Abstract
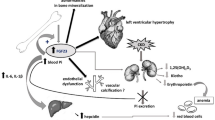
The roles of calcitonin, parathormone and calcitriol in the regulation of plasma calcium and phosphate are well-established. However, in autosomal-dominant hypophosphatemic rickety patients, studies have revealed normal plasma levels of calcium, associated with normal thyroid and parathyroid functions, but decreased levels of phosphate and calcitriol despite adequate reserves of vitamin D. Also, in tumoral calcinosis, persistent hyperphosphatemia with increased levels of 1,25(OH)2D3 have been observed. These studies indicate the involvement of factors other than the ones already known. The first decade of this century/millennium has led to the discovery of the involvement of fibroblast growth factor-23, furin protease and α-klotho in the homeostasis of calcium and phosphate, which is the subject of this mini-review.
Similar content being viewed by others
References
Winkler DG, Sutherland MK, Geoghegan JC, Yu C, Hayes T, Skonier JE, Shpektor D, Jonas M, Kovacevich BR, Staehling-Hampton K, Appleby M, Brunkow ME, Latham JA. Osteocyte control of bone formation via sclerostin, a novel BMP antagonist. EMBO J. 2003;22:6267–76.
Perwad F, Azam N, Zhang MYH, Yamashita T, Tenenhouse HS, Portale AA. Dietary and serum phosphorus regulate fibroblast growth factor-23 expression and 1,25-dihydroxyvitamin D metabolism in mice. Endocrinology. 2005;146:5358–64.
Kida Y, Fukumoto S, Yamashita T, Jonsson K, Econs M, Juppner H. Fibroblast growth factor-23 in oncogenic osteomalacia and X-linked hypophosphatemia. New Engl J Med. 2003;349:505–6.
Jonsson KB, Zahradnik R, Larsson T, White KE, Sugimoto T, Imanishi Y, Yamamoto T, Hampson G, Koshiyama H, Lunggren O, Oba K, Yang IM, Miyauchi A, Econs MJ, Juppner H. Fibroblast growth factor-23 in oncogenic osteomalacia and X-linked hypophosphatemia. New Engl J Med. 2003;348:1656–63.
Rodriguez M, Lopez I, Munoz J, Aguilera-Tejero E, Almaden Y. FGF23 and mineral metabolism, implications in CKD-MBD. Nefroligia. 2012;32(3):275–8.
Larson T, Davis SI, Garringer HJ, Mooney SD, Draman MS, Cullen MJ, White KE. Fibroblast growth factor-23 mutants causing familial tumoral calcinosis are differentially processed. Endocrinology. 2005;146:3883–91.
Bergwitz C, Banerjee S, Abu-Zahra H, Kaji H, Miyauchi A, Sugimoto T, Juppner H. Defective O-glycosylation due to a novel homozygous S129P mutation is associated with lack of fibroblast growth factor-23 secretion and tumoral calcinosis. J Clin Endocrinol Metab. 2009;94(11):4267–74.
Nabeshima Y. Regulation of calcium homeostasis. Clin Calcium. 2010;20(11):1677–85.
Antonuicci DM, Yamashita T, Portale AA. Dietary phosphorus regulates serum fibroblast growth factor-23 concentrations in healthy men. J Clin Endocrinol Metab. 2006;91:3144–9.
Ubaidus S, Li M, Sultana S, de Freitas PHL, Oda K, Maeda T, Takagi R, Amizuka N. FGF-23 is mainly synthesized by osteocytes in the regularly distributed osteocytic lacunar canalicular system established after physiological bone remodeling. J Electron Microsc (Tokyo). 2009;58:381–92.
Liu S, Quarles LD. How fibroblast growth factor-23 works. J Am Soc Nephrol. 2007;18(6):1637–47.
Perward F, Zhang MYH, Tenehouse HS, Portale. Fibroblast growth factor-23 impairs phosphorus and vitamin D metabolism in vivo and suppresses 25-hydroxy vitamin D 1-α hydroxylase expression in vitro. Am J Renal Physiol. 2007;293:1577–83.
White KE, Larsson TE, Econs MJ. The roles of specific genes implicated as circulating factors involved in normal and disordered phosphate homeostasis: frizzled related protein-4, matrix extracellular phosphoglyco- protein and fibroblast growth factor-23. Endocrinol Rev. 2006;27:221–41.
Gattineni J, Bates C, Twombley K, Dwarkanath V, Robinson ML, Goetz R, Mohammadi M, Baum M. FGF-23 decreases renal NaPi-2a and NaPi-2c expression and induces hypophosphatemia in vivo predominantly via FGF-receptor-1. Am J Physiol Renal Physiol. 2009;297:F282–91.
Ramon I, Kleynen P, Body JJ, Karmali R. Fibroblast growth factor-23 and its role in phosphate homeostasis. Eur J Endocrinol. 2010;162:1–10.
Masi L, Gozzini A, Franchi A, Companacci D, Amedei A, Falchetti A, Franceschelli F, Marcucci G, Tanini A, Capanna R, Brandi ML. A novel recessive mutation of fibroblast growth factor-23 in tumoral calcinosis. J Bone Joint Surg. 2009;91:1190–8.
Nabeshima Y. Discovery of α-klotho and FGF-23 unveiled new insight into calcium and phosphate homeostasis. Clin Calcium. 2008;18(7):923–34.
Razzaque MS, Lanske B. The emerging role of the fibroblast growth factor-23-klotho axis in renal regulation of phosphate homeostasis. J Endocrinol. 2007;194:1–10.
de Groot T, van der Hagen EA, Verkaart S, Te Boekhorst VA, Bindels RJ, Hoenderop JG. The role of the TRPV5 N-terminus in channel activity, tetramerization and trafficking. J Biol Chem. 2011;286(37):32132–9.
Urakawa L, Yamazak Y, Shimada T, Lijima K, Haseqawa H, Okawa K, Fujita T, Fukumoto S, Yamashita T. Klotho converts FGF receptor into a specific receptor for FGF23. Nature. 2006;444:770–4.
Wise RJ, Barr PJ, Wong PA, Kiefer MC, Brake AJ, Kaufman RJ. Expression of a human proprotein processing enzyme: correct cleavage of the von Willebrand factor precursor at a paired basic amino acid site. Proc Natl Acad Sci USA. 1991;87(23):9378–82.
Thomas G. Furin at the cutting edge: from protein traffic to embryogenesis and disease. Nat Rev Mol Cell Biol. 2002;3(10):753–66.
Conflict of Interest
Since this is a single author review article and involving no experimental work, research funding or support, I certify that there is no conflict with anyone in this regard.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mattoo, R.L. The Roles of Fibroblast Growth Factor (FGF)-23, α-Klotho and Furin Protease in Calcium and Phosphate Homeostasis : A Mini-Review. Ind J Clin Biochem 29, 8–12 (2014). https://doi.org/10.1007/s12291-013-0324-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12291-013-0324-1