Abstract
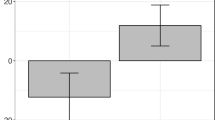
Current research in cerebellar cognitive and linguistic functions makes plausible the idea that the cerebellum is involved in processing temporally contiguous linguistic input. In order to assess this hypothesis, a lexical decision task was constructed to study the effects of cerebellar transcranial magnetic stimulation on semantic noun-to-verb priming based on association (e.g. ‘soap–cleaning’) or similarity (e.g. ‘robbery–stealing’). The results demonstrated a selective increase in associative priming size after stimulation of a lateral cerebellar site. The findings are discussed in the contexts of a cerebellar role in linguistic expectancy generation and the corticocerebellar ‘prefrontal’ reciprocal loop.


Similar content being viewed by others
Notes
A simple node-counting scheme (path). The relatedness score is inversely proportional to the number of nodes along the shortest path between the synsets. The shortest possible path occurs when the two synsets are the same, in which case the length is 1. Thus, the maximum relatedness value is 1.
As the software cannot assess the semantic relatedness of words of different grammatical categories (here, nouns and verbs), it was ensured that, in each pair, the noun was homonymous with a verb of the same basic semantic properties, e.g. ‘release’, ‘fear’.
Of course, semantic associative relatedness implies that the forms may co-occur, be it in a non-immediate fashion. However, such loose co-occurrences could easily be found in the categorically related pairs as well. For example, in (1) below, the semantic associatively related pair ‘stripper-entertaining’ (a) can loosely co-occur in speech, like the semantic categorically related one ‘murder-killing’ (b).
(a)‘[…] being a male stripper isn’t just about taking clothes off, it’s about entertaining people […]’
(b)‘It would seem that one is guilty of murder through killing someone by chance […]’ (Google search)
Indeed, the priming sizes of the three participants that had participated in earlier versions of this task were significantly larger overall than those of the rest of the no TMS subjects (Relatedness × Experience with task: F (1, 43) = 5.09, MSe = 342.85, p < 0.03; a large priming effect for the Experienced group: Relatedness: F (1, 1) = 19,063.58, MSe = 0.20, p < 0.005).
Distribution of subjects across lists: list A: n = 12; list B: n = 13; list C: n = 11; list D: n = 10; lateral group: A 3; B 3; C 3; D 3; medial group: A 3; B 4; C 2; D 2; no TMS group 1: A 3; B 3; C 3; D 3; no TMS group 2: A 3; B 3; C 3; D 2.
Abbreviations
- ANOVA:
-
Analysis of variance
- cm:
-
Centimetre
- mm:
-
Millimetre
- ms:
-
Millisecond(s)
- (r)TMS:
-
(Repetitive) transcranial magnetic stimulation
- SD:
-
Standard deviation
- SEM:
-
Standard error of the mean
- BNC:
-
British National Corpus
References
Argyropoulos GP. Neocerebellar emulation in language processing. In: Alter K, Horne M, Lindgren M, Roll M, von Koss Torkildsen J, editors. Brain talk: discourse with and in the brain. Papers from the first Birgit Rausing language program conference in linguistics. Lund: Lund University, Media Tryck; 2009. p. 193–206.
Argyropoulos GP. Cerebellar theta-burst stimulation selectively enhances lexical associative priming. Cerebellum. 2011;10(3):540–50.
Argyropoulos GP. Cortico-cortical and cortico-cerebellar computations in language change. In: Scott-Phillips TC, Tamariz M, Cartmill EA, Hurford JR, editors. The evolution of language: proceedings of the 7th international conference on the evolution of language. Singapore: World Scientific; 2012. p. 11–8.
Argyropoulos GP, Kimiskidis V, Papagiannopoulos S. Theta-burst stimulation of the right neocerebellar vermis selectively disrupts the practice-induced acceleration of lexical decisions. Behav Neurosci. 2011;125(5):724–34.
Bellebaum C, Daum I. Mechanisms of cerebellar involvement in associative learning. Cortex. 2011;47:128–36.
Canavan AG, Sprengelmeyer R, Diener HC, Hömberg V. Conditional associative learning is impaired in cerebellar disease in humans. Behav Neurosci. 1994;108:475–85.
Christian KM, Thompson RF. Long-term storage of an associative memory trace in the cerebellum. Behav Neurosci. 2005;119:526–37.
