Abstract
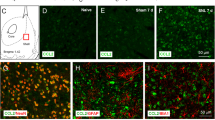
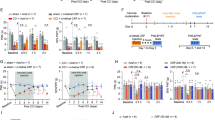
The medium spiny neurons (MSNs) in the nucleus accumbens (NAc) integrate excitatory and inhibitory synaptic inputs and gate motivational and emotional behavior output. Here we report that the relative intensity of excitatory and inhibitory synaptic inputs to MSNs of the NAc shell was decreased in mice with neuropathic pain induced by spinal nerve ligation (SNL). SNL increased the frequency, but not the amplitude of spontaneous inhibitory postsynaptic currents (sIPSCs), and decreased both the frequency and amplitude of spontaneous excitatory postsynaptic currents (sEPSCs) in the MSNs. SNL also decreased the paired-pulse ratio (PPR) of evoked IPSCs but increased the PPR of evoked EPSCs. Moreover, acute bath application of C–C motif chemokine ligand 2 (CCL2) increased the frequency and amplitude of sIPSCs and sEPSCs in the MSNs, and especially strengthened the amplitude of N-methyl-D-aspartate receptor (NMDAR)-mediated miniature EPSCs. Further Ccl2 overexpression in the NAc in vivo decreased the peak amplitude of the sEPSC/sIPSC ratio. Finally, Ccr2 knock-down improved the impaired induction of NMDAR-dependent long-term depression (LTD) in the NAc after SNL. These results suggest that CCL2/CCR2 signaling plays a role in the integration of excitatory/inhibitory synaptic transmission and leads to an increase of the LTD induction threshold at the synapses of MSNs during neuropathic pain.







Similar content being viewed by others
References
Schwartz N, Temkin P, Jurado S, Lim BK, Heifets BD, Polepalli JS, et al. Chronic pain. Decreased motivation during chronic pain requires long-term depression in the nucleus accumbens. Science 2014, 345: 535–542.
Ren W, Centeno MV, Berger S, Wu Y, Na X, Liu X. The indirect pathway of the nucleus accumbens shell amplifies neuropathic pain. Nat Neurosci 2016, 19: 220–222.
Wu XB, Jing PB, Zhang ZJ, Cao DL, Gao MH, Jiang BC, et al. Chemokine receptor CCR2 contributes to neuropathic pain and the associated depression via increasing NR2B-mediated currents in both D1 and D2 dopamine receptor-containing medium spiny neurons in the nucleus accumbens shell. Neuropsychopharmacology 2018, 43: 2320–2330.
Chang PC, Pollema-Mays SL, Centeno MV, Procissi D, Contini M, Baria AT, et al. Role of nucleus accumbens in neuropathic pain: linked multi-scale evidence in the rat transitioning to neuropathic pain. Pain 2014, 155: 1128–1139.
Goffer Y, Xu D, Eberle SE, D’Amour J, Lee M, Tukey D, et al. Calcium-permeable AMPA receptors in the nucleus accumbens regulate depression-like behaviors in the chronic neuropathic pain state. J Neurosci 2013, 33: 19034–19044.
Xu D, Su C, Lin HY, Manders T, Wang J. Persistent neuropathic pain increases synaptic GluA1 subunit levels in core and shell subregions of the nucleus accumbens. Neurosci Lett 2015, 609: 176–181.
Su C, D’Amour J, Lee M, Lin HY, Manders T, Xu D, et al. Persistent pain alters AMPA receptor subunit levels in the nucleus accumbens. Mol Brain 2015, 8: 46.
Nisenbaum ES, Grace AA, Berger TW. Functionally distinct subpopulations of striatal neurons are differentially regulated by GABAergic and dopaminergic inputs–II. vitro analysis. Neuroscience 1992, 48: 579–593.
Russo SJ, Mazei-Robison MS, Ables JL, Nestler EJ. Neurotrophic factors and structural plasticity in addiction. Neuropharmacology 2009, 56(Suppl 1): 73–82.
Wilson CJ, Kawaguchi Y. The origins of two-state spontaneous membrane potential fluctuations of neostriatal spiny neurons. J Neurosci 1996, 16: 2397–2410.
