Abstract
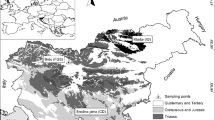
Carbon sources were estimated by measuring carbon isotope ratios (δ13C and Δ14C) with accelerator mass spectrometry (AMS) in forest soils of different lithology. Six locations were selected in temperate deciduous and coniferous stands in Slovenia (Žirovski vrh, Idrija, Kočevski Rog, Pohorje, Gorišnica, and Rakitna), where carbonate rocks consisting of limestone and dolomite are abundant as underlying bedrock. Carbon isotope fractionation would not have occurred in two carbonaceous soils, since the values of both δ13C and Δ14C changed consistently in these soils after thermal (550°C, 2 h) or chemical (1 M HCl, 24 h) treatments. Organic components were found to be predominant carbon sources (70–100%) in the uppermost portions (0–2 cm in depth). In deeper portions at a depth of about 30–35 cm, soil carbon may be derived completely from underlying carbonate minerals in Idria, western part of Slovenia. The Combination of heat and chemical treatments with AMS provides practical information on soil carbon sources in carbonaceous soils.





Similar content being viewed by others
References
Amundson R, Stern L, Baisden T, Wang Y (1998) The isotopic composition of soil and soil-resourced CO2. Geoderma 82:83–114
Breecker D, Sharp ZD (2008) A field and laboratory method for monitoring the concentration and isotopic composition of soil CO2. Rapid Commun Mass Spectrom 22:449–454
Cegnar T (2011) Climate of Slovenia at glance, Climate of Slovenia 1971–2000, The Environmental Agency of the Republic of Slovenia. http://www.arso.gov.si/
Fujiyoshi R, Sawamura S (2004) Mesoscale variability of vertical profiles of environmental radionuclides (40K, 226Ra, 210Pb and 137Cs) in temperate forest soils in Germany. Sci Total Environ 320:177–188
Fujiyoshi R, Haraki Y, Sumiyoshi T, Amano T, Kobal I, Vaupotič J (2010) Tracing the sources of gaseous components (222Rn, CO2 and its carbon isotopes) in soil air under a cool-temperate deciduous stand in Sapporo, Japan. Environ Geochem Health 32(1):73–82
Fujiyoshi R, Yamaguchi T, Okamoto K, Sumiyoshi T, Kobal I, Vaupotič J (2011) Tracing depositional consequences of environmental radionuclides (137Cs and 210Pb) in Slovenian forest soils. Cent Eur J Geosci 3(3):291–301
Genty D, Vokal B, Obelič B, Massault M (1998) Bomb 14C time history recorded in two modern stalagmites—importance of soil organic matter dynamics and bomb 14C distribution over continents. Earth Planet Sci Lett 160:795–809
Gunasekaran S, Anbalagan G (2007) Thermal decomposition of natural dolomite. Bull Mater Sci 30(4):339–344
Guo J, Atarashi-Ando M, Amano H (2003) Variation of 14C, 137Cs and stable carbon composition in forest soil and its implications. J Radioanal Nucl Chem 255(1):223–229
Jin L, Ogrinc N, Hamilton SK, Szramek K, Kanduč T, Walter LM (2009) Inorganic carbon isotope systematics in soil profiles undergoing silicate and carbonate weathering. Chem Geol 264:139–153
Kanduč T, Szramek K, Ogrinc N, Walter LM (2007) Origin and cycling of riverine inorganic carbon in the Sava River watershed (Slovenia) inferred from the major solutes and stable carbon isotopes. Biogeochemistry 86:137–154
Kayler ZE, Sulzman E, Rugh WD, Mix AC, Bond BJ (2010) Characterizing the impact of diffusive and advective soil gas transport on the measurement and interpretation of the isotopic signal of soil respiration. Soil Biol Biochem 42:435–444
Kuzyakov Y, Gavrichkova O (2010) Time lag between photosynthesis and carbon dioxide efflux from soil: a review of mechanisms and controls. Global Change Biol 16:3346–3386
Liu W, Moriizumi J, Yamazawa H, Iida T (2006) Depth profiles of radiocarbon and carbon isotopic compositions of organic matter and CO2 in a forest soil. J Environ Radioact 90:210–223
Lund K, Fogler HS, McCune CC, Acidization I (1973) The dissolution of dolomite in hydrochloric acid. Chem Eng Sci 28:691–700
Myorga E, Aufdenkampe AK, Masiello CA, Krusche AV, Hedges JI, Auay PD (2005) Young organic matter as a source of carbon dioxide outgassing from Amazon Rivers. Nature 436(28):538–541
Phillips CL, Nickerson N, Risk D, Kayler ZE, Anderson C, Mix A, Bond BJ (2010) Soil moisture effects on the carbon isotope composition of soil respiration. Rapid Commun Mass Spectrom 24:1271–1280
Poage MA, Feng X (2004) A theoretical analysis of steady state δ13C profiles of soil organic matter. Global Biogeochem Cycles 18:1–13
Schroeder PA, Austin JC, Dowd JF (2006) Estimating long-term soil respiration rates from carbon isotopes occluded in gibbsite. Geochim Cosmochim Acta 70:5692–5697
Schwab V, Spangenberg JE (2004) Organic geochemistry across the Permian–Triassic transition at the Idrijca Valley, Western Slovenia. Appl Geochem 19:55–72
Stuiver M, Quay PD (1981) Atmospheric C-14 changes resulting from fossil-fuel CO2 release and cosmic ray flux variability. Earth Planet Sci Lett 53:349–362
Tans P (1981) A compilation of bomb 14C data for use in global carbon model calculations. In: Bolin B (ed) Carbon cycle modelling (SCOPE 16). Wiley, New York, pp 131–157
Vrščaj B, Prus T, Lobnik F (2005) Soil information and soil data use in Slovenia. Eur Soil Bureau Res Rep 9:331–344
You SJ, Yin Y, Allen HE (1999) Partitioning of organic matter in soils: effect of pH and water/soil ratio. Sci Total Environ 227:155–160
Acknowledgments
This work was partially supported by the Japan Atomic Energy Agency (JAEA) for measuring carbon isotopes by AMS (2009A-F09, 2010A-F05), and by the Joint Research Program between Japan and Slovenia (JSPS) from 2010 to 2011. We would like to thank Dr. Primož Simončič of Slovenian Forestry Institute for helping with location settings in a protected forest site (Kočevski Rog) in Slovenia, and the staff members of the Mutsu AMS facility of JAEA (Aomori Prefecture, Japan) for providing isotope data of excellent quality.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fujiyoshi, R., Amano, H., Sakuta, Y. et al. Practical evaluation of carbon sources of forest soils in Slovenia from stable and radio-carbon isotope measurements. Environ Earth Sci 67, 133–140 (2012). https://doi.org/10.1007/s12665-011-1486-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12665-011-1486-x