Abstract
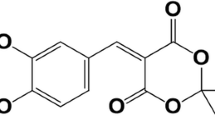
COA-Cl, a novel adenosine-like nucleic acid analog, has recently been shown to exert neuroprotective effects and to increase dopamine levels both in vivo and in vitro. Therefore, we hypothesized that COA-Cl could protect dopaminergic neurons against toxic insults. Thus, the present study aimed to investigate the protective effects of COA-Cl against hydrogen peroxide (H2O2)- and 6-hydroxydopamine (6-OHDA)-induced toxicity in PC12 cells and to elucidate the possible mechanisms. PC12 cells were incubated with COA-Cl (100 μM) with or without H2O2 or 6-OHDA (200 μM) for 24 h. Treatment with COA-Cl attenuated the decrease in cell viability, SOD activity and the Bcl-2/Bax ratio caused by H2O2. In addition, COA-Cl attenuated the increase in LDH release, ROS production, caspase-3 activity, and apoptosis induced by H2O2. Further, COA-Cl enhanced the protection of PC12 cells against the toxicity caused by 6-OHDA, as evidenced by an increase in cell viability and the Bcl-2/Bax ratio, and a decrease in LDH release. Our results are the first to demonstrate that COA-Cl potentially protects PC12 cells against toxicity induced by H2O2 and 6-OHDA, implying that COA-Cl could be a promising neuroprotective agent for the treatment of Parkinson’s disease.






Similar content being viewed by others
Data Availability
Not applicable.
References
Albers DS, Beal MF (2000) Mitochondrial dysfunction and oxidative stress in aging and neurodegenerative disease. J Neural Transm Suppl 59:133–154
Bernstein AI, Garrison SP, Zambetti GP, O’Malley KL (2011) 6-OHDA generated ROS induces DNA damage and p53- and PUMA-dependent cell death. Mol Neurodegener 6:2
Blum D, Torch S, Lambeng N, Nissou M, Benabid AL, Sadoul R, Verna JM (2001) Molecular pathways involved in the neurotoxicity of 6-OHDA, dopamine and MPTP: contribution to the apoptotic theory in Parkinson’s disease. Prog Neurobiol 65:135–172
Burns RS, Chiueh CC, Markey SP, Ebert MH, Jacobowitz DM, Kopin IJ (1983) A primate model of parkinsonism: selective destruction of dopaminergic neurons in the pars compacta of the substantia nigra by N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. Proc Natl Acad Sci USA 80:4546–4550
Cheng WY, Tong H, Miller EW, Chang CJ, Remington J, Zucker RM, Bromberg PA, Samet JM, Hofer TP (2010) An integrated imaging approach to the study of oxidative stress generation by mitochondrial dysfunction in living cells. Environ Health Perspect 118:902–908
Cho ES, Lee KW, Lee HJ (2008) Cocoa procyanidins protect PC12 cells from hydrogen-peroxide-induced apoptosis by inhibiting activation of p38 MAPK and JNK. Mutat Res 640(1–2):123–130
Choudhary GS, Al-Harbi S, Almasan A (2015) Caspase-3 activation is a critical determinant of genotoxic stress-induced apoptosis. Methods Mol Biol 1219:1–9
Cui K, Luo X, Xu K, Ven Murthy MR (2004) Role of oxidative stress in neurodegeneration: recent developments in assay methods for oxidative stress and nutraceutical antioxidants. Prog Neuropsychopharmacol Biol Psychiatry 28:771–799
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39:889–909
Dias V, Junn E, Mouradian MM (2013) The role of oxidative stress in Parkinson’s disease. J Parkinsons Dis 3:461–491
Ding X, Wang D, Li L, Ma H (2016) Dehydroepiandrosterone ameliorates H2O2-induced Leydig cells oxidation damage and apoptosis through inhibition of ROS production and activation of PI3K/Akt pathways. Int J Biochem Cell Biol 70:126–139
