Abstract
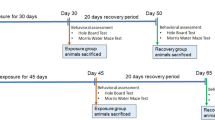
Acrolein is a clear, colorless liquid and a highly reactive α, β-unsaturated aldehyde. Acrolein, a byproduct and initiator of oxidative stress, has a major role in the pathogenesis of disorders including pulmonary, cardiovascular, atherosclerosis, and neurodegenerative diseases. Environmental or dietary exposure and endogenous production are common sources of acrolein. Widespread exposure to acrolein is a major risk for human health; therefore, we decided to investigate the neurological effects of acrolein. In this study, we used male Sprague-Dawley rats and exposed them orally to acrolein (0.5, 1, 3, and 5 mg/kg/day) for 90 days and investigated the neurobehavioral and electrophysiological disturbances. We also assessed the correlation between neurotoxicity and CSF concentration of acrolein in the rats. The results showed that chronic oral administration of acrolein at 5 mg/kg/day impaired learning and memory in the neurobehavioral tests. In addition, acrolein decreased the release of excitatory neurotransmitters such as glutamate in electrophysiological studies. Our data demonstrated that chronic oral exposure of acrolein at a dose of 5 mg/kg leads to a direct correlation between neurotoxicity and its CSF concentration. In conclusion, exposure to acrolein as a major pollutant in the environment may cause cognitive problems and may have serious neurocognitive effects on humans.













Similar content being viewed by others
Data Availability
All data produced or analyzed in this study are included in this manuscript.
References
Abraham K, Andres S, Palavinskas R, Berg K, Appel KE, Lampen A (2011) Toxicology and risk assessment of acrolein in food. Mol Nutr Food Res 55:1277–1290. https://doi.org/10.1002/mnfr.201100481
Ahmadian-Moghadam H, Sadat-Shirazi MS, Seifi F et al (2019) Transgenerational influence of parental morphine exposure on pain perception, anxiety-like behavior and passive avoidance memory among male and female offspring of Wistar rats. EXCLI J 18:1019–1036. https://doi.org/10.17179/excli2019-1845
Alamed J, Wilcock DM, Diamond DM, Gordon MN, Morgan D (2006) Two-day radial-arm water maze learning and memory task; robust resolution of amyloid-related memory deficits in transgenic mice. Nat Protoc 1:1671–1679. https://doi.org/10.1038/nprot.2006.275
Ambaw A, Zheng L, Tambe MA, Strathearn KE, Acosta G, Hubers SA, Liu F, Herr SA, Tang J, Truong A, Walls E, Pond A, Rochet JC, Shi R (2018) Acrolein-mediated neuronal cell death and alpha-synuclein aggregation: implications for Parkinson’s disease. Mol Cell Neurosci 88:70–82. https://doi.org/10.1016/j.mcn.2018.01.006
Azechi H, Hakamada K, Yamamoto T (2019) A new inbred strain of Fawn-Hooded rats demonstrates mania-like behavioural and monoaminergic abnormalities. IBRO Reports 7:98–106. https://doi.org/10.1016/j.ibror.2019.11.001
Bao M, Joza PJ, Masters A, Rickert WS (2014) Analysis of selected carbonyl compounds in tobacco samples by using pentafluorobenzylhydroxylamine derivatization and gas chromatography-mass spectrometry. Beiträge Zur Tab Int to Tob Res 26:86–97. https://doi.org/10.2478/cttr-2014-0017