Courchesne E, Allen G. Prediction and preparation, fundamental functions of the cerebellum. Learn Mem. 1997;4:1–35.
Daum I, Ackermann H, Schugens MM, Reimold C, Dichgans J, Birbaumer N. The cerebellum and cognitive functions in humans. Behav Neurosci. 1993;107:411–9.
Del Olmo MF, Cheeran B, Koch G, Rothwell JC. Role of the cerebellum in externally paced rhythmic finger movements. J Neurophysiol. 2007;98:145–52.
Demirtas-Tatlidede A, Freitas C, Cromer JR, Safar L, Ongur D, Stone WS, Seidman LJ, Schmahmann JD, Pascual-Leone A. Safety and proof of principle study of cerebellar vermal theta burst stimulation in refractory schizophrenia. Schizophr Res. 2010;124:91–100.
Devlin JT, Watkins KE. Stimulating language: insights from TMS. Brain. 2007;130:610–22.
Drepper J, Timmann D, Kolb FP, Diener HC. Non- motor associative learning in patients with isolated degenerative cerebellar disease. Brain. 1999;122:87–97.
Fellbaum C (ed) (1998) WordNet: an electronic lexical database. Cambridge: MIT. http://wordnet.princeton.edu/
Ferretti TR, McRae K, Hatherell A. Integrating verbs, situation schemas, and thematic role concepts. J Mem Lang. 2001;44:516–47.
Ferretti TR, Gagné C, McRae K. Thematic role focusing by participle inflections: evidence from conceptual combination. J Exp Psychol Learn Mem Cogn. 2003;29(1):118–27.
Ferretti TR, Kutas M, McRae K. Verb aspect and the activation of event knowledge. J Exp Psychol Learn Mem Cogn. 2007;33(1):182–96.
Ferrucci R, Marceglia S, Vergari M, Cogiamanian F, Mrakic-Sposta S, Mameli F, Zago S, Barbieri S, Priori A. Cerebellar transcranial direct current stimulation impairs the practice-dependent proficiency increase in working memory. J Cogn Neurosci. 2008;20:1687–97.
Fierro B, Giglia G, Palermo A, Pecoraro C, Scalia S, Brighina F. Modulatory effects of 1 Hz rTMS over the cerebellum on motor cortex excitability. Exp Brain Res. 2007;176:440–7.
Fiez JA, Petersen SE, Cheney MK, Raichle ME. Impaired non-motor learning and error detection associated with cerebellar damage. A single case study. Brain. 1992;115:155–78.
Fiez JA, Raichle M. Linguistic processing. Int Rev Neurobiol. 1997;41:233–54.
Forster KI, Davis C. The density constraint on form-priming in the naming task: interference from a masked prime. J Mem Lang. 1991;30:1–25.
Frings M, Dimitrova A, Schorn CF, Elles H-G, Hein-Kropp C, Gizewski ER, Diener HC, Timmann D. Cerebellar involvement in verb generation: an fMRI study. Neurosci Lett. 2006;409:19–23.
Gebhart AL, Petersen SE, Thach WT. Role of the posterolateral cerebellum in language. Ann NY Acad Sci. 2002;978:318–33.
Hagoort P. Semantic priming in Broca’s aphasics at a short SOA: no support for an automatic access deficit. Brain Lang. 1997;56:287–300.
Hashimoto M, Ohtsuka K. Transcranial magnetic stimulation over the posterior cerebellum during visual saccades in man. Brain. 1995;118:1185–93.
Huang Y, Edwards MJ, Rounis E, Bhatia KP, Rothwell JC. Theta burst stimulation of the human motor cortex. Neuron. 2005;45:201–6.
Jarvis BG. DirectRT (Version 2008.1.0.11) [Computer software]. New York: Empirisoft Corporation; 2008.
Kalla R, Muggleton NG, Cowey A, Walsh V. Human dorsolateral prefrontal cortex is involved in visual search for conjunctions but not features: a theta TMS study. Cortex. 2009;45:1085–109.
Kelly RM, Strick PL. Cerebellar loops with motor cortex and prefrontal cortex of a nonhuman primate. J Neurosci. 2003;23:8432–44.
Koch G, Mori F, Marconi B, Codecà C, Pecchioli C, Salerno, Torriero S, Lo Gerfo E, Mir P, Oliveri M, Caltagirone C. Changes in intracortical circuits of the human motor cortex following theta burst stimulation of the lateral cerebellum. Clin Neurophysiol. 2008;119:2559–69.
