Abstract
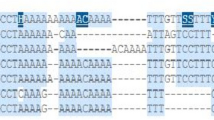
Matrix metalloproteinase-9 (gelatinase B) plays a key role in cancer invasion and metastasis by degrading the extracellular matrix and basement membrane barriers. A cytosine (C) > thymidine (T) single nucleotide polymorphism (SNP) at position −1562 in the MMP-9 promoter is reported to influence the expression of the gene. Genotyping of MMP-9 −1562 C→T promoter polymorphism in 140 gastric cancer patients and 132 healthy control subjects was carried out in order to evaluate its association with progression and development of gastric cancer. The SNP was genotyped by tetra-primer amplification refractory mutation system-polymerase chain reaction followed by agarose gel electrophoresis. Statistical methods were adopted to test for the significance of the results. Risk factor profile of the patients revealed age above 50 years, smoking, alcoholism as the factors associated with the disease. The distribution of genotype frequencies in gastric cancer patients were 28.7 % of CC, 45.5 % of CT and 25.7 % of TT, whereas in control subjects 31.8 % of CC, 53.03 % of CT and 15.15 % of TT, respectively. The allelic frequencies were 51.51 % of C and 48.48 % of T in patient group and 58.33 % of C and 41.66 % of T in controls respectively. The present study shows the possible association of epidemiological risk factors with gastric cancer. There is an increased frequency of T allele in the disease compared to control subjects. However, there is no association of the MMP-9 −1562 C→T promoter polymorphism in the development of gastric cancer.
Similar content being viewed by others
References
Lauren CCR, Evely CFS, da Diana Mendes F, Mariana FL, Adriana CG, Danielle QC. Interrelationship between MYC gene numerical aberrations and protein expression in individuals from northern Brazil with early gastric adenocarcinoma. Cancer Genet Cytogenet. 2008;181(1):31–5.
Schwartz G. Invasion and metastasis in gastric cancer: in vitro and in vivo models with clinical considerations. Semin Oncol. 1996;23:316–24.
Neugut AI, Hayek M, Howe G. Epidemiology of gastric cancer. Semin Oncol. 1996;23:281–91.
Sun HK, Tae EJ, Ji UK, Kye CK, Jong WP, Seung MN. Prognostic implications for gastric carcinoma based on loss of heterozygosity genotypes correlation with clinicopathologic variables. Cancer Genet Cytogenet. 2004;153:26–31.
Folgueras AR, Pendas AM, Sanchez LM, Lopez-Otin C. Matrix metalloproteinases in cancer: from new functions to improved inhibition strategies. Int J Dev Biol. 2004;48:411–24.
Hibbs MS, Hoidal JR, Kang AHJ. Expression of a metalloproteinase that degrades native type V collagen and denatured collagens by cultured human alveolar macrophages. Clin Invest. 1987;80:1644–50.
Wilhelm SM, Collier IE, Marmer BL, Eisen AZ, Grant GA, Goldberg GI. SV40-transformed human lung fibroblasts secrete a 92 kDa type IV collagenase which is identical to that secreted by normal human macrophages. J Biol Chem. 1989;264:17213–21.
Murphy G, Ward R, Hembry RM, Reynolds JJ, Kuhn K, Tryggvason K. Characterization of gelatinase from pig polymorphonuclear leucocytes. A metalloproteinase resembling tumour type IV collagenase. Biochem J. 1989;258:463–72.
Senior RM, Griffin GL, Fliszar CJ, Shapiro SD, Goldberg GI, Welgus HG. Human 92- and 72-kilodalton type IV collagenases are elastases. J Biol Chem. 1991;266:7870–5.
Welgus HG, Campbell EJ, Cury JD, Eisen AZ, Senior RM, Wilhelm SM, et al. Neutral metalloproteinases produced by human mononuclear phagocytes. Enzyme profile, regulation, and expression during cellular development. J Clin Invest. 1990;86:1496–502.
Campbell EJ, Cury JD, Shapiro SD, Goldberg GI, Welgus HG. Neutral proteinases of human mononuclear phagocytes: cellular differentiation markedly alters cell phenotype for serine proteinases, metalloproteinases, and TIMP. J Immunol. 1991;146:1286–93.
Watanabe H, Nakanishi I, Yamashita K, Hayakawa T. OkadaY. Matrix metalloproteinase-9 (92 kDa gelatinase/type IV collagenase) from U937 monoblastoid cells: correlation with cellular invasion. J Cell Sci. 1993;104:991–9.
