Abstract
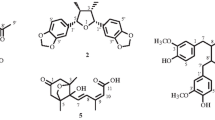
Column chromatographic separation of the roots of cultivated-wild ginseng (Jangnoisam) led to the isolation of seven polyacetylenes (1–7). Their structures were determined by spectroscopic methods to be panaxynol (1), ginsenoyne-A (2), panaxydol (3), 10-methoxy heptadeca-1-ene-4, 6-dyne-3, 9-diol (4) (3R, 9R, 10R)-panaxytriol (5), panaxyne (6), and ginsenoyne-C (7). These compounds were isolated from this source for the first time. The compounds were tested for their cytotoxic activity against four human cancer cell lines (A549, SK-OV-3, SK-MEL-2, and HCT-15) in vitro using the SRB method. Panaxydol (3) and panaxyne (6) showed significant and selective cytotoxicity against SK-OV-3 with ED50 values 2.93 and 1.40 μM, respectively.
Similar content being viewed by others
References
Ahn, B. Z. and Kim, S. I., Relation between structure and cytotoxic activity of panaxydol analogs against L1210 cells. Arch. Pharm. (Weinheim, Germany), 321, 61–63 (1988).
Ahn, G. I., Park, C, H., Lee, E. O., Lee, H. J., Lee, J. H., Kim, K. H., Rhee, Y. H., Jang, Y. S., Kim, S. T., and Kim, S. H., Apoptotic effect of ethanol extracts of ojungbangamtang and acidic polysaccharide of Korea red ginseng in a MCF7/adR multidrug-resistance breast cancer cells. Yakhak Hoechi, 50, 272–277 (2006).
Baek, N. I., Kim, D. S., Lee, Y. H., Park, J. D., Lee, Ch. B., and Kim I. S., Cytotoxicities of ginseng saponins and their degradation products against some cancer cell lines., Arch. Pharm. Res., 8, 164–168 (1995).
Choi, Y. E., Kim, Y. S., Yi, M. J., Park, W. G., Yi, J. S., Chun, S. R., Han, S. S., and Lee, S. J., Physiological and chemical characteristics of field-and mountain-cultivated ginseng roots. J. Plant. Biol., 50, 198–205 (2007).
Fujimoto, Y., Satoh, M., Takeuchi, N., and Kirisawa, M., Cytotoxic acetylene from Panax quinquefolium. Chem. Pharm. Bull., 39, 521–523 (1991).
Fujimoto, Y., Wang, H., Satoh, M., and Takeuchi, N., Polyacetylenes from Panax quinquefolium. Phytochemistry, 35, 1255–1257 (1994).
Hirakura, K., Morita, M., Nakajima, K., Ikeya, Y., and Mitsuhashi, H., Polyacetylenes from the roots of Panax ginseng. Phytochemistry, 30, 3327–3333 (1991).
Hirakura, K., Morita, M., Nakajima, K., Ikeya, Y., and Mitsuhashi, H., The constituents of Panax ginseng. Part 3.Three acetylenic compounds from roots of Panax ginseng. Phytochemistry, 31, 899–903 (1992).
Hirakura, K., Morita, M., Niitsu, K., Ikeya, Y., and Maruno, M., The constituents of Panax ginseng. Part 4. Linoleoylated polyacetylenes from the root of Panax ginseng. Phytochemistry, 35, 963–967 (1994).
Kang, K. S., Yokozawa, T., Yamabe, N., Kim, H. Y., and Park, J. H., ESR study on the structure and hydroxyl radical-scavenging activity relationships of ginsenosides isolated from Panax ginseng C. A. Meyer. Bio. Pharm. Bull., 30, 917–921 (2007).
Kim, S. I., Kang, K. S., Kim, H. Y., and Ahn, B. Z., Panaxyne, a new cytotoxic polyyne from Panax ginseng root against L1210 cell. Saengyak Hakhoechi, 20, 71–75 (1989).
Kim, J. H. and Kim, J. K., Antioxidant activity and functional component analysis of Korean mountain Ginseng’s different sections. Han’guk Sikp’um Yongyang Kwahak Hoechi, 35, 1315–1321 (2006).
Kitagawa, I., Taniyama, T., Yoshikawa, M., Ikennishi, Y., and Nakagawa, Y., Chemical studies on crude drug processing. VI. Chemical structures of malonyl-ginsenosides Rb1, Rb2, Rc, and Rd isolated from the root of Panax ginseng C. A. Meyer. Chem.Pharm.Bull., 37, 2961–2970 (1989).
Lee, S. W., Kim, K., Rho, M. Ch., Chung, M. Y., Kim, Y. H., Lee, S. K., Lee, H. S., and Kim, Y. K., New polyacetylenes, DGAT inhibitors from the roots of Panax ginseng. Planta Medica, 70, 197–200 (2004).
Lee, W. M., Kim, S. D., Kim, K. S., Song, Y. B., Kwak, Y. S., Cho, J. Y., Park, H. J., Oh, J. W., and Rhee, M. H., Proto-panaxadiol modulates LPS-induced inflammatory activity in murine macrophage RAW264.7 cells. J. Ginseng Res., 30, 181–187 (2006).
Mayer, S. F., Steinreiber, A., Orru, R. V. A., and Faber, K., Chemoenzymatic Asymmetric Total Syntheses of Antitumor Agents (3R,9R,10R)-and (3S,9R,10R)-Panaxytriol and (R)-and (S)-Falcarinol from Panax ginseng Using an Enantioconvergent Enzyme-Triggered Cascade Reaction. J. Org. Chem., 67, 9115–9121 (2002).
Matsunaga, H., Katano, M., Yamamoto, H., Fujito, H., and Takata, K., Cytotoxic activity of poly-acetylene compounds in Panax ginseng C. A. Meyer. Chem. Pharm. Bull., 38, 3480–3482 (1990).
Park, J. D., Recent studies on the chemical constituents of Korean ginseng (Panax ginseng C. A. Meyer). J. Ginseng Res., 20, 389–415 (1996).
Ryu, J. H., Jang, S. R., Lee, S. Y., Lee, H. J., and Han, Y. N., Inhibitors of nitric oxide synthesis from ginseng in activated macrophages. J. Ginseng Res., 22, 181–187 (1998).
Skehan, P., Storeng, R., Scudiero, D., Monks, A., McMahon, J., Vistica, D., Warren, J.T., Bokesch, H., Kenney, S., and Boyd, M. R., New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst., 82, 1107–1112 (1990).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, M.C., Seo, D.S., Choi, S.U. et al. Polyacetylenes from the roots of cultivated-wild ginseng and their cytotoxicity in vitro . Arch. Pharm. Res. 31, 154–159 (2008). https://doi.org/10.1007/s12272-001-1134-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-001-1134-1