Abstract
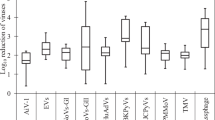
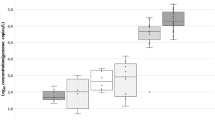
Irrigation water is a doorway for the pathogen contamination of fresh produce. We quantified pathogenic viruses [human adenoviruses, noroviruses of genogroups I and II, group A rotaviruses, Aichi virus 1 (AiV-1), enteroviruses (EnVs), and salivirus (SaliV)] and examined potential index viruses [JC and BK polyomaviruses (JCPyVs and BKPyVs), pepper mild mottle virus (PMMoV), and tobacco mosaic virus (TMV)] in irrigation water sources in the Kathmandu Valley, Nepal. River, sewage, wastewater treatment plant (WWTP) effluent, pond, canal, and groundwater samples were collected in September 2014, and in April and August 2015. Viruses were concentrated using an electronegative membrane-vortex method and quantified using TaqMan (MGB)-based quantitative PCR (qPCR) assays with murine norovirus as a molecular process control to determine extraction-reverse transcription-qPCR efficiency. Tested pathogenic viruses were prevalent with maximum concentrations of 5.5–8.8 log10 copies/L, and there was a greater abundance of EnVs, SaliV, and AiV-1. Virus concentrations in river water were equivalent to those in sewage. Canal, pond, and groundwater samples were found to be less contaminated than river, sewage, and WWTP effluent. Seasonal dependency was clearly evident for most of the viruses, with peak concentrations in the dry season. JCPyVs and BKPyVs had a poor detection ratio and correspondence with pathogenic viruses. Instead, the frequently proposed PMMoV and the newly proposed TMV were strongly predictive of the pathogen contamination level, particularly in the dry season. We recommend utilizing canal, pond, and groundwater for irrigation to minimize deleterious health effects and propose PMMoV and TMV as indexes to elucidate pathogenic virus levels in environmental samples.






Similar content being viewed by others
References
Abanyie, F., Harvey, R. R., Harris, J. R., Wiegand, R. E., Gaul, L., Desvignes-Kendrick, M., et al. (2015). 2013 multistate outbreaks of Cyclospora cayetanensis infections associated with fresh produce: Focus on the Texas investigations. Epidemiology and Infection, 143(16), 3451–3458. https://doi.org/10.1017/S0950268815000370.
Ahmed, W., Wan, C., Goonetilleke, A., & Gardner, T. (2010). Evaluating sewage-associated JCV and BKV polyomaviruses for sourcing human fecal pollution in a coastal river in Southeast Queensland, Australia. Journal of Environmental Quality, 39(5), 1743–1750. https://doi.org/10.2134/jeq2010.0062.
Albinana-Gimenez, N., Miagostovich, M. P., Calgua, B., Huguet, J. M., Matia, L., & Girones, R. (2009). Analysis of adenoviruses and polyomaviruses quantified by qPCR as indicators of water quality in source and drinking-water treatment plants. Water Research, 43(7), 2011–2019. https://doi.org/10.1016/j.watres.2009.01.025.
Baggi, F., Demarta, A., & Peduzzi, R. (2001). Persistence of viral pathogens and bacteriophages during sewage treatment: Lack of correlation with indicator bacteria. Research Microbiology, 152(8), 743–751.
Balique, F., Colson, P., Barry, A. O., Nappez, C., Ferretti, A., Moussawi, K. Al, et al. (2013). Tobacco mosaic virus in the lungs of mice following intra-tracheal inoculation. PLoS ONE, 8, 1–12. https://doi.org/10.1371/journal.pone.0054993.
Benabbes, L., Ollivier, J., Schaeffer, J., Parnaudeau, S., Rhaissi, H., Nourlil, J., et al. (2013). Norovirus and other human enteric viruses in moroccan shellfish. Food and Environmental Virology, 5(1), 35–40.
Bernard, H., Faber, M., Wilking, H., Haller, S., Höhle, M., Schielke, A., et al. (2014). Large multistate outbreak of norovirus gastroenteritis associated with frozen strawberries, Germany, 2012. Euro Surveillance: Bulletin Européen Sur Les Maladies Transmissibles =. European Communicable Disease Bulletin, 19(8), 20719. https://doi.org/10.2807/1560-7917.ES2014.19.8.20719.
