Abstract
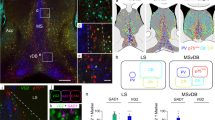
The septum is a basal forebrain region located between the lateral ventricles in rodents. It consists of lateral and medial divisions. Medial septal projections regulate hippocampal theta rhythm whereas lateral septal projections are involved in processes such as affective functions, memory formation, and behavioral responses. Gamma-aminobutyric acidergic neurons of the septal region possess the 65 and 67 isoforms of the enzyme glutamic acid decarboxylase. Although data on the glutamic acid decarboxylase isoform distribution in the septal region generally appears to indicate glutamic acid decarboxylase 67 dominance, different studies have given inconsistent results in this regard. The aim of this study was therefore to obtain information on the distributions of both of these glutamic acid decarboxylase isoforms in the septal region in transgenic mice. Two animal groups of glutamic acid decarboxylase–green fluorescent protein knock-in transgenic mice were utilized in the experiment. Brain sections from the region were taken for anti-green fluorescent protein immunohistochemistry in order to obtain estimated quantitative data on the number of gamma-aminobutyric acidergic neurons. Following the immunohistochemical procedures, the mean numbers of labeled cells in the lateral and medial septal nuclei were obtained for the two isoform groups. Statistical analysis yielded significant results which indicated that the 65 isoform of glutamic acid decarboxylase predominates in both lateral and medial septal nuclei (unpaired two-tailed t-test p < 0.0001 for LS, p < 0.01 for MS). This study is the first to reveal the dominance of glutamic acid decarboxylase isoform 65 in the septal region in glutamic acid decarboxylase–green fluorescent protein transgenic mice.





Similar content being viewed by others
References
Alreja M, Shanabrough M, Liu W, Leranth C (2000a) Opioids suppress IPSCs in neurons of the rat medial septum/diagonal band of Broca: involvement of mu-opioid receptors and septohippocampal GABAergic neurons. J Neurosci 20:1179–1189
Alreja M, Wu M, Liu W, Atkins JB, Leranth C, Shanabrough M (2000b) Muscarinic tone sustains impulse flow in the septohippocampal GABA but not cholinergic pathway: implications for learning and memory. J Neurosci 20:8103–8110
Asada H (1996) Mice lacking the 65 kDa isoform of glutamic acid decarboxylase (GAD65) maintain normal levels of GAD67 and GABA in their brains but are susceptible to seizures. Biochem Biophys Res Commun 229:891–895
Buzsáki G (2002) Theta oscillations in the hippocampus. Neuron 33:325–340
Castaneda MT, Garrido-Sanabria ER, Hernandez S et al (2005) Glutamic acid decarboxylase isoforms are differentially distributed in the septal region of the rat. Neurosci Res 52:107–119
Chalfie M (1995) Green fluorescent protein. Photochem Photobiol 62:651–656
Colom LV (2006) Septal networks: relevance to theta rhythm, epilepsy and Alzheimer’s disease. J Neurochem 96:609–623
Colom LV, Castaneda MT, Reyna T, Hernandez S, Garrido-Sanabria E (2005) Characterization of medial septal glutamatergic neurons and their projection to the hippocampus. Synapse 58:151–164
Deller T, Leranth C, Frotscher M (1994) Reciprocal connections of lateral septal neurons and neurons in the lateral hypothalamus in the rat: a combined phaseolus vulgaris-leucoagglutinin and fluoro-gold immunocytochemical study. Neurosci Lett 168:119–122
Drugan RC, Morrow AL, Weizman R et al (1988) Stress-induced behavioral depression in the rat is associated with a decrease in GABA receptor-mediated chloride ion flux and brain benzodiazepine receptor occupancy. Brain Res 487:45–51
Erlander MG, Tillakaratne NJ, Feldblum S, Patel N, Tobin AJ (1991) Two genes encode distinct glutamate decarboxylases. Neuron 7:91–100
Esclapez M, Tillakaratne NJ, Tobin AJ, Houser CR (1993) Comparative localization of mRNAs encoding two forms of glutamic acid decarboxylase with nonradioactive in situ hybridization methods. J Comp Neurol 331:339–362
Esclapez M, Tillakaratne NJ, Kaufman DL, Tobin AJ, Houser CR (1994) Comparative localization of two forms of glutamic acid decarboxylase and their mRNAs in rat brain supports the concept of functional differences between the forms. J Neurosci 14:1834–1855
Feldblum S, Erlander MG, Tobin AJ (1993) Different distributions of GAD65 and GAD67 mRNAs suggest that the two glutamate decarboxylases play distinctive functional roles. J Neurosci Res 34:689–706
Franklin KBJ, Paxinos G (1997) The mouse brain in stereotaxic coordinates. Academic, London
Garrido-Sanabria ER, Castaneda MT, Banuelos C, Perez-Cordova MG, Hernandez S, Colom LV (2006) Septal GABAergic neurons are selectively vulnerable to pilocarpine-induced status epilepticus and chronic spontaneous seizures. Neuroscience 142:871–883
Huang H, Michetti C, Busnelli M et al (2013) Chronic and acute intranasal oxytocin produce divergent social effects in mice. Neuropsychopharmacology 39:1102–1114
