air Quality Research. https://doi.org/10.4209/aaqr.2019.10.0523.
Llorens, F., Villar-Piqué, A., Candelise, N., Ferrer, I., & Zerr, I. (2019). Tau protein as a biological fluid biomarker in neurodegenerative dementias. In Cognitive Disorders. https://doi.org/10.5772/intechopen.73528.
Yaqub, A., Faheem, I., Anjum, K. M., Ditta, S. A., Yousaf, M. Z., Tanvir, F., & Raza, C. (2020). Neurotoxicity of ZnO nanoparticles and associated motor function deficits in mice. Applied Nanoscience (Switzerland), 10(1), 177–185. https://doi.org/10.1007/s13204-019-01093-3.
Sood, K., Kaur, J., & Khatri, M. (2017). comparative neurotoxicity evaluation of zinc oxide nanoparticles by crawling assay on Drosophila melanogaster. International Journal of Engineering Technology Science and Research, 4(4), 440–444.
Elshama, S. S., El-Kenawy, A. E. M., & Osman, H. E. H. (2017). Histopathological study of zinc oxide nanoparticle-induced neurotoxicity in rats. Current Topics in Toxicology, 13, 95–103.
Milivojević, T., Glavan, G., Božič, J., Sepčić, K., Mesarič, T., & Drobne, D. (2015). Neurotoxic potential of ingested ZnO nanomaterials on bees. Chemosphere, 120, 547–554. https://doi.org/10.1016/j.chemosphere.2014.07.054.
Hosseini, S. M., Moshrefi, A. H., Amani, R., Razavimehr, S. V., Aghajanikhah, M. H., Sokouti, Z., & Holari, B. B. (2019). Subchronic effects of different doses of zinc oxide nanoparticle on reproductive organs of female rats: An experimental study. International Journal of Reproductive BioMedicine, 17(2), 107–118. https://doi.org/10.18502/ijrm.v17i2.3988.
Husain, W. M., Araak, J. K., & Ibrahim, O. b. M. S. (2019). Effect of different doses from ZnONPS on the pituitary-testes axis function in adult male rats. Advances in Animal and Veterinary Sciences, 7(7), 550–556. https://doi.org/10.17582/journal.aavs/2019/7.7.550.556.
Yousef, M. I., Al-Hamadani, M. Y. I., & Kamel, M. A. (2019). Reproductive toxicity of aluminum oxide nanoparticles and zinc oxide nanoparticles in male rats. Nanoparticle, 1(1), 3.
Tang, Y., Chen, B., Hong, W., Chen, L., Yao, L., Zhao, Y., et al. (2019). ZnO nanoparticles induced male reproductive toxicity based on the effects on the endoplasmic reticulum stress signaling pathway. International Journal of Nanomedicine, 14, 9563–9576. https://doi.org/10.2147/IJN.S223318.
Hancock, J. L. (1951). A staning tecknique for the study of temperature shock in semen. Nature, 167, 323–325.
Krzanowska, H. (1982). Sperm quantity in bred lines of mice and their crosses. Acta. Boil, 5, 279–290.
Radhi, M. J., & Al-Bairuty, G. (2019). Effect of zinc oxide nanoparticles (ZnO-NPs) on weights of some reproductive organs and sperm abnormalities in the tail of epididymis of albino mice. Pharm. Sci. & Res., 11(1), 243–246 Retrieved from https://www.researchgate.net/publication/330823862.
Kong, T., Zhang, S. H., Zhang, C., Zhang, J. L., Yang, F., Wang, G. Y., et al. (2020). The effects of 50 nm unmodified nano-ZnO on lipid metabolism and semen quality in male mice. Biological Trace Element Research, 194(2), 432–442. https://doi.org/10.1007/s12011-019-01792-6.
Han, Z., Yan, Q., Ge, W., Liu, Z. G., Gurunathan, S., De Felici, M., et al. (2016). Cytotoxic effects of ZnO nanoparticles on mouse testicular cells. International Journal of Nanomedicine, 11, 5187–5203. https://doi.org/10.2147/IJN.S111447.
Mozaffari, Z., Parivar, K., Roodbari, N. H., & Irani, S. (2015). Histopathological evaluation of the toxic effects of zinc oxide (ZnO) nanoparticles on testicular tissue of NMRI adult mice. Advanced Studies in Biology, 7, 275–291. https://doi.org/10.12988/asb.2015.5425.
