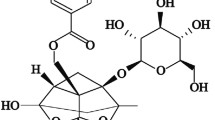
Abstract
Ganoderic Acid A (GA) has many pharmacological effects such as anti-tumor, antibacterial, anti-inflammatory, and immunosuppressive effects. However, the protective effect of GA on liver injury has not been reported. This study aimed to investigate the action of GA on insufficient methionine and choline combined with high-fat diet (HFD)-induced non-alcoholic fatty liver disease (NAFLD) in rats. NAFLD model was established by insufficient methionine and choline combined with high fat feeding to rats. The levels of Acetyl-CoA carboxylase, fatty acid synthase, sterol regulatory element binding protein, liver X receptors, AMP-activated protein kinase, peroxisome proliferator-activated receptor α, PPARg coactivator 1α and NF-κB pathway in the liver were detected by western blot. The results of this study demonstrated that the expression of GA can not only significantly decrease the live weight and liver weight per body weight of HFD mice, but also restore the alanine aminotransferase, aspartate aminotransferase, total bilirubin levels, triglyceride and cholesterol in serum. In addition, the expression of GA increased the levels of high-density lipoprotein cholesterol in serum, ameliorated pathological changes and decreased NAS score of mice’s liver. In conclusion, the treatment with GA could improve NAFLD in rats by regulating the levels of signaling events involved in free fatty acid production, lipid oxidation and liver inflammation.








Similar content being viewed by others
References
Arkan MC, Hevener AL, Greten FR, Maeda S, Li ZW, Long JM, Wynshaw Boris A, Poli G, Olefsky J, Karin M (2005) IKK-beta links inflammation to obesity-induced insulin resistance. Nat Med 11(2):191–198. https://doi.org/10.1038/nm1185
Argyrou C, Moris D, Vernadakis S (2017) Hepatocellular carcinoma development in non-alcoholic fatty liver disease and non-alcoholic steatohepatitis. Is it going to be the “Plague” of the 21st century? A literature review focusing on pathogenesis, prevention and treatment. J BUON 22:6–20. https://doi.org/10.1002/9781118924938.ch1
Bagherniya M, Nobili V, Blesso CN, Sahebkar A (2018) Medicinal plants and bioactive natural compounds in the treatment of non-alcoholic fatty liver disease: a clinical review. Pharmacol Res 130:213–240. https://doi.org/10.1016/j.phrs.2017.12.020
Barish GD, Narkar VA, Evans RM (2006) PPAR delta: a dagger in the heart of the metabolic syndrome. J Clin Invest 116:590–597. https://doi.org/10.1172/JCI27955
Barroso E, Rodríguez-Calvo R, Serrano-Marco L, Astudillo AM, Balsinde J, Palomer X, Vázquez-Carrera M (2011) The PPARβ/δ activator GW501516 prevents the down-regulation of AMPK caused by a high-fat diet in liver and amplifies the PGC-1α-Lipin 1-PPARα pathway leading to increased fatty acid oxidation. Endocrinology 152:1848–1859. https://doi.org/10.1210/en.2010-1468
Berger J, Moller DE (2002) The mechanisms of action of PPARs. Annu Rev Med 53:409–435. https://doi.org/10.1146/annurev.med.53.082901.104018
Boh B, Berovic M, Zhang J, Zhibin L (2007) Ganoderma lucidum and its pharmaceutically active compounds. Biotechnol Annu Rev 13:265–301. https://doi.org/10.1016/S1387-2656(07)13010-6
Chang CJ, Lin C-S, Lu C-C, Martel J, Ko Y-F, Ojcius DM, Tseng S-F, Wu T-R, Chen Y-YM, Young JD (2015) Ganoderma lucidum reduces obesity in mice by modulating the composition of the gut microbiota. Nat Commun 6:7489. https://doi.org/10.1038/ncomms8489