Koch G, Brusa L, Carrillo F, Lo Gerfo E, Torriero S, Oliveri M, Mir P, Caltagirone C, Stanzione P. Cerebellar magnetic stimulation decreases levodopa-induced dyskinesias in Parkinson disease. Neurology. 2009;73:113–9.
Larsell O, Jansen J. The comparative anatomy and histology of the cerebellum, Vol 3 III: the human cerebellum, cerebellar connections, and cerebellar cortex. Minneapolis: University of Minnesota Press; 1972.
Leech G. 100 million words of English: the British National Corpus. Lang Res. 1992;28(1):1–13.
Leggio MG, Silveri MC, Petrosini L, Molinari M. Phonological grouping is specifically affected in cerebellar patients: a verbal fluency study. J Neurol Neurosurg Psychiatry. 2000;69:102–6.
Liu CL, Tseng P, Chiau HY, Liang WK, Hung DL, Tzeng OJ, Muggleton NG, Juan CH. The location probability effects of saccade reaction times are modulated in the frontal eye fields but not in the supplementary eye field. Cereb Cortex. 2010;21(6):1416–25.
McCormick DA, Thompson RF. Cerebellum: essential involvement in the classically conditioned eyelid response. Science. 1984;223:296–9.
McRae K, Hare M, Elman JL, Ferretti T. A basis for generating expectancies for verbs from nouns. Mem Cognit. 2005;33:1174–84.
Miall RC, King D. State estimation in the cerebellum. Cerebellum. 2008;7:572–6.
Middleton FA, Strick PL. Cerebellar projections to the prefrontal cortex of the primate. J Neurosci. 2001;21:700–12.
Moss HE, Ostrin RK, Tyler LK, Marslen-Wilson WD. Accessing different types of lexical semantic information: evidence from priming. J Exp Psychol Learn Mem Cogn. 1995;21(4):863–83.
Ober B. RT and non-RT methodology for semantic priming research with Alzheimer’s disease patients: a critical review. J Clin Exp Neuropsychol. 2002;24:883–911.
Ohtsuka K, Enoki T. Transcranial magnetic stimulation over the posterior cerebellum during smooth pursuit eye movements in man. Brain. 1998;121:429–35.
Oliveri M, Koch G, Torriero S, Caltagirone C. Increased facilitation of the primary motor cortex following 1 Hz repetitive transcranial magnetic stimulation of the contralateral cerebellum in normal humans. Neurosci Lett. 2005;376:188–93.
Oliveri M, Torriero S, Koch G, Salerno S, Petrosini L, Caltagirone C. The role of transcranial magnetic stimulation in the study of cerebellar cognitive function. Cerebellum. 2007;6:95–101.
Oliveri M, Bonni S, Turriziani P, Koch G, Lo Gerfo E, Torriero S, Vicario CM, Petrosini L, Caltagirone C. Motor and linguistic linking of space and time in the cerebellum. PLoS One. 2009;4(11):e7933.
Palmer ED, Rosen HJ, Ojemann JG, Buckner RL, Kelley WM, Petersen SE. An event-related fMRI study of overt and covert word stem completion. NeuroImage. 2001;14:182–93.
Paulin M. Neural representations of moving systems. In: Schmahmann JD, editor. The cerebellum and cognition, international review of neurobiology, vol. 41. San Diego: Academic; 1997. p. 515–33.
Pedersen T, Patwardhan S, Michelizzi J. WordNet::similarity—measuring the relatedness of concepts. AAAI. 2004;04:1024–5.
Perea M, Gotor A. Associative and semantic priming effects occur at very short stimulus-onset asynchronies in lexical decision and naming. Cognition. 1997;62:223–40.
Perea M, Rosa E. The effects of associative and semantic priming in the lexical decision task. Psychol Res. 2002;66:180–94.
Petersen SE, Fox PT, Posner ML, Mintun M, Raichle ME. Positron emission tomographic studies of the processing of single words. J Cogn Neurosci. 1989;1:153–70.
Pollatsek A, Well AD. On the use of counterbalanced designs in cognitive research: a suggestion for a better and more powerful analysis. J Exp Psychol Learn Mem Cogn. 1995;21:785–94.
Ramnani N. The primate cortico-cerebellar system: anatomy and function. Nat Rev Neurosci. 2006;7:511–22.