D’orsi B, Mateyka J, Prehn JHM (2017) Control of mitochondrial physiology and cell death by the Bcl-2 family proteins Bax and Bok. Neurochem Int 109:162–170
Duan X-HN, Wang W-L, Dai R, Yan H-W, Han C-N, Liu L-S (2015) Current situation of PC12 cell use in neuronal injury study. Int J Biotechnol Wellness Ind 4:61–66
Dumont A, Hehner SP, Hofmann TG, Ueffing M, Dröge W, Schmitz ML (1999) Hydrogen peroxide-induced apoptosis is CD95-independent, requires the release of mitochondria-derived reactive oxygen species and the activation of NF-kappaB. Oncogene 18:747–757
Franco-Iborra S, Vila M, Perier C (2018) Mitochondrial quality control in neurodegenerative diseases: focus on Parkinson’s disease and Huntington’s disease. Front Neurosci 12:342
Greene LA, Tischler AS (1976) Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Proc Natl Acad Sci USA 73:2424–2428
Hadipour E, Tayarani-Najaran Z, Fereidoni M (2020) Vitamin K2 protects PC12 cells against Aβ (1–42) and H2O2-induced apoptosis via p38 MAP kinase pathway. Nutr Neurosci 23:343–352
Hanrott K, Gudmunsen L, O’Neill MJ, Wonnacott S (2006) 6-hydroxydopamine-induced apoptosis is mediated via extracellular auto-oxidation and caspase 3-dependent activation of protein kinase Cdelta. J Biol Chem 281:5373–5382
Heusinkveld HJ, Westerink RHS (2017) Comparison of different in vitro cell models for the assessment of pesticide-induced dopaminergic neurotoxicity. Toxicol In Vitro 45:81–88
Huang WC, Chen PC, Jou SB, Cheng JT (2001) Protein kinase C and changes in manganese superoxide dismutase gene expression in cultured glial cells. Clin Exp Pharmacol Physiol 28:822–825
Jamal M, Tsukamoto I, Takata M, Ito A, Tanaka N, Miki T, Takakura A, Ameno K, Kubota Y, Konishi R, Kinoshita H (2019) COA-Cl induces dopamine release and tyrosine hydroxylase phosphorylation: In vivo reverse microdialysis and in vitro analysis. Brain Res 1706:68–74
Jiang B, Liu JH, Bao YM, An LJ (2003) Hydrogen peroxide-induced apoptosis in pc12 cells and the protective effect of puerarin. Cell Biol Int 27:1025–1031
Juknat AA, Mendez Mdel V, Quaglino A, Fameli CI, Mena M, Kotler ML (2005) Melatonin prevents hydrogen peroxide-induced Bax expression in cultured rat astrocytes. J Pineal Res 38:84–92
Kiningham KK, Oberley TD, Lin SM, Mattingly CA, St Clair DK (1999) Overexpression of manganese superoxide dismutase protects against mitochondrial-initiated poly (ADP-ribose) polymerase-mediated cell death. FASEB J 13:1601–1610
Kishimoto Y, Tsukamoto I, Nishigawa A, Nishimoto A, Kirino Y, Kato Y, Konishi R, Maruyama T, Sakakibara N (2018) Data on COA-Cl administration to the APP/PS2 double-transgenic mouse model of Alzheimers disease: improved hippocampus-dependent learning and unchanged spontaneous physical activity. Data Brief 20:1877–1883
Latchoumycandane C, Anantharam V, Jin H, Kanthasamy A, Kanthasamy A (2011) Dopaminergic neurotoxicant 6-OHDAinduces oxidative damage through proteolytic activation of PKCδ cell culture and animal models of Parkinson’s disease. Toxicol Appl Pharmacol 256:314–323
Lee YM, He W, Liou YC (2021) The redox language in neurodegenerative diseases: oxidative post-translational modifications by hydrogen peroxide. Cell Death Dis 12:58