Daemen S, Kutmon M, Evelo CT (2013) A pathway approach to investigate the function and regulation of SREBPs. Genes Nutr 8:289–300. https://doi.org/10.1007/s12263-013-0342-x
Dahlhoff C, Worsch S, Sailer M, Hummel BA, Fiamoncini J, Uebel K, Obeid R, Scherling C, Geisel J, Bader BL (2014) Methyl-donor supplementation in obese mice prevents the progression of NAFLD, activates AMPK and decreases acyl-carnitine levels. Mol Metab 3:565–580. https://doi.org/10.1016/j.molmet.2014.04.010
Guo F, Huang C, Liao X, Wang Y, He Y, Feng R, Li Y, Sun C (2011) Beneficial effects of mangiferin on hyperlipidemia in high-fat-fed hamsters. Mol Nutr Food Res 55:1809–1818. https://doi.org/10.1002/mnfr.201100392
Min HK, Kapoor A, Fuchs M, Mirshahi F, Zhou H, Maher J, Kellum J, Warnick R, Contos MJ, Sanyal AJ (2012) Increased hepatic synthesis and dysregulation of cholesterol metabolism is associated with the severity of nonalcoholic fatty liver disease. Cell Metab 15:665–674. https://doi.org/10.1016/j.cmet.2012.04.004
Horton JD, Goldstein JL, Brown MS (2002) SREBPs: activators of the complete program of cholesterol and fatty acid synthesis in the liver. J Clin Invest 109(9):1125–1131. https://doi.org/10.1172/JCI15593
Hou Y, Moreau F, Chadee K (2012) PPARgamma is an E3 ligase that induces the degradation of NFkappaB/p65. Nat Commun 3:1300. https://doi.org/10.1038/ncomms2270
Hsu WH, Chen TH, Lee BH, Hsu YW, Pan TM (2014) Monascin and ankaflavin act as natural AMPK activators with PPARα agonist activity to down-regulate nonalcoholic steatohepatitis in high-fat diet-fed C57BL/6 mice. Food Chem Toxicol 64:94–103. https://doi.org/10.1016/j.fct.2013.11.015
Huang CZ, Tung YT, Hsia SM, Wu CH, Yen GC (2017) The hepatoprotective effect of Phyllanthus emblica L. fruit on high fat diet-induced non-alcoholic fatty liver disease (NAFLD) in SD rats. Food Funct 8:842–850. https://doi.org/10.1039/c6fo01585a
Jung TW, Kang C, Goh J, Chae SI, Kim HC, Lee TJ, Abd El-Aty AM, Jeong JH (2018) WISP1 promotes non-alcoholic fatty liver disease and skeletal muscle insulin resistance via TLR4/JNK signaling. J Cell Physiol 233(8):6077–6087. https://doi.org/10.1002/jcp.26449
Kim J, Wei Y, Sowers JR (2011) Role of mitochondrial dysfunction in insulin resistance. Circ Res 102(4):401–414. https://doi.org/10.1161/CIRCRESAHA.107.165472
Kohjima M, Enjoji M, Higuchi N, Kato M, Kotoh K, Yoshimoto T, Fujino T, Yada M, Yada R, Harada N, Takayanagi R, Nakamuta M (2007) Re-evaluation of fatty acid metabolism-related gene expression in nonalcoholic fatty liver disease. Int J Mol Med 20(3):351–358. https://doi.org/10.3892/ijmm.20.3.351
Kohjima M, Higuchi N, Kato M, Kotoh K, Yoshimoto T, Fujino T, Yada M, Yada R, Harada N, Enjoji M, Takayanagi R, Nakamuta M (2008) SREBP-1c, regulated by the insulin and AMPK signaling pathways, plays a role in nonalcoholic fatty liver disease. Int J Mol Med 21(4):507–511. https://doi.org/10.3892/ijmm.21.4.507
Lee JH, Moon JM, Kim YH, Lee B, Choi SY, Song BJ, Kim DK, Lee YM (2019) Chrysanthemum indicum effect of enzymatic treatment of linné extracts on lipid accumulation and adipogenesis in high-fat-diet-induced obese male mice. Nutrients. https://doi.org/10.3390/nu11020269
Limacabello E, Garcíamediavilla MV, Miquilenacolina ME, Vargascastrillón J, Lozanorodríguez T, Fernándezbermejo M, Olcoz JL, Gonzálezgallego J, Garcíamonzón C, Sánchezcampos S (2011) Enhanced expression of pro-inflammatory mediators and liver X-receptor-regulated lipogenic genes in non-alcoholic fatty liver disease and hepatitis C. Clin Sci 120(6):239–250. https://doi.org/10.1042/CS20100387