Kash SF, Johnson RS, Tecott LH et al (1997) Epilepsy in mice deficient in the 65-kDa isoform of glutamic acid decarboxylase. Proc Natl Acad Sci USA 94:14060–14065
Kaufman DL, McGinnis JF, Krieger NR, Tobin AJ (1986) Brain glutamate decarboxylase cloned in lambda gt-11: fusion protein produces gamma-aminobutyric acid. Science 232:1138–1140
Köhler C, Chan-Palay V (1983) Distribution of gamma aminobutyric acid containing neurons and terminals in the septal area: an immunohistochemical study using antibodies to glutamic acid decarboxylase in the rat brain. Anat Embryol 167:53–65
Köhler C, Chan-Palay V, Wu JY (1984) Septal neurons containing glutamic acid decarboxylase immunoreactivity project to the hippocampal region in the rat brain. Anat Embryol (Berlin) 169:41–44
Leranth C, Frotscher M (1989) Organization of the septal region in the rat brain: cholinergic–GABAergic interconnections and the termination of hippocampus–septal fibers. J Comp Neurol 289:304–314
Lopez-Bendito G (2004) Preferential origin and layer destination of GAD65–GFP cortical interneurons. Cereb Cortex 14:1122–1133
Marczynski TJ (1998) GABAergic deafferentation hypothesis of brain aging and Alzheimer’s disease revisited. Brain Res Bull 45:341–379
McLaughlin BJ, Barber R, Saito K, Roberts E, Wu JY (1975a) Immunocytochemical localization of glutamate decarboxylase in rat spinal cord. J Comp Neurol 164:305–321
McLaughlin BJ, Wood JG, Saito K, Roberts E, Wu JY (1975b) The fine structural localization of glutamate decarboxylase in developing axonal processes and presynaptic terminals of rodent cerebellum. Brain Res 85:355–371
Numan R (2000) The behavioral neuroscience of the septal region. Springer, New York
Oliva AA Jr, Jiang M, Lam T, Smith KL, Swann JW (2000) Novel hippocampal interneuronal subtypes identified using transgenic mice that express green fluorescent protein in GABAergic interneurons. J Neurosci 20:3354–3368
Panula P, Revuelta AV, Cheney DL, Wu JY, Costa E (1984) An immunohistochemical study on the location of GABAergic neurons in rat septum. J Comp Neurol 222:69–80
Paxinos G, Franklin KB (2013) The mouse brain in stereotaxic coordinates. Elsevier, San Diego
Rimvall K, Sheikh SN, Martin DL (1993) Effects of increased gamma-aminobutyric acid levels on GAD, protein and mRNA levels in rat cerebral cortex. J Neurochem 60:714–720
Risold PY, Swanson LW (1996) Structural evidence for functional domains in the rat hippocampus. Science 272:1484–1486
Risold PY, Swanson LW (1997) Connections of the rat lateral septal complex. Brain Res Rev 24:115–195
Sheehan TP, Chambers RA, Russell DS (2004) Regulation of affect by the lateral septum: implications for neuropsychiatry. Brain Res Rev 46:71–117
Soghomonian JJ, Laprade N (1997) Glutamate decarboxylase (GAD67 and GAD65) gene expression is increased in a subpopulation of neurons in the putamen of parkinsonian monkeys. Synapse 27:122–132
Storm-Mathisen J, Leknes AK, Bore AT et al (1983) First visualization of glutamate and GABA in neurones by immunocytochemistry. Nature 301:517–520
Swanson LW, Cowan WM (1979) The connections of the septal region in the rat. J Comp Neurol 186:621–655
Tamamaki N, Yanagawa Y, Tomioka R, Miyazaki J, Obata K, Kaneko T (2003) Green fluorescent protein expression and colocalization with calretinin, parvalbumin, and somatostatin in the GAD67–GFP knock-in mouse. J Comp Neurol 467:60–79
Wang X (2014) Immunofluorescently labeling glutamic acid decarboxylase 65 coupled with confocal imaging for identifying GABAergic somata in the rat dentate gyrus: a comparison with labeling glutamic acid decarboxylase 67. J Chem Neuroanat 61–62:51–63
Yuan PQ, Granas C, Kallstrom L et al (1997) Differential distribution of glutamic acid decarboxylase-65 and glutamic acid decarboxylase-67 messenger RNAs in the entopeduncular nucleus of the rat. Neuroscience 78:87–97
Zhao C, Driessen T, Gammie SC (2012) Glutamic acid decarboxylase 65 and 67 expression in the lateral septum is up-regulated in association with the postpartum period in mice. Brain Res 1470:35–44
Zhao C, Eisinger B, Gammie SC (2013) Characterization of GABAergic neurons in the mouse lateral septum: a double fluorescence in situ hybridization and immunohistochemical study using tyramide signal amplification. PLoS One 8:e73750. http://www.plosone.org. Accessed 30 Jan 2014
Acknowledgments
We would like to thank Dr. Paul Bolam and Dr. Juan Mena-Segovia (University of Oxford, Anatomical Neuropharmacology Unit) for providing transgenic mice brains for the experiments conducted in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Grant sponsor: Marmara University Scientific Research Projects Unit (SAG-C-TUP-250608-0162) to Dr. Umit S. Sehirli.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Verimli, U., Sehirli, U.S. Glutamic acid decarboxylase isoform distribution in transgenic mouse septum: an anti-GFP immunofluorescence study. Anat Sci Int 91, 398–406 (2016). https://doi.org/10.1007/s12565-015-0316-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12565-015-0316-8