Rogers SL, Friedman RB. The underlying mechanisms of semantic memory in Alzheimer’s disease and semantic dementia. Neuropsychologia. 2008;46:12–21.
Sack AT. Transcranial magnetic stimulation, causal structure–function mapping and networks of functional relevance. Curr Opin Neurobiol. 2006;16:593–9.
Sack AT, Kadosh RC, Schuhmann T, Moerel M, Walsh V, Goebel R. Optimizing functional accuracy of TMS in cognitive studies: a comparison of methods. J Cogn Neurosci. 2008;21(2):207–21.
Schmahmann JD. The role of the cerebellum in affect and psychosis. J Neurolinguistics. 2000;13:189–214.
Stewart LM, Walsh V, Rothwell JC. Motor and phosphine thresholds: a transcranial magnetic stimulation correlation study. Neuropsychologia. 2001;39:415–9.
Théoret H, Haque J, Pascual-Leone A. Increased variability of paced finger tapping accuracy following repetitive magnetic stimulation of the cerebellum in humans. Neurosci Lett. 2001;306:29–32.
Thompson-Schill SL, Kurtz KJ, Gabrieli JDE. Effects of semantic and associative relatedness on automatic priming. J Mem Lang. 1998;38:440–58.
Timmann D, Drepper J, Calabrese S, Bürgerhoff K, Maschke M, Kolb FP, Daum I, Diener HC. Use of sequence information in associative learning in control subjects and cerebellar patients. Cerebellum. 2004;3:75–82.
Timmann D, Drepper J, Frings M, Maschke M, Richter S, Gerwig M, Kolb FP. The human cerebellum contributes to motor, emotional and cognitive associative learning. A review. Cortex. 2010;46:845–57.
Timmann D, Drepper J, Maschke M, Kolb FP, Böring D, Thilmann AF, Diener HC. Motor deficits cannot explain impaired cognitive associative learning in cerebellar patients. Neuropsychologia. 2002;40:788–800.
Torriero S, Oliveri M, Koch G, Caltagirone C, Petrosini L. Interference of left and right cerebellar rTMS with procedural learning. J Cogn Neurosci. 2004;16:1605–11.
Torriero S, Oliveri M, Koch G, Lo Gerfo E, Salerno S, Petrosini L, Caltagirone C. Cortical networks of procedural learning: evidence from cerebellar damage. Neuropsychologia. 2007;45:1208–14.
Torriero S, Oliveri M, Koch G, Lo Gerfo E, Salerno S, Ferlazzo F, Caltagirone C, Petrosini L. Changes in cerebello-motor connectivity during procedural learning by actual execution and observation. J Cogn Neurosci. 2010;23(2):338–48.
Tucker J, Harding AE, Jahanshahi M, Nixon PD, Rushworth M, Quinn NP, Thompson PD, Passingham RE. Associative learning in patients with cerebellar ataxia. Behav Neurosci. 1996;110:1229–34.
Ugawa Y, Uesaka Y, Terao Y, Hanajima R, Kanazawa I. Magnetic stimulation over the cerebellum in humans. Ann Neurol. 1995;37:703–13.
Vallesi A, Shallice T, Walsh V. Role of the prefrontal cortex in the foreperiod effect: TMS evidence for dual mechanisms in temporal preparation. Cereb Cortex. 2007;17:466–74.
Wolpert DM, Miall RC, Kawato M. Internal models in the cerebellum. Trends Cogn Sci. 1998;2:338–47.
Acknowledgments
The authors would like to thank Dr. Thomas Bak, Prof. Simon Garrod, Prof. Jim Hurford, Prof. Vassilios Kimiskidis, Prof. Maria Leggio, Prof. Ken McRae, Prof. Massimo Oliveri, Prof. Martin Pickering, Prof. Richard Shillcock, Prof. Steve Small, Prof Ana Solodkin, Dr. Patrick Sturt, as well as the reviewers of the journal for their precious support and encouragement. NGM was supported by the UK Medical Research Council and by the National Science Council, Taiwan (100-2410-H-008-074-MY3).
Conflict of Interest
There were no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Argyropoulos, G.P., Muggleton, N.G. Effects of Cerebellar Stimulation on Processing Semantic Associations. Cerebellum 12, 83–96 (2013). https://doi.org/10.1007/s12311-012-0398-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-012-0398-y