Masarone M, Federico A, Abenavoli L, Loguercio C, Persico M (2014) Non alcoholic fatty liver: epidemiology and natural history. Rev Recent Clin Trials 9(3):126–133. https://doi.org/10.2174/1574887109666141216111143
Miura K, Seki E, Ohnishi H, Brenner DA (2010) Role of toll-like receptors and their downstream molecules in the development of nonalcoholic fatty liver disease. Gastroenterol Res Pract 2010:70–80. https://doi.org/10.1155/2010/362847
Morgan K, Uyuni A, Nandgiri G, Mao L, Castaneda L, Kathirvel E, French SW, Morgan TR (2008) Altered expression of transcription factors and genes regulating lipogenesis in liver and adipose tissue of mice with high fat diet-induced obesity and nonalcoholic fatty liver disease. Eur Eur J Gastroenterol Hepatol 20(9):843–854. https://doi.org/10.1097/MEG.0b013e3282f9b203
Repa JJ, Liang G, Ou J, Bashmakov Y, Lobaccaro JM, Shimomura I, Shan B, Brown MS, Goldstein JL, Mangelsdorf DJ (2000) Regulation of mouse sterol regulatory element-binding protein-1c gene (SREBP-1c) by oxysterol receptors, LXRalpha LXRbeta. Genes Dev 14(22):2819–2830. https://doi.org/10.1101/gad.844900
Safaei A, Oskouie AA, Mohebbi SR, Rezaei-Tavirani M, Mahboubi M, Peyvandi M, Okhovatian F, Zamanian-Azodi M (2016) Metabolomic analysis of human cirrhosis, hepatocellular carcinoma, non-alcoholic fatty liver disease and non-alcoholic steatohepatitis diseases. Gastroenterol Hepatol Bed Bench 9(3):158–173. https://doi.org/10.22037/ghfbb.v0i0.952
Schmitz ML, Mattioli I, Buss H, Kracht M (2014) NF-kappaB: a multifaceted transcription factor regulated at several levels. ChemBioChem 5:1348–1358. https://doi.org/10.1002/cbic.200400144
Wan B, Li Y, Sun S, Yang Y, Lv Y, Wang L, Song M, Chen M, Wu C, Pan H, Zhang X (2019) Ganoderic acid A attenuates lipopolysaccharide-induced lung injury in mice. Biosci Rep 39(5):1. https://doi.org/10.1042/BSR20190301
Wang XC, Rui-Jiao X, Yi L, Dong-Mei W, Yi-Jian Y, Olivier L (2012) The species identity of the widely cultivated Ganoderma, G. lucidum (Ling-zhi). China. PLoS ONE 7(7):e40857. https://doi.org/10.1371/journal.pone.0040857
Yeung F, Hoberg JE, Ramsey CS, Keller MD, Jones DR, Frye RA, Mayo MW (2004) Modulation of NF-kappaB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J 23:2369–2380. https://doi.org/10.1038/sj.emboj.7600244
Yoon S, Kim J, Lee H, Lee H, Lim J, Yang H, Shin SS, Yoon M (2017) The effects of herbal composition Gambigyeongsinhwan (4) on hepatic steatosis and inflammation in Otsuka Long-Evans Tokushima fatty rats and HepG2 cells. J Ethnopharmacol 195:204–213. https://doi.org/10.1016/j.jep.2016.11.020
Yu J, Chu ES, Hui AY, Cheung KF, Chan HL, Leung WK, Farrell GC, Sung JJ (2007) Lipoprotein lipase activator ameliorates the severity of dietary steatohepatitis. Biochem Biophys Res Commun 356(1):53–59. https://doi.org/10.1016/j.bbrc.2007.02.129
Zhu J, Jin J, Ding J, Ding JX, Li SY, Cen PP, Wang KY, Wang H, Xia JB (2018) Ganoderic acid A improves high fat diet-induced obesity, lipid accumulation and insulin sensitivity through regulating SREBP pathway. Chem Biol Interact 290:77–87. https://doi.org/10.1016/j.cbi.2018.05.014
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict interest associated with this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, F., Shi, K., Dong, J. et al. Ganoderic acid A attenuates high-fat-diet-induced liver injury in rats by regulating the lipid oxidation and liver inflammation. Arch. Pharm. Res. 43, 744–754 (2020). https://doi.org/10.1007/s12272-020-01256-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-020-01256-9