Liu, X. Q., Zhang, H. F., Zhang, W. D., Zhang, P. F., Hao, Y. N., Song, R., et al. (2016). Regulation of neuroendocrine cells and neuron factors in the ovary by zinc oxide nanoparticles. Toxicology Letters, 256, 19–32. https://doi.org/10.1016/j.toxlet.2016.05.007.
Alkaladi, A., Afifi, M., Ali, H., & Saddick, S. (2020). Hormonal and molecular alterations induced by sub-lethal toxicity of zinc oxide nanoparticles on Oreochromis niloticus. Saudi Journal of Biological Sciences, 27(5), 1296–1301. https://doi.org/10.1016/j.sjbs.2020.01.010.
Deepa, S., Murugananthkumar, R., Raj Gupta, Y., Gowda K.S, M., & Senthilkumaran, B. (2019). Effects of zinc oxide nanoparticles and zinc sulfate on the testis of common carp, Cyprinus carpio. Nanotoxicology, 13(2), 240–257. doi:https://doi.org/10.1080/17435390.2018.1541259
O’Donnell, B., Huo, L., Polli, J. R., Qiu, L., Collier, D. N., Zhang, B., & Pan, X. (2017). ZnO Nanoparticles enhanced germ cell apoptosis in Caenorhabditis elegans, in comparison with ZnCl2. Toxicological Sciences, 156(2), 336–343. https://doi.org/10.1093/toxsci/kfw258.
Adam, N., Vergauwen, L., Blust, R., & Knapen, D. (2015). Gene transcription patterns and energy reserves in Daphnia magna show no nanoparticle specific toxicity when exposed to ZnO and CuO nanoparticles. Environmental Research, 138, 82–92. https://doi.org/10.1016/j.envres.2015.02.014.
Jo, E., Seo, G., Kwon, J. T., Lee, M., & Lee, B. cheun, Eom, I., … Choi, K. (2013). Exposure to zinc oxide nanoparticles affects reproductive development and biodistribution in offspring rats. Journal of Toxicological Sciences. https://doi.org/10.2131/jts.38.525.
Willhite, C. C., & Mirkes, P. E. (2014). Developmental toxicology. In Encyclopedia of Toxicology: Third Edition. https://doi.org/10.1016/B978-0-12-386454-3.00014-2.
Cordier, S. (2008). Evidence for a role of paternal exposures in developmental toxicity. In Basic and Clinical Pharmacology and Toxicology. https://doi.org/10.1111/j.1742-7843.2007.00162.x.
Aikawa, N. (2020). A novel screening test to predict the developmental toxicity of drugs using human induced pluripotent stem cells. Journal of Toxicological Sciences. https://doi.org/10.2131/jts.45.187.
Teng, C., Jia, J., Wang, Z., Sharma, V. K., & Yan, B. (2019). Size-dependent maternal-fetal transfer and fetal developmental toxicity of ZnO nanoparticles after oral exposures in pregnant mice. Ecotoxicology and Environmental Safety, 182. https://doi.org/10.1016/j.ecoenv.2019.109439.
Chen, B., Hong, W., Yang, P., Tang, Y., Zhao, Y., Aguilar, Z. P., & Xu, H. (2020). Nano zinc oxide induced fetal mice growth restriction, based on oxide stress and endoplasmic reticulum stress. Nanomaterials, 10(2). https://doi.org/10.3390/nano10020259.
Manzo, S., Schiavo, S., Oliviero, M., Toscano, A., Ciaravolo, M., & Cirino, P. (2017). Immune and reproductive system impairment in adult sea urchin exposed to nanosized ZnO via food. Science of the Total Environment, 599–600, 9–13. https://doi.org/10.1016/j.scitotenv.2017.04.173.
Verma, S. K., Panda, P. K., Jha, E., Suar, M., & Parashar, S. K. S. (2017). Altered physiochemical properties in industrially synthesized ZnO nanoparticles regulate oxidative stress; Induce in vivo cytotoxicity in embryonic zebrafish by apoptosis. Scientific Reports, 7(1). https://doi.org/10.1038/s41598-017-14039-y.
Zhao, X., Wang, S., Wu, Y., You, H., & Lv, L. (2013). Acute ZnO nanoparticles exposure induces developmental toxicity, oxidative stress and DNA damage in embryo-larval zebrafish. Aquatic Toxicology, 136–137, 49–59. https://doi.org/10.1016/j.aquatox.2013.03.019.