Lindenboim L, Yuan J, Stein R (2000) Bcl-xS and Bax induce different apoptotic pathways in PC12 cells. Oncogene 19:1783–1793
Lopez MV, Cuadrado MP, Ruiz-Poveda OM, Del Fresno AM, Accame ME (2007) Neuroprotective effect of individual ginsenosides on astrocytes primary culture. Biochim Biophys Acta 1770:1308–1316
Lu F, Nakamura T, Okabe N, Himi N, Nakamura-Maruyama E, Shiromoto T, Narita K, Tsukamoto I, Xi G, Keep RF, Miyamoto O (2016) COA-Cl, a novel synthesized nucleoside analog, exerts neuroprotective effects in the acute phase of intracerebral hemorrhage. J Stroke Cerebrovasc Dis 25:2637–2643
Matsura M, Kai Y, Fujii H, Ito K (1999) Yamada, Hydrogen peroxide-induced apoptosis in HL-60 cells requires caspase-3 activation. Free Radic Res 30:73–83
McKeague AL, Wilson DJ, Nelson J (2003) Staurosporine-induced apoptosis and hydrogen peroxide-induced necrosis in two human breast cell lines. Br J Cancer 88:125–131
Michel PP, Hirsch EC, Hunot S (2016) Understanding dopaminergic cell death pathways in Parkinson disease. Neuron 90:675–691
Minjie X, Wei W, Zhou Z, Yongfei Y (2005) Capillary electrophoresis analysis of hydrogen peroxide induced apoptosis in PC12 cells. J Pharm Biomed Anal 39:853–860
Nesi G, Sestito S, Digiacomo M, Rapposelli S (2017) Oxidative stress, mitochondrial abnormalities and proteins deposition: multitarget approaches in Alzheimer’s Disease. Curr Top Med Chem 17:3062–3079
Nishikido T, Oyama JI, Shiraki A, Tsukamoto I, Igarashi J, Node K (2019) COA-Cl (2-Cl-C.OXT-A) can promote coronary collateral development following acute myocardial infarction in mice. Sci Rep 9:2533
Okabe N, Nakamura E, Himi N, Narita K, Tsukamoto I, Maruyama T, Sakakibara N, Nakamura T, Itano T, Miyamoto O (2013) Delayed administration of the nucleic acid analog 2Cl-C.OXT-A attenuates brain damage and enhances functional recovery after ischemic stroke. Brain Res 1506:115–131
Oraki Kohshour M, Najafi L, Heidari M, Ghaffari Sharaf M (2013) Antiproliferative effect of H2O2 against human acute myelogenous leukemia KG1 cell line. J Acupunct Meridian Stud 6:134–141
Pantic I, Cumic J, Skodric SR, Dugalic S, Brodski C (2021) Oxidopamine and oxidative stress: Recent advances in experimental physiology and pharmacology. Chem Biol Interact 336:109380
Poljsak B, Šuput D, Milisav I (2013) Achieving the balance between ROS and antioxidants: when to use the synthetic antioxidants. Oxid Med Cell Longev 2013:956792
Porter AG, Janicke RU (1999) Emerging roles of caspase-3 in apoptosis. Cell Death Differ 6:99–104
Ramazani E, Tayarani-Najaran Z, Fereidoni M (2019) Celecoxib, indomethacin, and ibuprofen prevent 6-hydroxydopamine-induced PC12 cell death through the inhibition of NFκB and SAPK/JNK pathways. Iran J Basic Med Sci 22(5):477–484
Redza-Dutordoir M, Averill-Bates DA (2016) Activation of apoptosis signalling pathways by reactive oxygen species. Biochim Biophys Acta 1863:2977–2992
Rhee SG (1999) Redox signaling: hydrogen peroxide as intracellular messenger. Exp Mol Med 31:53–59
Sakakibara N, Igarashi J, Takata M, Demizu Y, Misawa T, Kurihara M, Konishi R, Kato Y, Maruyama T, Tsukamoto I (2015) Synthesis and evaluation of novel carbocyclic oxetanocin A (COA-Cl) derivatives as potential tube formation agents. Chem Pharm Bull (Tokyo) 63:701–709
Sakamoto I, Himi N, Hayashi N, Okabe N, Nakamura-Maruyama E, Tsukamoto I, Hasegawa T, Miyamoto O (2021) The protective effect and mechanism of COA-Cl in acute phase after spinal cord injury. Neurosci Res 170:114–121