Bouwknegt, M., Verhaelen, K., Rzezutka, A., Kozyra, I., Maunula, L., von Bonsdorff, C., et al. (2015). Quantitative farm-to-fork risk assessment model for norovirus and hepatitis a virus in european leafy green vegetable and berry fruit supply chains. International Journal of Food Microbiology, 198, 50–58.
Callejón, R. M., Rodríguez-Naranjo, M. I., Ubeda, C., Hornedo-Ortega, R., Garcia-Parrilla, M. C., & Troncoso, A. M. (2015). Reported foodborne outbreaks due to fresh produce in the United States and European Union: trends and causes. Foodborne Pathogens and Disease, 12(1), 32–38. https://doi.org/10.1089/fpd.2014.1821.
Cheong, S., Lee, C., Song, S. W., Choi, W. C., Lee, C. H., & Kim, S. J. (2009). Enteric viruses in raw vegetables and groundwater used for irrigation in South Korea. Applied Environmental Microbiology, 75(24), 7745–7751. https://doi.org/10.1128/AEM.01629-09.
Colson, P., Richet, H., Desnues, C., Balique, F., Moal, V., Grob, J., et al. (2010). Pepper mild mottle virus, a plant virus associated with specific immune responses, fever, abdominal pains, and pruritus in humans. PLoS ONE, 5(4), e10041.
da Silva, A. K., Le Saux, J.-C., Parnaudeau, S., Pommepuy, M., Elimelech, M., & Le Guyader, F. S. (2007). Evaluation of removal of noroviruses during wastewater treatment, using real-time reverse transcription-PCR: Different behaviors of genogroups I and II. Applied and Environmental Microbiology, 73(24), 7891–7897.
Drexler, J. F., Baumgarte, S., de Souza Luna, L. K., Eschbach-Bludau, M., Lukashev, A. N., & Drosten, C. (2011). Aichi virus shedding in high concentrations in patients with acute diarrhea. Emerging Infectious Diseases, 17(8), 1544–1547. https://doi.org/10.3201/eid1708.101556.
El-Senousy, W. M., Costafreda, M. I., Pintó, R. M., & Bosch, A. (2013). Method validation for norovirus detection in naturally contaminated irrigation water and fresh produce. International Journal of Food Microbiology, 167(1), 74–79. https://doi.org/10.1016/j.ijfoodmicro.2013.06.023.
Garner, D., & Kathariou, S. (2016). Fresh produce-associated listeriosis outbreaks, sources of concern, teachable moments, and insights. Journal of Food Protection, 2(2), 337–344. https://doi.org/10.4315/0362-028X.JFP-15-387.
Gerba, P. C., & Choi, Y. P. (2006). Role of irrigation water in crop contamination by viruses. In S. M. Goyal (Ed.), Viruses in Food (pp. 257–263). New York: Springer.
Hamza, I. A., Jurzik, L., Überla, K., & Wilhelm, M. (2011). Evaluation of pepper mild mottle virus, human picobirnavirus and Torque teno virus as indicators of fecal contamination in river water. Water Research, 45(3), 1358–1368. https://doi.org/10.1016/j.watres.2010.10.021.
Hanning, I. B., Nutt, J. D., & Ricke, S. C. (2009). Salmonellosis outbreaks in the United States due to fresh produce: Sources and potential intervention measures. Foodborne Pathogens and Disease, 6(6), 635–648. https://doi.org/10.1089/fpd.2008.0232.
Haramoto, E., Katayama, H., Asami, M., & Akiba, M. (2012). Development of a novel method for simultaneous concentration of viruses and protozoa from a single water sample. Journal of Virological Methods, 182(1–2), 62–69. https://doi.org/10.1016/j.jviromet.2012.03.011.
Haramoto, E., & Kitajima, M. (2017). Quantification and genotyping of Aichi Virus 1 in water samples in the Kathmandu Valley, Nepal. Food and Environmental Virology, 9(3), 350–353. https://doi.org/10.1007/s12560-017-9283-7.
Haramoto, E., Kitajima, M., Katayama, H., & Ohgaki, S. (2010). Real-time PCR detection of adenoviruses, polyomaviruses, and torque teno viruses in river water in Japan. Water Research, 44(6), 1747–1752. https://doi.org/10.1016/j.watres.2009.11.043.
Haramoto, E., Kitajima, M., Kishida, N., Konno, Y., Katayama, H., Asami, M., et al. (2013a). Occurrence of pepper mild mottle virus in drinking water sources in Japan. Applied and Environmental Microbiology, 79(23), 7413–7418.
