Abstract
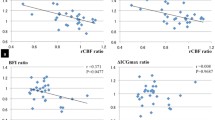
Paradoxical reduction of cerebral blood flow (CBF) after administration of the vasodilator acetazolamide is the most severe stage of cerebrovascular reactivity failure and is often associated with an increased oxygen extraction fraction (OEF). In this study, we aimed to reveal the mechanism underlying this phenomenon by focusing on the ratio of CBF to cerebral blood volume (CBV) as a marker of regional cerebral perfusion pressure (CPP). In 37 patients with unilateral internal carotid or middle cerebral arterial (MCA) steno-occlusive disease and 8 normal controls, the baseline CBF (CBFb), CBV, OEF, cerebral oxygen metabolic rate (CMRO2), and CBF after acetazolamide loading in the anterior and posterior MCA territories were measured by 15O positron emission tomography. Paradoxical CBF reduction was found in 28 of 74 regions (18 of 37 patients) in the ipsilateral hemisphere. High CBFb (>47.6 mL/100 mL/min, n = 7) was associated with normal CBFb/CBV, increased CBV, decreased OEF, and normal CMRO2. Low CBFb (<31.8 mL/100 mL/min, n = 9) was associated with decreased CBFb/CBV, increased CBV, increased OEF, and decreased CMRO2. These findings demonstrated that paradoxical CBF reduction is not always associated with reduction of CPP, but partly includes high-CBFb regions with normal CPP, which has not been described in previous studies.
Similar content being viewed by others
References
Symon L. Experimental evidence for “intracerebral steal” following CO2 inhalation. Scand J Clin Lab Invest Suppl 1968, 102: XIII:A.
Symon L, Pasztor E, Branston NM. The distribution and density of reduced cerebral blood fl ow following acute middle cerebral artery occlusion: an experimental study by the technique of hydrogen clearance in baboons. Stroke 1974, 5: 355–364.
Regli F, Yamaguchi T, Waltz AG. Effects of acetazolamide on cerebral ischemia and infarction after experimental occlusion of middle cerebral artery. Stroke 1971, 2: 456–460.
Vorstrup S, Henriksen L, Paulson OB. Effect of acetazolamide on cerebral blood fl ow and cerebral metabolic rate for oxygen. J Clin Invest 1984, 74: 1634–1639.
Vorstrup S, Brun B, Lassen NA. Evaluation of the cerebral vasodilatory capacity by the acetazolamide test before EC-IC bypass surgery in patients with occlusion of the internal carotid artery. Stroke 1986, 17: 1291–1298.
Vorstrup S. Tomographic cerebral blood flow measurements in patients with ischemic cerebrovascular disease and evaluation of the vasodilatory capacity by the acetazolamide test. Acta Neurol Scand Suppl 1988, 114: 1–48.
Kuwabara Y, Ichiya Y, Sasaki M, Yoshida T, Masuda K. Time dependency of the acetazolamide effect on cerebral hemodynamics in patients with chronic occlusive cerebral arteries. Early steal phenomenon demonstrated by [15O]H2O positron emission tomography. Stroke 1995, 26: 1825–1829.
Okazawa H, Yamauchi H, Toyoda H, Sugimoto K, Fujibayashi Y, Yonekura Y. Relationship between vasodilatation and cerebral blood flow increase in impaired hemodynamics: a PET study with the acetazolamide test in cerebrovascular disease. J Nucl Med 2003, 44: 1875–1883.
Baron JC, Bousser MG, Rey A, Guillard A, Comar D, Castaigne P. Reversal of focal “misery-perfusion syndrome” by extra-intracranial arterial bypass in hemodynamic cerebral ischemia. A case study with 15O positron emission tomography. Stroke 1981, 12: 454–459.
Gibbs JM, Wise RJ, Leenders KL, Jones T. Evaluation of cerebral perfusion reserve in patients with carotid-artery occlusion. Lancet 1984, 1: 310–314.
Sette G, Baron JC, Mazoyer B, Levasseur M, Pappata S, Crouzel C. Local brain haemodynamics and oxygen metabolism in cerebrovascular disease. Positron emission tomography. Brain 1989, 112(Pt 4): 931–951.
Schumann P, Touzani O, Young AR, Morello R, Baron JC, MacKenzie ET. Evaluation of the ratio of cerebral blood fl ow to cerebral blood volume as an index of local cerebral perfusion pressure. Brain 1998, 121(Pt 7): 1369–1379.
North American Symptomatic Carotid Endarterectomy Trial Collaborators. Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Engl J Med 1991, 325: 445–453.
Matsumoto K, Kitamura K, Mizuta T, Tanaka K, Yamamoto S, Sakamoto S, et al. Performance characteristics of a new 3-dimensional continuous-emission and spiral-transmission high-sensitivity and high-resolution PET camera evaluated with the NEMA NU 2-2001 standard. J Nucl Med 2006, 47: 83–90.
Ibaraki M, Miura S, Shimosegawa E, Sugawara S, Mizuta T, Ishikawa A, et al. Quantification of cerebral blood flow and oxygen metabolism with 3-dimensional PET and 15O: validation by comparison with 2-dimensional PET. J Nucl Med 2008, 49: 50–59.
Mintun MA, Raichle ME, Martin WR, Herscovitch P. Brain oxygen utilization measured with O-15 radiotracers and positron emission tomography. J Nucl Med 1984, 25: 177–187.