O’Donnell P, Grace AA. Synaptic interactions among excitatory afferents to nucleus accumbens neurons: hippocampal gating of prefrontal cortical input. J Neurosci 1995, 15: 3622–3639.
Castillo PE, Chiu CQ, Carroll RC. Long-term plasticity at inhibitory synapses. Curr Opin Neurobiol 2011, 21: 328–338.
Potter LE, Paylor JW, Suh JS, Tenorio G, Caliaperumal J, Colbourne F, et al. Altered excitatory-inhibitory balance within somatosensory cortex is associated with enhanced plasticity and pain sensitivity in a mouse model of multiple sclerosis. J Neuroinflammation 2016, 13: 142.
Cheriyan J, Sheets PL. Peripheral nerve injury reduces the excitation-inhibition balance of basolateral amygdala inputs to prelimbic pyramidal neurons projecting to the periaqueductal gray. Mol Brain 2020, 13: 100.
Massaly N, Copits BA, Wilson-Poe AR, Hipolito L, Markovic T, Yoon HJ, et al. Pain-induced negative affect is mediated via recruitment of the nucleus accumbens kappa opioid system. Neuron 2019, 102(564–573): e566.
Gao YJ, Zhang L, Samad OA, Suter MR, Yasuhiko K, Xu ZZ, et al. JNK-induced MCP-1 production in spinal cord astrocytes contributes to central sensitization and neuropathic pain. J Neurosci 2009, 29: 4096–4108.
Zhou Y, Tang H, Xiong H. Chemokine CCL2 enhances NMDA receptor-mediated excitatory postsynaptic current in rat hippocampal slices-a potential mechanism for HIV-1-associated neuropathy?. J Neuroimmune Pharmacol 2016, 11: 306–315.
Luscher C, Malenka RC. NMDA receptor-dependent long-term potentiation and long-term depression (LTP/LTD). Cold Spring Harb Perspect Biol 2012, 4.
Spiga S, Talani G, Mulas G, Licheri V, Fois GR, Muggironi G, et al. Hampered long-term depression and thin spine loss in the nucleus accumbens of ethanol-dependent rats. Proc Natl Acad Sci U S A 2014, 111: E3745-3754.
Chiou CS, Huang CC, Liang YC, Tsai YC, Hsu KS. Impairment of long-term depression in the anterior cingulate cortex of mice with bone cancer pain. Pain 2012, 153: 2097–2108.
Wei F, Li P, Zhuo M. Loss of synaptic depression in mammalian anterior cingulate cortex after amputation. J Neurosci 1999, 19: 9346–9354.
Zhang ZJ, Cao DL, Zhang X, Ji RR, Gao YJ. Chemokine contribution to neuropathic pain: respective induction of CXCL1 and CXCR2 in spinal cord astrocytes and neurons. Pain 2013, 154: 2185–2197.
Thomas MJ, Beurrier C, Bonci A, Malenka RC. Long-term depression in the nucleus accumbens: a neural correlate of behavioral sensitization to cocaine. Nat Neurosci 2001, 4: 1217–1223.
Baliki MN, Petre B, Torbey S, Herrmann KM, Huang L, Schnitzer TJ, et al. Corticostriatal functional connectivity predicts transition to chronic back pain. Nat Neurosci 2012, 15: 1117–1119.
Makary MM, Polosecki P, Cecchi GA, DeAraujo IE, Barron DS, Constable TR, et al. Loss of nucleus accumbens low-frequency fluctuations is a signature of chronic pain. Proc Natl Acad Sci U S A 2020, 117: 10015–10023.
Britt JP, Benaliouad F, McDevitt RA, Stuber GD, Wise RA, Bonci A. Synaptic and behavioral profile of multiple glutamatergic inputs to the nucleus accumbens. Neuron 2012, 76: 790–803.
Lee AT, Vogt D, Rubenstein JL, Sohal VS. A class of GABAergic neurons in the prefrontal cortex sends long-range projections to the nucleus accumbens and elicits acute avoidance behavior. J Neurosci 2014, 34: 11519–11525.