Butterfield DA, Castegna A, Lauderback CM, Drake J (2002) Evidence that amyloid beta-peptide-induced lipid peroxidation and its sequelae in Alzheimer’s disease brain contribute to neuronal death. Neurobiol Aging 23:655–664. https://doi.org/10.1016/S0197-4580(01)00340-2
Chen C-H, Joshi AU, Mochly-Rosen D (2016) The role of mitochondrial aldehyde dehydrogenase 2 (ALDH2) in neuropathology and neurodegeneration. Acta Neurol Taiwanica 25:111–123
Chen HJ, Wang CC, Chan DC, Chiu CY, Yang RS, Liu SH (2019) Adverse effects of acrolein, a ubiquitous environmental toxicant, on muscle regeneration and mass. J Cachexia Sarcopenia Muscle 10:165–176. https://doi.org/10.1002/jcsm.12362
Esterbauer H, Schaur RJ, Zollner H (1991) Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radic Biol Med 11:81–128. https://doi.org/10.1016/0891-5849(91)90192-6
Falcone E, Ahmed IMM, Oliveri V, Bellia F, Vileno B, el Khoury Y, Hellwig P, Faller P, Vecchio G (2020) Acrolein and copper as competitive effectors of α-Synuclein. Chem Eur J 26:1871–1879. https://doi.org/10.1002/chem.201904885
Farzanehfar V, Faizi M, Naderi N, Kobarfard F (2017) Development of an analytical method for dibutyl phthalate determination using Surrogate analyte approach. Iran. J. Pharm. Res 16:140–145. https://doi.org/10.22037/ijpr.2017.1968
Farzanehfar V, Naderi N, Kobarfard F, Faizi M (2016) Determination of dibutyl phthalate neurobehavioral toxicity in mice. Food Chem Toxicol 94:221–226. https://doi.org/10.22037/ijpr.2017.1968
Fulton JL, Horwitz MA, Bruske EL, Johnson JS (2018) Asymmetric organocatalytic sulfa-Michael addition to enone diesters. J Org Chem 83:3385–3391. https://doi.org/10.1021/acs.joc.8b00007
Gholami Pourbadie H, Naderi N, Janahmadi M, Mehranfard N, Motamedi F (2016) Calcium channel blockade attenuates abnormal synaptic transmission in the dentate gyrus elicited by entorhinal amyloidopathy. Synapse 70:408–417. https://doi.org/10.1002/syn.21915
Guan X, Wang Y, Kai G, Zhao S, Huang T, Li Y, Xu Y, Zhang L, Pang T (2019) Cerebrolysin ameliorates focal cerebral ischemia injury through neuroinflammatory inhibition via CREB/PGC-1α pathway. Front Pharmacol 10:1245. https://doi.org/10.3389/fphar.2019.01245
Haghani M, Shabani M, Javan M, Motamedi F, Janahmadi M (2012) CB1 cannabinoid receptor activation rescues amyloid β-induced alterations in behaviour and intrinsic electrophysiological properties of rat hippocampal CA1 pyramidal neurones. Cell Physiol Biochem 29:391–406. https://doi.org/10.1159/000338494
Haldenwanger A, Eling P, Kastrup A, Hildebrandt H (2010) Correlation between cognitive impairment and CSF biomarkers in amnesic MCI, non-amnesic MCI, and Alzheimer’s disease. J Alzheimers Dis 22:971–980. https://doi.org/10.3233/JAD-2010-101203
Henning RJ, Johnson GT, Coyle JP, Harbison RD (2017) Acrolein can cause cardiovascular disease: a review. Cardiovasc Toxicol 17:227–236. https://doi.org/10.1007/s12012-016-9396-5
Hernández-Álvarez D, Mena-Montes B, Toledo-Pérez R, Pedraza-Vázquez G, López-Cervantes SP, Morales-Salazar A, Hernández-Cruz E, Lazzarini-Lechuga R, Vázquez-Cárdenas RR, Vilchis-DeLaRosa S, Posadas-Rodríguez P, Santín-Márquez R, Rosas-Carrasco O, Ibañez-Contreras A, Alarcón-Aguilar A, López-Díazguerrero NE, Luna-López A, Königsberg M (2019) Long-term moderate exercise combined with metformin treatment induces an hormetic response that prevents strength and muscle mass loss in old female wistar rats. Oxidative Med Cell Longev 2019:1–14. https://doi.org/10.1155/2019/3428543