Bai, W., Zhang, Z., Tian, W., He, X., Ma, Y., Zhao, Y., & Chai, Z. (2010). Toxicity of zinc oxide nanoparticles to zebrafish embryo: A physicochemical study of toxicity mechanism. Journal of Nanoparticle Research, 12(5), 1645–1654. https://doi.org/10.1007/s11051-009-9740-9.
Choi, J. S., Kim, R. O., Yoon, S., & Kim, W. K. (2016). Developmental toxicity of zinc oxide nanoparticles to zebrafish (Danio rerio): A transcriptomic analysis. PLoS ONE, 11(8). https://doi.org/10.1371/journal.pone.0160763.
Cong, Y., Jin, F., Wang, J., & Mu, J. (2017). The embryotoxicity of ZnO nanoparticles to marine medaka, Oryzias melastigma. Aquatic Toxicology, 185, 11–18. https://doi.org/10.1016/j.aquatox.2017.01.006.
Yan, Y., Wang, G., Huang, J., Zhang, Y., Cheng, X., Chuai, M., et al. (2020). Zinc oxide nanoparticles exposure-induced oxidative stress restricts cranial neural crest development during chicken embryogenesis. Ecotoxicology and Environmental Safety. https://doi.org/10.1016/j.ecoenv.2020.110415.
Xiaoli, F., Junrong, W., Xuan, L., Yanli, Z., Limin, W., Jia, L., & Longquan, S. (2017). Prenatal exposure to nanosized zinc oxide in rats: Neurotoxicity and postnatal impaired learning and memory ability. Nanomedicine, 12(7), 777–795. https://doi.org/10.2217/nnm-2016-0397.
Lee, J., Yu, W. J., Song, J., Sung, C., Jeong, E. J., Han, J. S., et al. (2016). Developmental toxicity of intravenously injected zinc oxide nanoparticles in rats. Archives of Pharmacal Research, 39(12), 1682–1692. https://doi.org/10.1007/s12272-016-0767-z.
Xiang, L., Zhao, H. M., Li, Y. W., Huang, X. P., Wu, X. L., Zhai, T., et al. (2015). Effects of the size and morphology of zinc oxide nanoparticles on the germination of Chinese cabbage seeds. Environmental Science and Pollution Research, 22(14), 10452–10462. https://doi.org/10.1007/s11356-015-4172-9.
Liu, J., Zhao, Y., Ge, W., Zhang, P., Liu, X., Zhang, W., et al. (2017). Oocyte exposure to ZnO nanoparticles inhibits early embryonic development through the γ-H2AX and NF-κB signaling pathways. Oncotarget, 8(26), 42673–42692. https://doi.org/10.18632/oncotarget.17349.
Zhao, X., Ren, X., Zhu, R., Luo, Z., & Ren, B. (2016). Zinc oxide nanoparticles induce oxidative DNA damage and ROS-triggered mitochondria-mediated apoptosis in zebrafish embryos. Aquatic Toxicology, 180, 56–70. https://doi.org/10.1016/j.aquatox.2016.09.013.
Li, J., Chen, Z., Huang, R., Miao, Z., Cai, L., & Du, Q. (2018). Toxicity assessment and histopathological analysis of nano-ZnO against marine fish (Mugilogobius chulae) embryos. Journal of Environmental Sciences (China), 73, 78–88. https://doi.org/10.1016/j.jes.2018.01.015.
Choi, J. S., Song, J., Yoon, S., & Kim, W. K. (2018). Predicting ZnO nanoparticle and ZnSO4 toxicity from zebrafish embryo gene expression. Molecular and Cellular Toxicology, 14(1), 19–26. https://doi.org/10.1007/s13273-018-0003-7.
Morgalev, Y. N., Gosteva, I. A., Morgaleva, T. G., Morgalev, S. Y., Kostenko, E. V., & Kudryavtsev, B. A. (2018). Parameters of embryogenesis in zebrafish Danio rerio as indicators of the ecological toxicity of zinc oxide nanoparticles. Nanotechnologies in Russia, 13(5–6), 311–316. https://doi.org/10.1134/S1995078018030114.
Oliviero, M., Schiavo, S., Rametta, G., Miglietta, M. L., & Manzo, S. (2017). Different sizes of ZnO diversely affected the cytogenesis of the sea urchin Paracentrotus lividus. Science of the Total Environment, 607–608, 176–183. https://doi.org/10.1016/j.scitotenv.2017.07.038.