Satoh T, Sakai N, Enokido Y, Uchiyama Y, Hatanaka H (1996) Free radical-independent protection by nerve growth factor and Bcl-2 of PC12 cells from hydrogen peroxide-triggered apoptosis. J Biochem 120:540–546
Shin JH, Kim SW, Lim CM, Jeong JY, Piao CS, Lee JK (2009) alphaB-crystallin suppresses oxidative stress-induced astrocyte apoptosis by inhibiting caspase-3 activation. Neurosci Res 64:355–361
Simola N, Morelli M, Carta AR (2007) The 6-hydroxydopamine model of Parkinson’s disease. Neurotox Res 11:151–167
Sokolova T, Gutterer JM, Hirrlinger J, Hamprecht B, Dringen R (2001) Catalase in astroglia-rich primary cultures from rat brain: immunocytochemical localization and inactivation during the disposal of hydrogen peroxide. Neurosci Lett 297:129–132
Sundqvist M, Christenson K, Bjornsdottir H, Osla V, Karlsson A, Dahlgren C, Speert DP, Fasth A, Brown KL, Bylund J (2017) Elevated mitochondrial reactive oxygen species and cellular redox imbalance in human NADPH-oxidase-deficient phagocytes. Front Immunol 8:1828
Takata MK, Yamaguchi F, Nakanose K, Watanabe Y, Hatano N, Tsukamoto I, Nagata M, Izumori K, Tokuda M (2005) Neuroprotective effect of D-psicose on 6-hydroxydopamine-induced apoptosis in rat pheochromocytoma (PC12) cells. J Biosci Bioeng 100:511–516
Trist BG, Hare DJ, Double KL (2019) Oxidative stress in the aging substantia nigra and the etiology of Parkinson’s disease. Aging Cell 18:e13031
Tsao CW, Cheng JT, Shen CL, Lin YS (1996) 6-Hydroxydopamine induces thymocyte apoptosis in mice. J Neuroimmunol 65:91–95
Tsujimoto Y (1998) Role of Bcl-2 family proteins in apoptosis: apoptosomes or mitochondria? Genes Cells 3:697–707
Tsukamoto I, Sakakibara N, Maruyama T, Igarashi J, Kosaka H, Kubota Y, Tokuda M, Ashino H, Hattori K, Tanaka S, Kawata M, Konishi R (2010) A novel nucleic acid analogue shows strong angiogenic activity. Biochem Biophys Res Commun 399:699–704
Wu Y, Wang D, Wang X, Wang Y, Ren F, Chang D, Chang Z, Jia B (2011) Caspase 3 is activated through caspase 8 instead of caspase 9 during H2O2-induced apoptosis in HeLa cells. Cell Physiol Biochem 27:539–546
Yang J, Yang J, Liang SH, Xu Y, Moore A, Ran C (2016) Imaging hydrogen peroxide in Alzheimer’s disease via cascade signal amplification. Sci Rep 6:35613
Zou XD, Guo SQ, Hu ZW, Li WL (2016) NAMPT protects against 6-hydroxydopamine-induced neurotoxicity in PC12 cells through modulating SIRT1 activity. Mol Med Rep 13(5):4058–4064
Author information
Authors and Affiliations
Contributions
All authors whose names appear on the submission (1) made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data, or the creation of new software used in the work; (2) drafted the work or revised it critically for important intellectual content; (3) approved the version to be published; and (4) agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12640_2022_587_MOESM1_ESM.ppt
Supplementary file1 (PPT 335 KB) Fig. 7 Full gels for all Western blot images used for quantification in Fig. 4
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jamal, M., Tsukamoto, I., Maki, T. et al. COA-Cl Evokes Protective Responses Against H2O2-and 6-OHDA-Induced Toxic Injury in PC12 Cells. Neurotox Res 40, 2061–2071 (2022). https://doi.org/10.1007/s12640-022-00587-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-022-00587-3