Hosseinzadeh M, Pourbadie HG, Khodagholi F, Daftari M, Naderi N, Motamedi F (2019) Preconditioning with toll-like receptor agonists attenuates seizure activity and neuronal hyperexcitability in the pilocarpine rat model of epilepsy. Neuroscience 408:388–399. https://doi.org/10.1016/j.neuroscience.2019.04.020
Huang Y-J, Jin M-H, Pi R-B, Zhang JJ, Ouyang Y, Chao XJ, Chen MH, Liu PQ, Yu JC, Ramassamy C, Dou J, Chen XH, Jiang YM, Qin J (2013a) Acrolein induces Alzheimer’s disease-like pathologies in vitro and in vivo. Toxicol Lett 217:184–191. https://doi.org/10.1016/j.toxlet.2012.12.023
Huang Y-J, Zhang L, Shi L-Y, Wang YY, Yang YB, Ke B, Zhang TY, Qin J (2018) Caloric restriction ameliorates acrolein-induced neurotoxicity in rats. Neurotoxicology 65:44–51. https://doi.org/10.1016/j.neuro.2018.01.003
Huang Y, Jin M, Pi R, Zhang J, Chen M, Ouyang Y, Liu A, Chao X, Liu P, Liu J, Ramassamy C, Qin J (2013b) Protective effects of caffeic acid and caffeic acid phenethyl ester against acrolein-induced neurotoxicity in HT22 mouse hippocampal cells. Neurosci Lett 535:146–151. https://doi.org/10.1016/j.neulet.2012.12.051
Hughes EG, Peng X, Gleichman AJ, Lai M, Zhou L, Tsou R, Parsons TD, Lynch DR, Dalmau J, Balice-Gordon RJ (2010) Cellular and synaptic mechanisms of anti-NMDA receptor encephalitis. J Neurosci 30:5866–5875. https://doi.org/10.1523/JNEUROSCI.0167-10.2010
Hühmer AF, Biringer RG, Amato H, Fonteh AN, Harrington MG (2006) Protein analysis in human cerebrospinal fluid: physiological aspects, current progress and future challenges. Dis Markers 22:3–26. https://doi.org/10.1155/2006/158797
Igarashi K, Kashiwagi K (2011) Protein-conjugated acrolein as a biochemical marker of brain infarction. Mol Nutr Food Res 55:1332–1341. https://doi.org/10.1002/mnfr.201100068
Igarashi K, Uemura T, Kashiwagi K (2020) Assessing acrolein for determination of the severity of brain stroke, dementia, renal failure, and Sjögren’s syndrome. Amino Acids 52:119–127. https://doi.org/10.1007/s00726-019-02700-x
Igarashi K, Yoshida M, Waragai M, Kashiwagi K (2015) Evaluation of dementia by acrolein, amyloid-β and creatinine. Clin Chim Acta 450:56–63. https://doi.org/10.1016/j.cca.2015.07.017
Jung HY, Yoo DY, Park JH, Kim J, Chung J, Kim D, Won M‑H, Yoon Y, Hwang I (2018) Age-dependent changes in vesicular glutamate transporter 1 and 2 expression in the gerbil hippocampus. Mol Med Rep 17:6465–6471. https://doi.org/10.3892/mmr.2018.8705
Kaminskas LM, Pyke SM, Burcham PC (2004) Strong protein adduct trapping accompanies abolition of acrolein-mediated hepatotoxicity by hydralazine in mice. J Pharmacol Exp Ther 310:1003–1010. https://doi.org/10.1124/jpet.104.067330
Lahouel A, Kebieche M, Lakroun Z, Rouabhi R, Fetoui H, Chtourou Y, Djamila Z, Soulimani R (2016) Neurobehavioral deficits and brain oxidative stress induced by chronic low dose exposure of persistent organic pollutants mixture in adult female rat. Environ Sci Pollut Res 23:19030–19040. https://doi.org/10.1007/s11356-016-6913-9