Bonfanti, P., Moschini, E., Saibene, M., Bacchetta, R., Rettighieri, L., Calabri, L., et al. (2015). Do nanoparticle physico-chemical properties and developmental exposure window influence nano ZnO embryotoxicity in Xenopus laevis? International Journal of Environmental Research and Public Health, 12(8), 8828–8848. https://doi.org/10.3390/ijerph120808828.
Saliani, M., Jalal, R., & Goharshadi, E. K. (2016). Mechanism of oxidative stress involved in the toxicity of ZnO nanoparticles against eukaryotic cells. Nanomedicine journal, 3(1), 1–14. https://doi.org/10.7508/nmj.2016.01.001.
Handa, N., Bhardwaj, R., Kaur, H., Poonam, Kapoor D, Rattan, A., … Kapoor, N. (2015). Selenium: An antioxidative protectant in plants under stress. In Plant Metal Interaction: Emerging Remediation Techniques. doi:https://doi.org/10.1016/B978-0-12-803158-2.00007-2
Liu, Z., Ren, Z., Zhang, J., Chuang, C. C., Kandaswamy, E., Zhou, T., & Zuo, L. (2018). Role of ROS and nutritional antioxidants in human diseases. Frontiers in Physiology. https://doi.org/10.3389/fphys.2018.00477.
Pinho, A. R., Rebelo, S., & de Lourdes Pereira, M. (2020). The impact of zinc oxide nanoparticles on male (In)fertility. Materials. https://doi.org/10.3390/ma13040849.
Hammond, B., & Hess, M. L. (1985). The oxygen free radical system: Potential mediator of myocardial injury. Journal of the American College of Cardiology, 6(1), 215–220. https://doi.org/10.1016/S0735-1097(85)80278-3.
Siddique, T., Deng, H. X., & Ajroud-Driss, S. (2013). Chapter 132 – Motor neuron disease. In Emery and Rimoin’s Principles and Practice of Medical Genetics. https://doi.org/10.1016/B978-0-12-383834-6.00141-5.
Jeeva, J. S., Sunitha, J., Ananthalakshmi, R., Rajkumari, S., Ramesh, M., & Krishnan, R. (2015). Enzymatic antioxidants and its role in oral diseases. Journal of Pharmacy and Bioallied Sciences. https://doi.org/10.4103/0975-7406.163438.
Ighodaro, O. M., & Akinloye, O. A. (2018). First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria Journal of Medicine, 54(4), 287–293. https://doi.org/10.1016/j.ajme.2017.09.001.
Nandi, A., Yan, L. J., Jana, C. K., & Das, N. (2019). Role of catalase in oxidative stress- and age-associated degenerative diseases. Oxidative Medicine and Cellular Longevity. https://doi.org/10.1155/2019/9613090.
A.H., M., A., Q., I., Q., A.H., N., & R., B. (2020). Accumulation and trafficking of zinc oxide nanoparticles in an invertebrate model, Bombyx mori, with insights on their effects on immuno-competent cells. Scientific reports.
Abdel-Halim, K. Y., Osman, S. R., & Abdou, G. Y. (2020). In vivo evaluation of oxidative stress and biochemical alteration as biomarkers in glass clover snail, Monacha cartusiana exposed to zinc oxide nanoparticles. Environmental Pollution. https://doi.org/10.1016/j.envpol.2019.113120.
Chang, Y. T., Chang, W. N., Tsai, N. W., Huang, C. C., Te Kung, C., Su, Y. J., et al. (2014). The roles of biomarkers of oxidative stress and antioxidant in alzheimer’s disease: A systematic review. BioMed Research International. https://doi.org/10.1155/2014/182303.
Saxena, R. (2014). Arthritis as a disease of aging and changes in antioxidant status. In Aging: Oxidative Stress and Dietary Antioxidants. https://doi.org/10.1016/B978-0-12-405933-7.00005-6.
Fuchs, P., Perez-Pinzon, M. A., & Dave, K. R. (2013). Cerebral ischemia in diabetics and oxidative stress. In Diabetes: Oxidative Stress and Dietary Antioxidants (pp. 15–23). https://doi.org/10.1016/B978-0-12-405885-9.00002-4.
Ayala, A., Muñoz, M. F., & Argüelles, S. (2014). Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxidative Medicine and Cellular Longevity. https://doi.org/10.1155/2014/360438.