Haramoto, E., Kitajima, M., & Otagiri, M. (2013b). Development of a reverse transcription-quantitative PCR assay for detection of salivirus/klassevirus. Applied and Environmental Microbiology, 79(11), 3529–3532. https://doi.org/10.1128/AEM.00132-13.
Haramoto, E., & Otagiri, M. (2013). Prevalence and genetic diversity of klassevirus in wastewater in Japan. Food and Environmental Virology, 5(1), 46–51.
Haramoto, E., & Otagiri, M. (2014). Occurrence of human cosavirus in wastewater and river water in Japan. Food and Environmental Virology, 6(1), 62–66.
Haramoto, E., Yamada, K., & Nishida, K. (2011). Prevalence of protozoa, viruses, coliphages and indicator bacteria in groundwater and river water in the Kathmandu Valley, Nepal. Transactions of the Royal Society of Tropical Medicine and Hygiene, 105(12), 711–716. https://doi.org/10.1016/j.trstmh.2011.08.004.
Hata, A., Kitajima, M., & Katayama, H. (2013). Occurrence and reduction of human viruses, F-specific RNA coliphage genogroups and microbial indicators at a full-scale wastewater treatment plant in Japan. Journal of Applied Microbiology, 114(2), 545–554. https://doi.org/10.1111/jam.12051.
Heim, A., Ebnet, C., Harste, G., & Pring-Åkerblom, P. (2003). Rapid and quantitative detection of human adenovirus DNA by real-time PCR. Journal of Medical Virology, 70(2), 228–239. https://doi.org/10.1002/jmv.10382.
Hoa-Tran, T. N., Nakagomi, T., Sano, D., Sherchand, J. B., Pandey, B. D., Cunliffe, N. A., et al. (2015). Molecular epidemiology of noroviruses detected in Nepalese children with acute diarrhea between 2005 and 2011: Increase and predominance of minor genotype GII.13. Infection, Genetics and Evolution, 30, 27–36. https://doi.org/10.1016/j.meegid.2014.12.003.
Hu, Q., Niu, Y., Zhang, K., Liu, Y., & Zhou, X. (2011). Virus-derived transgenes expressing hairpin RNA give immunity to Tobacco mosaic virus and Cucumber mosaic virus. Virology Journal, 8(1), 41. https://doi.org/10.1186/1743-422X-8-41.
IHME. (2010). GBD Profile: Nepal. Institute for Health Metrics and Evaluation, Seattle, WA 98121 USA. Retrieved April 15, 2015, from https://www.healthdata.org/sites/default/files/files/country_profiles/GBD/ihme_gbd_country_report_nepal.pdf.
Jothikumar, N., Kang, G., & Hill, V. R. (2009). Broadly reactive TaqMan® assay for real-time RT-PCR detection of rotavirus in clinical and environmental samples. Journal of Virological Methods, 155(2), 126–131. https://doi.org/10.1016/j.jviromet.2008.09.025.
Kageyama, T., Kojima, S., Shinohara, M., Uchida, K., Fukushi, S., Hoshino, F. B., et al. (2003). Broadly reactive and highly sensitive assay for Norwalk-like viruses based on real-time quantitative reverse transcription-PCR. Journal of Clinical Microbiology, 41(4), 1548–1557. https://doi.org/10.1128/JCM.41.4.1548.
Katayama, H., Shimasaki, A., & Ohgaki, S. (2002). Development of a virus concentration method and its application to detection of enterovirus and norwalk virus from Coastal Seawater development of a virus concentration method and its application to detection of enterovirus and Norwalk Virus from Coastal. Applied Environmental Microbiology, 68(3), 1033–1039. https://doi.org/10.1128/AEM.68.3.1033.
Kirby, A. E., Shi, J., Montes, J., Lichtenstein, M., & Moe, C. L. (2014). Disease course and viral shedding in experimental Norwalk Virus and Snow Mountain Virus infection. Journal of Medical Virology, 86(12), 2055–2064.
Kitajima, M., & Gerba, C. (2015). Aichi virus 1: Environmental occurrence and behavior. Pathogens, 4(2), 256–268. https://doi.org/10.3390/pathogens4020256.
Kitajima, M., Haramoto, E., Phanuwan, C., Katayama, H., & Furumai, H. (2012). Molecular detection and genotyping of human noroviruses in influent and effluent water at a wastewater treatment plant in Japan. Journal of Applied Microbiology, 112(3), 605–613. https://doi.org/10.1111/j.1365-2672.2012.05231.x.