Liu J-H, Wang T-W, Lin Y-Y, Ho WC, Tsai HC, Chen SP, Lin AMY, Liu TY, Wang HT (2020) Acrolein is involved in ischemic stroke-induced neurotoxicity through spermidine/spermine-N1-acetyltransferase activation. Exp Neurol 323:113066. https://doi.org/10.1016/j.expneurol.2019.113066
LoPachin RM, Barber DS, Gavin T (2008) Molecular mechanisms of the conjugated α, β-unsaturated carbonyl derivatives: relevance to neurotoxicity and neurodegenerative diseases. Toxicol Sci 104:235–249. https://doi.org/10.1093/toxsci/kfm301
Łuczaj W, Moniuszko A, Jarocka-Karpowicz I et al (2016) Tick-borne encephalitis–lipid peroxidation and its consequences. Scand J Clin Lab Invest 76:1–9. https://doi.org/10.3109/00365513.2015.1084040
Luo J, Robinson JP, Shi R (2005) Acrolein-induced cell death in PC12 cells: role of mitochondria-mediated oxidative stress. Neurochem Int 47:449–457. https://doi.org/10.1016/j.neuint.2005.07.002
Mehranfard N, Gholamipour-Badie H, Motamedi F et al (2014) The effect of paxilline on early alterations of electrophysiological properties of dentate gyrus granule cells in pilocarpine-treated rats. Iran J Pharm Es 13:125–132. https://doi.org/10.22037/ijpr.2014.1467
Mizoi M, Yoshida M, Saiki R, Waragai M, Uemura K, Akatsu H, Kashiwagi K, Igarashi K (2014) Distinction between mild cognitive impairment and Alzheimer’s disease by CSF amyloid β40 and β42, and protein-conjugated acrolein. Clin Chim Acta 430:150–155. https://doi.org/10.1016/j.cca.2014.01.007
Moghe A, Ghare S, Lamoreau B, Mohammad M, Barve S, McClain C, Joshi-Barve S (2015) Molecular mechanisms of acrolein toxicity: relevance to human disease. Toxicol Sci 143:242–255. https://doi.org/10.1093/toxsci/kfu233
Moriguchi S (2011) Pharmacological study on Alzheimer’s drugs targeting calcium/calmodulin-dependent protein kinase II. J Pharmacol Sci 117:6–11. https://doi.org/10.1254/jphs.11R06CP
Morris RG (2013) NMDA receptors and memory encoding. Neuropharmacology 74:32–40. https://doi.org/10.1016/j.neuropharm.2013.04.014
Neher E, Sakmann B (1992) The patch clamp technique. Sci Am 266:44–51. https://doi.org/10.1038/scientificamerican0392-44
Nieus T, Sola E, Mapelli J, Saftenku E, Rossi P, D'Angelo E (2006) LTP regulates burst initiation and frequency at mossy fiber–granule cell synapses of rat cerebellum: experimental observations and theoretical predictions. J Neurophysiol 95:686–699. https://doi.org/10.1152/jn.00696.2005
Pirmoghani A, Salehi I, Moradkhani S, Karimi SA, Salehi S (2019) Effect of Crataegus extract supplementation on diabetes induced memory deficits and serum biochemical parameters in male rats. IBRO Reports 7:90–96. https://doi.org/10.1016/j.ibror.2019.10.004
Radanovic M, Oshiro CA, Freitas TQ, Talib LL, Forlenza OV (2019) Correlation between CSF biomarkers of Alzheimer’s disease and global cognition in a psychogeriatric clinic cohort. Brazilian J Psychiatry 41:479–484. https://doi.org/10.1590/1516-4446-2018-0296
Rodigast M, Mutzel A, Iinuma Y, Haferkorn S, Herrmann H (2015) Characterisation and optimisation of a sample preparation method for the detection and quantification of atmospherically relevant carbonyl compounds in aqueous medium. Atmos Meas 8:2409–2416. https://doi.org/10.5194/amt-8-2409-2015