Ma, B., Villalta, P. W., Balbo, S., & Stepanov, I. (2014). Analysis of a malondialdehyde-deoxyguanosine adduct in human leukocyte DNA by liquid chromatography nanoelectrospray-high-resolution tandem mass spectrometry. Chemical Research in Toxicology. https://doi.org/10.1021/tx5002699.
Cadet, J., Douki, T., Gasparutto, D., & Ravanat, J. L. (2003). Oxidative damage to DNA: Formation, measurement and biochemical features. In Mutation Research - Fundamental and Molecular Mechanisms of Mutagenesis. https://doi.org/10.1016/j.mrfmmm.2003.09.001.
Zhai, Q. Y., Ge, W., Wang, J. J., Sun, X. F., Ma, J. M., Liu, J. C., et al. (2018). Exposure to zinc oxide nanoparticles during pregnancy induces oocyte DNA damage and affects ovarian reserve of mouse offspring. Aging, 10(8), 2170–2189. https://doi.org/10.18632/aging.101539.
Pati, R., Das, I., Mehta, R. K., Sahu, R., & Sonawane, A. (2016). Zinc-oxide nanoparticles exhibit genotoxic, clastogenic, cytotoxic and actin depolymerization effects by inducing oxidative stress responses in macrophages and adult mice. Toxicological Sciences. https://doi.org/10.1093/toxsci/kfw010.
Obaidul Islam, M., Bacchetti, T. T., & Ferretti, G. (2019). Alterations of antioxidant enzymes and biomarkers of nitro-oxidative stress in tissues of bladder cancer. Oxidative Medicine and Cellular Longevity. https://doi.org/10.1155/2019/2730896.
Qing, X., Shi, D., Lv, X., Wang, B., Chen, S., & Shao, Z. (2019). Prognostic significance of 8-hydroxy-2′-deoxyguanosine in solid tumors: A meta-analysis. BMC Cancer. https://doi.org/10.1186/s12885-019-6189-9.
Abass, M. A., Selim, S. A., Selim, A. O., El-Shal, A. S., & Gouda, Z. A. (2017). Effect of orally administered zinc oxide nanoparticles on albino rat thymus and spleen. IUBMB Life. https://doi.org/10.1002/iub.1638.
Ma, D. D., & Yang, W. X. (2016). Engineered nanoparticles induce cell apoptosis: Potential for cancer therapy. Oncotarget. https://doi.org/10.18632/oncotarget.8553.
Bai, D. P., Zhang, X. F., Zhang, G. L., Huang, Y. F., & Gurunathan, S. (2017). Zinc oxide nanoparticles induce apoptosis and autophagy in human ovarian cancer cells. International Journal of Nanomedicine, 12, 6521–6535. https://doi.org/10.2147/IJN.S140071.
Wang, L., Chen, C., Guo, L., Li, Q., Ding, H., Bi, H., & Guo, D. (2018). Zinc oxide nanoparticles induce murine photoreceptor cell death via mitochondria-related signaling pathway. Artificial Cells, Nanomedicine and Biotechnology, 46(sup1), 1102–1113. https://doi.org/10.1080/21691401.2018.1446018.
Biswas, S. K. (2016). Does the interdependence between oxidative stress and inflammation explain the antioxidant paradox? Oxidative Medicine and Cellular Longevity. https://doi.org/10.1155/2016/5698931.
Liu, J., Kang, Y., Yin, S., Song, B., Wei, L., Chen, L., & Shao, L. (2017). Zinc oxide nanoparticles induce toxic responses in human neuroblastoma SHSY5Y cells in a size-dependent manner. International Journal of Nanomedicine, 12, 8085–8099. https://doi.org/10.2147/IJN.S149070.
Najafi, M., Motevaseli, E., Shirazi, A., Geraily, G., Rezaeyan, A., Norouzi, F., et al. (2018). Mechanisms of inflammatory responses to radiation and normal tissues toxicity: clinical implications. International Journal of Radiation Biology. https://doi.org/10.1080/09553002.2018.1440092.
Khare, P., Sonane, M., Nagar, Y., Moin, N., Ali, S., Gupta, K. C., & Satish, A. (2015). Size dependent toxicity of zinc oxide nano-particles in soil nematode Caenorhabditis elegans. Nanotoxicology, 9(4), 423–432. https://doi.org/10.3109/17435390.2014.940403.