Herscovitch P, Markham J, Raichle ME. Brain blood flow measured with intravenous H2(15)O. I. Theory and error analysis. J Nucl Med 1983, 24: 782–789.
Iida H, Jones T, Miura S. Modeling approach to eliminate the need to separate arterial plasma in oxygen-15 inhalation positron emission tomography. J Nucl Med 1993, 34: 1333–1340.
Hatazawa J, Fujita H, Kanno I, Satoh T, Iida H, Miura S, et al. Regional cerebral blood flow, blood volume, oxygen extraction fraction, and oxygen utilization rate in normal volunteers measured by the autoradiographic technique and the single breath inhalation method. Ann Nucl Med 1995, 9: 15–21.
Lammertsma AA, Jones T. Correction for the presence of intravascular oxygen-15 in the steady-state technique for measuring regional oxygen extraction ratio in the brain: 1. Description of the method. J Cereb Blood Flow Metab 1983, 3: 416–424.
Kanno I, Iida H, Miura S, Murakami M, Takahashi K, Sasaki H, et al. A system for cerebral blood fl ow measurement using an H215O autoradiographic method and positron emission tomography. J Cereb Blood Flow Metab 1987, 7: 143–153.
Iida H, Kanno I, Miura S, Murakami M, Takahashi K, Uemura K. Error analysis of a quantitative cerebral blood flow measurement using H2(15)O autoradiography and positron emission tomography, with respect to the dispersion of the input function. J Cereb Blood Flow Metab 1986, 6: 536–545.
Kuroda S, Shiga T, Ishikawa T, Houkin K, Narita T, Katoh C, et al. Reduced blood flow and preserved vasoreactivity characterize oxygen hypometabolism due to incomplete infarction in occlusive carotid artery diseases. J Nucl Med 2004, 45: 943–949.
Imaizumi M, Kitagawa K, Oku N, Hashikawa K, Takasawa M, Yoshikawa T, et al. Clinical significance of cerebrovascular reserve in acetazolamide challenge -comparison with acetazolamide challenge H2O-PET and Gas-PET. Ann Nucl Med 2004, 18: 369–374.
Isozaki M, Arai Y, Kudo T, Kiyono Y, Kobayashi M, Kubota T, et al. Clinical implication and prognosis of normal baseline cerebral blood fl ow with impaired vascular reserve in patients with major cerebral artery occlusive disease. Ann Nucl Med 2010, 24: 371–377.
Powers WJ. Cerebral hemodynamics in ischemic cerebrovascular disease. Ann Neurol 1991, 29: 231–240.
Lassen NA. The luxury-perfusion syndrome and its possible relation to acute metabolic acidosis localised within the brain. Lancet 1966, 2: 1113–1115.
Ackerman RH, Correia JA, Alpert NM, Baron JC, Gouliamos A, Grotta JC, et al. Positron imaging in ischemic stroke disease using compounds labeled with oxygen 15. Initial results of clinicophysiologic correlations. Arch Neurol 1981, 38: 537–543.
Wise RJ, Bernardi S, Frackowiak RS, Legg NJ, Jones T. Serial observations on the pathophysiology of acute stroke. The transition from ischaemia to infarction as reflected in regional oxygen extraction. Brain 1983, 106(Pt 1): 197–222.
Hakim AM. The cerebral ischemic penumbra. Can J Neurol Sci 1987, 14: 557–559.
Torigai T, Mase M, Ohno T, Katano H, Nisikawa Y, Sakurai K, et al. Usefulness of dual and fully automated measurements of cerebral blood flow during balloon occlusion test of the internal carotid artery. J Stroke Cerebrovasc Dis 2011, 22 (3): 197–204.
Faraci FM, Heistad DD. Regulation of large cerebral arteries and cerebral microvascular pressure. Circ Res 1990, 66: 8–17.
Furst H, Hartl WH, Janssen I. Patterns of cerebrovascular reactivity in patients with unilateral asymptomatic carotid artery stenosis. Stroke 1994, 25: 1193–1200.
Omar NM, Marshall JM. Age-related changes in the sympathetic innervation of cerebral vessels and in carotid vascular responses to norepinephrine in the rat: in vitro and in vivo studies. J Appl Physiol 2010, 109: 314–322.
Derdeyn CP, Videen TO, Yundt KD, Fritsch SM, Carpenter DA, Grubb RL, et al. Variability of cerebral blood volume and oxygen extraction: stages of cerebral haemodynamic impairment revisited. Brain 2002, 125: 595–607.
Kuroda S, Shiga T, Houkin K, Ishikawa T, Katoh C, Tamaki N, et al. Cerebral oxygen metabolism and neuronal integrity in patients with impaired vasoreactivity attributable to occlusive carotid artery disease. Stroke 2006, 37: 393–398.
Yamauchi H, Nishii R, Higashi T, Kagawa S, Fukuyama H. Silent cortical neuronal damage in atherosclerotic disease of the major cerebral arteries. J Cereb Blood Flow Metab 2011, 31: 953–961.
Samson Y, Baron JC, Bousser MG, Rey A, Derlon JM, David P, et al. Effects of extra-intracranial arterial bypass on cerebral blood flow and oxygen metabolism in humans. Stroke 1985, 16: 609–616.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Watabe, T., Shimosegawa, E., Kato, H. et al. Paradoxical reduction of cerebral blood flow after acetazolamide loading: a hemodynamic and metabolic study with 15O PET. Neurosci. Bull. 30, 845–856 (2014). https://doi.org/10.1007/s12264-013-1459-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-013-1459-z