Sachser RM, Haubrich J, Lunardi PS, de Oliveira AL (2017) Forgetting of what was once learned: exploring the role of postsynaptic ionotropic glutamate receptors on memory formation, maintenance, and decay. Neuropharmacology 112:94–103. https://doi.org/10.1016/j.neuropharm.2016.07.015
Segev A, Garcia-Oscos F, Kourrich S (2016) Whole-cell patch-clamp recordings in brain slices J Vis Exp e54024. https://doi.org/10.3791/54024
Selmanoğlu G, Özgün GM, Karacaoğlu E (2018) Acrolein-mediated neurotoxicity in growing Wistar male rats. Pestic Biochem Physiol 149:37–43. https://doi.org/10.1016/j.pestbp.2018.05.006
Shamoto-Nagai M, Maruyama W, Hashizume Y, Yoshida M, Osawa T, Riederer P, Naoi M (2007) In parkinsonian substantia nigra, α-synuclein is modified by acrolein, a lipid-peroxidation product, and accumulates in the dopamine neurons with inhibition of proteasome activity. J Neural Transm 114:1559–1567. https://doi.org/10.1007/s00702-007-0789-2
Shan S, Tian L, Fang R (2019) Chlorogenic acid exerts beneficial effects in 6-hydroxydopamine-induced neurotoxicity by inhibition of endoplasmic reticulum stress. Med Sci Monit 25:453–459. https://doi.org/10.12659/MSM.911166
Sheibani V, Mandegary A, Vazifekhahan E et al (2019) Zataria multiflora Boiss. Extract improves spatial memory and learning capacity in scopolamine-induced amnesic rats. Avicenna J Phytomed 9:587–596. https://doi.org/10.22038/AJP.2019.13540
Shi L-Y, Zhang L, Li H, Liu TL, Lai JC, Wu ZB, Qin J (2018) Protective effects of curcumin on acrolein-induced neurotoxicity in HT22 mouse hippocampal cells. Pharmacol Reports 70:1040–1046. https://doi.org/10.1016/j.pharep.2018.05.006
Shukitt-Hale B, McEwen JJ, Szprengiel A, Joseph JA (2004) Effect of age on the radial arm water maze—a test of spatial learning and memory. Neurobiol Aging 25:223–229. https://doi.org/10.1016/S0197-4580(03)00041-1
Sousa BC, Pitt AR, Spickett CM (2017) Chemistry and analysis of HNE and other prominent carbonyl-containing lipid oxidation compounds. Free Radic Biol Med 111:294–308. https://doi.org/10.1016/j.freeradbiomed.2017.02.003
Stevens JF, Maier CS (2008) Acrolein: sources, metabolism, and biomolecular interactions relevant to human health and disease. Mol Nutr Food Res 52:7–25. https://doi.org/10.1002/mnfr.200700412
Storme T, Deroussent A, Mercier L, Prost E, Re M, Munier F, Martens T, Bourget P, Vassal G, Royer J, Paci A (2009) New ifosfamide analogs designed for lower associated neurotoxicity and nephrotoxicity with modified alkylating kinetics leading to enhanced in vitro anticancer activity. J Pharmacol Exp Ther 328:598–609. https://doi.org/10.1124/jpet.108.144170
Togashi M, Terai T, Kojima H, Hanaoka K, Igarashi K, Hirata Y, Urano Y, Nagano T (2014) Practical fluorescence detection of acrolein in human plasma via a two-step tethering approach. Chem Commun 50:14946–14948. https://doi.org/10.1039/C4CC02578D
Tordera RM, Totterdell S, Wojcik SM, Brose N, Elizalde N, Lasheras B, del Rio J (2007) Enhanced anxiety, depressive-like behaviour and impaired recognition memory in mice with reduced expression of the vesicular glutamate transporter 1 (VGLUT1). Eur J Neurosci 25:281–290. https://doi.org/10.1111/j.1460-9568.2006.05259.x
Tsou H-H, Hsu W-C, Fuh J-L, Chen SP, Liu TY, Wang HT (2018) Alterations in acrolein metabolism contribute to Alzheimer’s disease. J Alzheimers Dis 61:571–580. https://doi.org/10.3233/JAD-170736