Kaya, H., Aydin, F., Gürkan, M., Yilmaz, S., Ates, M., Demir, V., & Arslan, Z. (2016). A comparative toxicity study between small and large size zinc oxide nanoparticles in tilapia (Oreochromis niloticus): Organ pathologies, osmoregulatory responses and immunological parameters. Chemosphere. https://doi.org/10.1016/j.chemosphere.2015.09.024.
Dobretsov, S., Sathe, P., Bora, T., Barry, M., Myint, M. T. Z., & Abri, M. A. (2020). Toxicity of Different zinc oxide nanomaterials at 3 trophic levels: implications for development of low-toxicity antifouling agents. Environmental Toxicology and Chemistry, 39(7), 1343–1354. https://doi.org/10.1002/etc.4720.
Zhou, Z., Son, J., Harper, B., Zhou, Z., & Harper, S. (2015). Influence of surface chemical properties on the toxicity of engineered zinc oxide nanoparticles to embryonic zebrafish. Beilstein Journal of Nanotechnology. https://doi.org/10.3762/bjnano.6.160.
Naveed Ul Haq, A., Nadhman, A., Ullah, I., Mustafa, G., Yasinzai, M., & Khan, I. (2017). Synthesis approaches of zinc oxide nanoparticles: the dilemma of ecotoxicity. Journal of Nanomaterials. https://doi.org/10.1155/2017/8510342.
Hua, J., Vijver, M. G., Richardson, M. K., Ahmad, F., & Peijnenburg, W. J. G. M. (2014). Particle-specific toxic effects of differently shaped zinc oxide nanoparticles to zebrafish embryos (Danio rerio). Environmental Toxicology and Chemistry, 33(12), 2859–2868. https://doi.org/10.1002/etc.2758.
Park, H. S., Shin, S. S., Meang, E. H., Hong, J. S., Park, J. I., Kim, S. H., et al. (2014). A 90-day study of subchronic oral toxicity of 20 nm, negatively charged zinc oxide nanoparticles in Sprague Dawley rats. International Journal of Nanomedicine. https://doi.org/10.2147/IJN.S57926.
Park, H. S., Kim, S. J., Lee, T. J., Kim, G. Y., Meang, E., Hong, J. S., et al. (2014). A 90-day study of sub-chronic oral toxicity of 20 nm positively charged zinc oxide nanoparticles in Sprague Dawley rats. International Journal of Nanomedicine. https://doi.org/10.2147/IJN.S57927.
Shubha, P., Gowda, M. L., Namratha, K., Manjunatha, H. B., & Byrappa, K. (2019). In vitro and In vivo evaluation of green-hydrothermal synthesized ZnO nanoparticles. Journal of Drug Delivery Science and Technology. https://doi.org/10.1016/j.jddst.2018.12.017.
Raisi Dehkourdi, B., Fatahian, S., & Shahanipoor, K. (2017). Synthesis, characterization and renal toxicity of ZnO and polyethylene glycol coated ZnO nanoparticles. Nanomedicine Journal.
Xia, T., Zhao, Y., Sager, T., George, S., Pokhrel, S., Li, N., et al. (2011). Decreased dissolution of ZnO by iron doping yields nanoparticles with reduced toxicity in the rodent lung and zebrafish embryos. ACS Nano. https://doi.org/10.1021/nn1028482.
Brown, D. M., Johnston, H. J., Gaiser, B., Pinna, N., Caputo, G., Culha, M., et al. (2018). A cross-species and model comparison of the acute toxicity of nanoparticles used in the pigment and ink industries. NanoImpact, 11, 20–32. https://doi.org/10.1016/j.impact.2018.02.001.
Vimercati, L., Cavone, D., Caputi, A., De Maria, L., Tria, M., Prato, E., & Ferri, G. M. (2020). Nanoparticles: An experimental study of zinc nanoparticles toxicity on marine crustaceans. General Overview on the Health Implications in Humans. Frontiers in Public Health, 8. https://doi.org/10.3389/fpubh.2020.00192.
Mumtaz, M. M., & Pohl, H. R. (2012). Interspecies uncertainty in molecular responses and toxicity of mixtures. EXS. https://doi.org/10.1007/978-3-7643-8340-4_12.
Wei, Y., Li, Y., Jia, J., Jiang, Y., Zhao, B., Zhang, Q., & Yan, B. (2016). Aggravated hepatotoxicity occurs in aged mice but not in young mice after oral exposure to zinc oxide nanoparticles. NanoImpact, 3–4, 1–11. https://doi.org/10.1016/j.impact.2016.09.003.