O-charoenrat P, Rhys-Evans PH, Eccles SA. Expression of matrix metalloproteinases and their inhibitors correlates with invasion and metastasis in squamous cell carcinoma of the head and neck. Arch Otolaryngol Head Neck Surg. 2001;127:813–20.
Folgueras AR, Pendas AM, Sanchez LM, Lopez-Otin C. Matrix metalloproteinases in cancer: from new functions to improved inhibition strategies. Int J Dev Biol. 2004;48:411–24.
Vaira D, Holton J, Cains S, Polydorou A, Falzon M, Dowsett J, et al. Urease test for Campylobacter pylori: care in interpretation. J Clin Pathol. 1988;41:812–3.
Lahiri DK, Nurnberger JI Jr. A rapid non-enzymatic method for the preparation of HMW DNA from blood for RFLP studies. Nucleic Acids Res. 1992;19(19):5444.
Brenner H, Rothenbacher D, Arndt V. Epidemiology of stomach cancer. Methods Mol Biol. 2009;472:467–77.
Baiping Z, Shu Y, de Moniek M, Alun E, et al. Functional polymorphism in the regulatory region of gelatinase B gene in relation to severity of coronary atherosclerosis. Circulation. 1999;99:1788–94.
Sugimoto M, Yoshida S, Kennedy S, Deguchi M, Ohara N, Maruo T. Matrix metalloproteinase-1 and -9 promoter polymorphisms and endometrial carcinoma risk in a Japanese population. J Soc Gynecol Invest. 2006;13(7):523–9.
Matsumura S, Oue N, Nakayama H, Kitadai Y, Yoshida K, Yamaguchi Y, et al. A single nucleotide polymorphism in the MMP-9 promoter affects tumor progression and invasive phenotype of gastric cancer. J Cancer Res Clin. 2005;131(1):19–25.
Parkin DM, Bray F, Ferlay J, Pisani P. Global Cancer Statistics, 2002. CA Cancer J Clin. 2005;55:74–108.
Sung NY, Choi KS, Park ES, Park K, Lee SY, Lee AK, et al. Smoking, alcohol and gastric cancer risk in Korean men: the National Health Insurance Corporation Study. Br J Cancer. 2007;97:700–4.
Matsuhashi T, Yamada N, Shinzawa H, Takahashi T. Effect of alcohol on tumor growth of hepatocellular carcinoma with type C cirrhosis. Intern Med. 1996;35(6):443–8.
Tan W, Bailey AP, Shparago M, Busby B, Covington J, Johnson JW, et al. Chronic alcohol consumption stimulates VEGF expression, tumor angiogenesis and progression of melanoma in mice. Cancer Biol Ther. 2007;6(8):1211–7.
Zhao W, Liu H, Xu S, Entschladen F, Niggemann B, Zanker KS, et al. Migration and metalloproteinases determine the invasive potential of mouse melanoma cells, but not melanin and telomerase. Cancer Lett. 2001;162(Suppl):S49–55.
Bodey B, Bodey B Jr, Siegel SE, Kaiser HE. Matrix metalloproteinase expression in malignant melanomas: tumor-extracellular matrix interactions in invasion and metastasis. In Vivo. 2001;15(1):57–64.
Hofmann UB, Eggert AA, Blass K, Brocker EB, Becker JC. Expression of matrix metalloproteinases in the micro environment of spontaneous and experimental melanoma metastases reflects the requirements for tumor formation. Cancer Res. 2003;63(23):8221–5.
Matsumura S, Oue N, Nakayama H, et al. A single nucleotide polymorphism in the MMP-9 promoter affects tumor progression and invasive phenotype of gastric cancer. J Cancer Res Clin. 2005;131(1):19–25.
Kubben FJGM, Sier CFM, Meijer MJW, van den Berg M, van der Reijden JJ, Griffioen G, et al. Clinical impact of MMP and TIMP gene polymorphisms in gastric cancer. Br J Cancer. 2006;95:744–51.
Zhang X, Miao X, Xiong P, et al. Matrix metalloproteinase (MMP-2 and -9 functional single nucleotide polymorphism and gastric cancer. Cancer. 2004;23(11):1233–7.
Acknowledgments
Financial support, from Department of Biotechnology (DBT), New Delhi is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krishnaveni, D., Bhayal, A.C., Sri Manjari, K. et al. MMP 9 Gene Promoter Polymorphism in Gastric Cancer. Ind J Clin Biochem 27, 259–264 (2012). https://doi.org/10.1007/s12291-012-0210-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12291-012-0210-2