Taverna S, van Dongen YC, Groenewegen HJ, Pennartz CM. Direct physiological evidence for synaptic connectivity between medium-sized spiny neurons in rat nucleus accumbens in situ. J Neurophysiol 2004, 91: 1111–1121.
Burke DA, Rotstein HG, Alvarez VA. Striatal local circuitry: a new framework for lateral inhibition. Neuron 2017, 96: 267–284.
Duguid IC, Smart TG. Retrograde activation of presynaptic NMDA receptors enhances GABA release at cerebellar interneuron-Purkinje cell synapses. Nat Neurosci 2004, 7: 525–533.
Gubellini P, Ben-Ari Y, Gaiarsa JL. Endogenous neurotrophins are required for the induction of GABAergic long-term potentiation in the neonatal rat hippocampus. J Neurosci 2005, 25: 5796–5802.
Sivakumaran S, Mohajerani MH, Cherubini E. At immature mossy-fiber-CA3 synapses, correlated presynaptic and postsynaptic activity persistently enhances GABA release and network excitability via BDNF and cAMP-dependent PKA. J Neurosci 2009, 29: 2637–2647.
Liu D, Tang QQ, Yin C, Song Y, Liu Y, Yang JX, et al. Brain-derived neurotrophic factor-mediated projection-specific regulation of depressive-like and nociceptive behaviors in the mesolimbic reward circuitry. Pain 2018, 159: 175.
Zhang H, Qian YL, Li C, Liu D, Wang L, Wang XY, et al. Brain-derived neurotrophic factor in the mesolimbic reward circuitry mediates nociception in chronic neuropathic pain. Biol Psychiatry 2017, 82: 608–618.
Liu Y, Zhang LI, Tao HW. Heterosynaptic scaling of developing GABAergic synapses: dependence on glutamatergic input and developmental stage. J Neurosci 2007, 27: 5301–5312.
Chiu CQ, Martenson JS, Yamazaki M, Natsume R, Sakimura K, Tomita S, et al. Input-specific nmdar-dependent potentiation of dendritic gabaergic inhibition. Neuron 2018, 97(368–377): e363.
Spicarova D, Adamek P, Kalynovska N, Mrozkova P, Palecek J. TRPV1 receptor inhibition decreases CCL2-induced hyperalgesia. Neuropharmacology 2014, 81: 75–84.
D’Amour JA, Froemke RC. Inhibitory and excitatory spike-timing-dependent plasticity in the auditory cortex. Neuron 2015, 86: 514–528.
Luo C, Kuner T, Kuner R. Synaptic plasticity in pathological pain. Trends Neurosci 2014, 37: 343–355.
Deng PY, Lei S. Long-term depression in identified stellate neurons of juvenile rat entorhinal cortex. J Neurophysiol 2007, 97: 727–737.
Thomas MJ, Malenka RC, Bonci A. Modulation of long-term depression by dopamine in the mesolimbic system. J Neurosci 2000, 20: 5581–5586.
Liu L, Wong TP, Pozza MF, Lingenhoehl K, Wang Y, Sheng M, et al. Role of NMDA receptor subtypes in governing the direction of hippocampal synaptic plasticity. Science 2004, 304: 1021–1024.
Bie B, Brown DL, Naguib M. Synaptic plasticity and pain aversion. Eur J Pharmacol 2011, 667: 26–31.
Bliss TV, Collingridge GL, Kaang BK, Zhuo M. Synaptic plasticity in the anterior cingulate cortex in acute and chronic pain. Nat Rev Neurosci 2016, 17: 485–496.
Acknowledgements
This work was supported by Grants from the National Natural Science Foundation of China (32030048, 31871064, and 31671091).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wu, XB., Zhu, Q. & Gao, YJ. CCL2/CCR2 Contributes to the Altered Excitatory-inhibitory Synaptic Balance in the Nucleus Accumbens Shell Following Peripheral Nerve Injury-induced Neuropathic Pain. Neurosci. Bull. 37, 921–933 (2021). https://doi.org/10.1007/s12264-021-00697-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-021-00697-6