Wyss-Coray, T. (2016). Ageing, neurodegeneration and brain rejuvenation. Nature. https://doi.org/10.1038/nature20411.
Lotti, M. (2002). Age-related sensitivity of the nervous system to neurotoxic insults. In Toxicology Letters (Vol. 127, pp. 183–187). https://doi.org/10.1016/S0378-4274(01)00499-4.
Tian, L., Lin, B., Wu, L., Li, K., Liu, H., Yan, J., et al. (2015). Neurotoxicity induced by zinc oxide nanoparticles: Age-related differences and interaction. Scientific Reports, 5. https://doi.org/10.1038/srep16117.
Singh, K. P., Dhasmana, A., & Rahman, Q. (2018). Elucidation the toxicity mechanism of zinc oxide nanoparticle using molecular docking approach with proteins. Asian Journal of Pharmaceutical and Clinical Research, 11(3), 441–446. https://doi.org/10.22159/ajpcr.2018.v11i3.23384.
Santos, L. H. S., Ferreira, R. S., & Caffarena, E. R. (2019). Integrating molecular docking and molecular dynamics simulations. In Methods in Molecular Biology (Vol. 2053, pp. 13–34). https://doi.org/10.1007/978-1-4939-9752-7_2.
Hosseinzadeh, G., Maghari, A., Farnia, S. M. F., & Moosavi-Movahedi, A. A. (2018). Interaction mechanism of insulin with ZnO nanoparticles by replica exchange molecular dynamics simulation. Journal of Biomolecular Structure and Dynamics, 36(14), 3623–3635. https://doi.org/10.1080/07391102.2017.1396254.
Buglak, A. A., Zherdev, A. V., & Dzantiev, B. B. (2019). Nano-(Q)SAR for cytotoxicity prediction of engineered nanomaterials. Molecules. https://doi.org/10.3390/molecules24244537.
Mandlik, V., Bejugam, P. R., & Singh, S. (2016). Application of artificial neural networks in modern drug discovery. In Artificial Neural Network for Drug Design, Delivery and Disposition. https://doi.org/10.1016/B978-0-12-801559-9.00006-5.
Huang, Y., Li, X., Xu, S., Zheng, H., Zhang, L., Chen, J., et al. (2020). Quantitative structure–activity relationship models for predicting inflammatory potential of metal oxide nanoparticles. Environmental Health Perspectives, 128(6), 1–13. https://doi.org/10.1289/EHP6508.
Cao, J., Pan, Y., Jiang, Y., Qi, R., Yuan, B., Jia, Z., et al. (2020). Computer-aided nanotoxicology: risk assessment of metal oxide nanoparticlesvianano-QSAR. Green Chemistry, 22(11), 3512–3521. https://doi.org/10.1039/d0gc00933d.
Zhou, Z., Tang, X., Dai, W., Shi, J., & Chen, H. (2017). Nano-QSAR models for predicting cytotoxicity of metal oxide nanoparticles (MONPs) to E. coli. Canadian Journal of Chemistry, 95(8), 863–866. https://doi.org/10.1139/cjc-2017-0172.
Bailey, J. (2018). Does the stress of laboratory life and experimentation on animals adversely affect research data? A critical review. ATLA Alternatives to Laboratory Animals. https://doi.org/10.1177/026119291804600501.
Wu, F., Harper, B. J., & Harper, S. L. (2019). Comparative dissolution, uptake, and toxicity of zinc oxide particles in individual aquatic species and mixed populations. Environmental Toxicology and Chemistry. https://doi.org/10.1002/etc.4349.
Pence, L., & Tegulu, B. P. (2017). Chapter 3- Models of investigating placental biology. In Animal Models and Human Reproduction. Wiley Blackwell.
Funding
The study is supported by the Fundamental Research Grant Scheme (FRGS/1/2019/SKK10/UNIM/02/1) from the Ministry of Higher Education Malaysia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
We furthermore declare that there is no ethical issue in our experiments.
Consent to participate
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for publication
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Research Involving Humans and Animals Statement
None.
Informed Consent
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chong, C.L., Fang, C.M., Pung, S.Y. et al. Current Updates On the In vivo Assessment of Zinc Oxide Nanoparticles Toxicity Using Animal Models. BioNanoSci. 11, 590–620 (2021). https://doi.org/10.1007/s12668-021-00845-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12668-021-00845-2