Kitajima, M., Hata, A., Yamashita, T., Haramoto, E., Minagawa, H., & Katayama, H. (2013). Development of a reverse transcription-quantitative PCR system for detection and genotyping of aichi viruses in clinical and environmental samples. Applied and Environmental Microbiology, 79(13), 3952–3958.
Kitajima, M., Iker, B. C., Pepper, I. L., & Gerba, C. P. (2014). Relative abundance and treatment reduction of viruses during wastewater treatment processes—Identification of potential viral indicators. Science of the Total Environment, 488–489, 290–296. https://doi.org/10.1016/j.scitotenv.2014.04.087.
Kitajima, M., Oka, T., Takagi, H., Tohya, Y., Katayama, H., Takeda, N., et al. (2010). Development and application of a broadly reactive real-time reverse transcription-PCR assay for detection of murine noroviruses. Journal of Virological Methods, 169(2), 269–273. https://doi.org/10.1016/j.jviromet.2010.07.018.
Koening, R., & Lesemann, D. E. (1985). Plant viruses in German Rivers and Lakes. I. Tombusviruses, a Potexvirus and Carnation Mottle Virus. Journal of Phytopathology, 112(2), 105–116.
Kurokawa, M., Ono, K., Nukina, M., Thapa, U., & Rai, S. K. (2004). Detection of diarrheagenic viruses from diarrheal fecal samples collected from children in Kathmandu, Nepal. Nepal Medical College Journal, 6(1), 17–23.
MoHP Nepal, New Era. (2007). Nepal Demographic and Health Survey 2006, Final Report. Ministry of Health and Population, 1–192.
Pal, A., Sirota, L., Maudru, T., Peden, K., & Lewis, A. M. (2006). Real-time, quantitative PCR assays for the detection of virus-specific DNA in samples with mixed populations of polyomaviruses. Journal of Virological Methods, 135(1), 32–42. https://doi.org/10.1016/j.jviromet.2006.01.018.
Piazzolla, P., Castellano, M. A., & Stradis, A. (1986). Presence of plant Viruses in some Rivers of Southern Italy. Journal of Phytopathology, 116(3), 244–246. https://doi.org/10.1111/j.1439-0434.1986.tb00917.x.
Pleše, N., Juretić, N., Mamula, D., Polák, Z., & Krajačić, M. (1996). Plant viruses in soil and water of forest ecosystems in Croatia. Phyton—Annales Rei Botanicae, 36(1), 135–143.
Rachmadi, A. T., Torrey, J. R., & Kitajima, M. (2016). Human polyomavirus: Advantages and limitations as a human-specific viral marker in aquatic environments. Water Research, 105, 456–469. https://doi.org/10.1016/j.watres.2016.09.010.
Rosario, K., Symonds, E. M., Sinigalliano, C., Stewart, J., & Breitbart, M. (2009). Pepper mild mottle virus as an indicator of fecal pollution. Applied and Environmental Microbiology, 75(22), 7261–7267. https://doi.org/10.1128/AEM.00410-09.
Rutkowski, T., Raschid-Sally, L., & Buechler, S. (2007). Wastewater irrigation in the developing world—Two case studies from the Kathmandu Valley in Nepal. Agricultural Water Management, 88(1–3), 83–91. https://doi.org/10.1016/j.agwat.2006.08.012.
Rzezutka, A., & Cook, N. (2004). Survival of human enteric viruses in the environment and food. FEMS Microbiology Reviews, 28(4), 441–453. https://doi.org/10.1016/j.femsre.2004.02.001.
Scott, C. A., Faruqui, N. I., & Raschid-Sally, L. (2004). Wastewater use in irrigated agriculture: Confronting the livelihood and environmental realities. Irrigation and Drainage Systems. https://doi.org/10.1079/9780851998237.0000.
Shieh, Y. S. C., Wait, D., Tai, L., & Sobsey, M. D. (1995). Methods to remove inhibitors in sewage and other fecal wastes for enterovirus detection by the polymerase chain reaction. Journal of Virological Methods, 54(1), 51–66. https://doi.org/10.1016/0166-0934(95)00025-P.
Shrestha, S., Haramoto, E., Sherchand, J. B., Hada, S., Rajbhandari, S., & Shindo, J. (2016a). Prevalence of protozoa and indicator Bacteria in wastewater irrigation sources in Kathmandu Valley, Nepal: Cases from Kirtipur, Bhaktapur, and Madhyapur Thimi Municipalities. Journal of Water Environment Technology, 14(3), 149–157. https://doi.org/10.2965/jwet.15-047.