Tully M, Zheng L, Shi R (2014) Acrolein detection: potential theranostic utility in multiple sclerosis and spinal cord injury. Expert Rev Neurother 14:679–685. https://doi.org/10.1586/14737175.2014.918849
Uchida K (1999) Current status of acrolein as a lipid peroxidation product. Trends Cardiovasc Med 9:109–113. https://doi.org/10.1016/S1050-1738(99)00016-X
Walls MK, Race N, Zheng L, Vega-Alvarez SM, Acosta G, Park J, Shi R (2016) Structural and biochemical abnormalities in the absence of acute deficits in mild primary blast-induced head trauma. J Neurosurg 124:675–686. https://doi.org/10.3171/2015.1.JNS141571
Wang H-T, Lin J-H, Yang C-H et al (2017) Acrolein induces mtDNA damages, mitochondrial fission and mitophagy in human lung cells. Oncotarget 8:70406–70421. https://doi.org/10.18632/oncotarget.19710
Waragai M, Yoshida M, Mizoi M, Saiki R, Kashiwagi K, Takagi K, Arai H, Tashiro J, Hashimoto M, Iwai N, Uemura K, Igarashi K (2012) Increased protein-conjugated acrolein and amyloid-β 40/42 ratio in plasma of patients with mild cognitive impairment and Alzheimer’s disease. J Alzheimers Dis 32:33–41. https://doi.org/10.3233/JAD-2012-120253
Yasuo M, Droma Y, Kitaguchi Y, Ito M, Imamura H, Kawakubo M, Hanaoka M (2019) The relationship between acrolein and oxidative stress in COPD: in systemic plasma and in local lung tissue. Int J Chron Obstruct Pulmon Dis 14:1527–1537. https://doi.org/10.2147/COPD.S208633
Zhang L, Geohagen BC, Gavin T, LoPachin RM (2016) Joint toxic effects of the type-2 alkene electrophiles. Chem Biol Interact 254:198–206. https://doi.org/10.1016/j.cbi.2016.06.014
Zhu Y, Liu B, Zheng X, Wu J, Chen S, Chen Z, Chen T, Huang Z, Lei W (2019) Partial decortication ameliorates dopamine depletion-induced striatal neuron lesions in rats. Int J Mol Med 44:1414–1424. https://doi.org/10.3892/ijmm.2019.4288
Zimcikova E, Simko J, Karesova I, Kremlacek J, Malakova J (2017) Behavioral effects of antiepileptic drugs in rats: are the effects on mood and behavior detectable in open-field test? Seizure 52:35–40. https://doi.org/10.1016/j.seizure.2017.09.015
Code Availability
Not applicable.
Funding
This study was supported by a grant (Grant No. 313/1997) from the Research Council of Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Author information
Authors and Affiliations
Contributions
Data analysis, writing - original draft preparation: Mona Khoramjouy; investigation: Mona Khoramjouy and Elmira Heidarli; conceptualization, writing - review and editing: Nima Naderi, Farzad Kobarfard, and Mehrdad Faizi; resources: Farzad Kobarfard; project administration, funding acquisition, and supervision: Mehrdad Faizi.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
All the experiments were done according to instructions of the ethical standards of the Institutional Animal Care and Use Committee (IACUC) of Shahid Beheshti University of Medical Sciences with the approval code IR.SBMU.PHNM.1396.920.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khoramjouy, M., Naderi, N., Kobarfard, F. et al. An Intensified Acrolein Exposure Can Affect Memory and Cognition in Rat. Neurotox Res 39, 277–291 (2021). https://doi.org/10.1007/s12640-020-00278-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-020-00278-x