Shrestha, S., Haramoto, E., Sherchand, J. B., Rajbhandari, S., Prajapati, M., & Shindo, J. (2016b). Seasonal variation of microbial quality of irrigation water in different sources in the Kathmandu Valley, Nepal. Naresuan University Engineering Journal, 11(1), 57–62.
Shukla, A. (2012). Wastewater production, treatment and use in Nepal. Retrieved April 15, 2017., from https://www.ais.unwater.org/ais/pluginfile.php/232/mod_page/content/134/Nepal_CountryPaper.pdf.
Sidhu, J. P. S., Hodgers, L., Ahmed, W., Chong, M. N., & Toze, S. (2012). Prevalence of human pathogens and indicators in stormwater runoff in Brisbane, Australia. Water Research, 46(20), 6652–6660. https://doi.org/10.1016/j.watres.2012.03.012.
Thebault, A., Teunis, F. M., Pendu, J. L., Guyader, F. S. L., & Denis, J. B. (2013). Infectivity of GI and GII noroviruses established from oyster related outbreaks. Epidemics, 5(2), 98–110.
Uchida, R., Pandey, B. D., Sherchand, J. B., Ahmed, K., Yokoo, M., Nakagomi, T., et al. (2006). Molecular epidemiology of rotavirus diarrhea among children and adults in Nepal: Detection of G12 strains with P[6] or P[8] and a G11P[25] strain. Journal of Clinical Microbiology, 44(10), 3499–3505. https://doi.org/10.1128/JCM.01089-06.
van Lier, J. B., & Huibers, F. P. (2010). From unplanned to planned agricultural use: Making an asset out of wastewater. Irrigation and Drainage Systems, 24(1–2), 143–152. https://doi.org/10.1007/s10795-009-9090-x.
Wetter, C., Conti, M., Altschuh, D., Tabillion, R., & van Regenmortel, M. H. V. (1984). Pepper mild mottle virus, a tobamovirus infecting pepper cultivars in Sicily. Phytopathology, 74(4), 405–410. https://doi.org/10.1094/Phyto-74-405.
Wyn-Jones, A. P., Carducci, A., Cook, N., D’Agostino, M., Divizia, M., Fleischer, J., et al. (2011). Surveillance of adenoviruses and noroviruses in European recreational waters. Water Research, 45(3), 1025–1038. https://doi.org/10.1016/j.watres.2010.10.015.
You, D., Hug, L., & Chen, Y. (2014). Levels & trends in child mortality. United Nations Child. Fund 3–5. https://dx.doi.org/10.1016/S0140-6736(10)60703-9. Retrieved from https://www.unicef.org/media/files/Levels_and_Trends_in_Child_Mortality_2014.pdf.
Zhang, T., Breitbart, M., Lee, W. H., Run, J. Q., Wei, C. L., Soh, S. W. L., et al. (2006). RNA viral community in human feces: Prevalence of plant pathogenic viruses. PLoS Biology, 4(1), 0108–0118. https://doi.org/10.1371/journal.pbio.0040003.
Acknowledgements
This study was financially supported by the Science and Technology Research Partnership for Sustainable Development (SATREPS) Programs led by Professor Futaba Kazama of the University of Yamanashi. It is also supported by the Grant-in Aid for Scientific Research (B) (No. 17H03332) from the Japan Society for the Promotion of Science (JSPS). The authors would like to thank members of Medical Microbiology and Public Health Research laboratory of Institute of Medicine in Nepal for helping to conduct microbial analysis.
Funding
This study was financially supported by the Science and Technology Research Partnership for Sustainable Development (SATREPS) Programs entitled ‘Hydro-microbiological Approach for Water Security in Kathmandu Valley, Nepal’ led by Professor Futaba Kazama of the University of Yamanashi which is jointly funded by the Japan International Cooperation Agency (JICA) and the Japan Science and Technology Agency (JST). It is also supported by the Grant-in Aid forthe Scientific Research (B) (No. 17H03332) from the Japan Society for the Promotion of Science (JSPS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Shrestha, S., Shrestha, S., Shindo, J. et al. Virological Quality of Irrigation Water Sources and Pepper Mild Mottle Virus and Tobacco Mosaic Virus as Index of Pathogenic Virus Contamination Level. Food Environ Virol 10, 107–120 (2018). https://doi.org/10.1007/s12560-017-9324-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12560-017-9324-2