Abstract
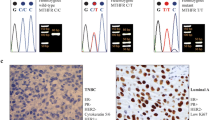
Breast cancer (BC) is the leading cause of cancer related death among women in 2014. The AURKA gene that encodes the protein called Aurora kinase A plays an important role in the progression of the cell cycle, by controlling and promoting the entry into the phase of mitosis. The single nucleotide polymorphism AURKA T91A (rs2273535) (Phe21Ile) has been identified as functional alternator of this kinase, the Ile allele is associated with the occurrence of chromosome segregation errors and tumor progression. Therefore, it is essential to know how BC risk is associated with histopathological characteristics, immunohistochemical characteristics, and genotype polymorphism in a high altitude Ecuadorian mestizo population. In this retrospective case-control study 200 individuals were analyzed. DNA was extracted from 100 healthy and 100 affected women. Genotypes were determined by genomic sequencing. We found significant association between the AURKA T91A (rs2273535) (Phe21Ile) genotype and an increased risk of BC development: Phe/Ile (odds ratio [OR] = 2.6; 95% confidence interval [CI] = 1.4–4.9; P = 0.004), Ile/Ile (OR = 3.8; 95% CI = 1.6–9.0; P = 0.002), and Phe/Ile + Ile/Ile (OR = 2.9; 95% CI = 1.6–5.2; P = 0.001). Additionally, the rs2273535 variant was associated with the tumor grade SBR III (OR = 9.6; 95% CI = 1.0–91.9; P = 0.048) and the Ki-67 ≥ 20 (OR = 16.5; 95% CI = 2.7–101.3; P = 0.002). In brief, this study provides the first evidence where the Ile allele of the AURKA gene could act as potentially predictive biomarker of BC in the high altitude Ecuadorian mestizo population that lives at 2800 m above sea level (masl).

Similar content being viewed by others
Change history
05 September 2017
An erratum to this article has been published.
References
Cancer Research UK. Worldwide cancer statistics. In: Cancer statistics. 2014. www.cancerresearchuk.org/cancer info/cancerstats/world. Accessed 7 Nov 2014
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
World Health Organization. International Agency for Research on Cancer. GLOBOCAN 2012: Estimated cancer incidence, mortality and prevalence worldwide in 2012. 2012. http://globocan.iarc.fr/ Default.aspx. Accessed 7 Nov 2016
Kumar V, Abbas A, Aster J. (2012) Robbins basic pathology. 9th ed. Philadelphia: Saunders
National Cancer Institute at the National Institutes of Health (2014) Histopathologic classification of breast cancer. In: Breast cancer treatment. http://www.cancer.gov/cancertopics/pdq/treatment/breast/healthprofessional/page2. Accessed 7 Nov 2014
Kumar R, Sharma A, Tiwari R (2012) Application of microarray in breast cancer: an overview. J Pharm Bioallied Sci 4:21–26
Banerji S, Cibulskis K, Rangel-Escareno C, Brown K, Carter S, Frederick A, Lawrance M, Sivachenko A, Sougnez C, Zou L, Cortes M, Fernandez-Lopez J, Peng S, Ardlie K, Auclair D, Bautista-Piña V, Duke F, Francis J, Jung J, Maffuz-Aziz A, Onofrio R, Parkin M, Pho N, Quintanar-Jurado V, Ramos A, Rebollar-Vega R, Rodriguez-Cuevas S, Romero-Cordoba S, Schumacher S, Stransky N, Thompson K, Uribe-Figueroa L, Baselga J, Beroukhim R, Polyak K, Sgroi D, Richardson A, Jimenez-Sanchez G, Lander E, Gabriel S, Garraway L, Golub T, Melendez-Zajgla J, Toker A, Getz G, Hidalgo-Miranda A, Meyerson M (2012) Sequence analysis of mutations and translocations across breast cancer subtypes. Nature 486:405–409
Eisenstein M (2015) Startups use short-read data to expand long-read sequencing market. Nat Biotechnol 33:433–435
Wong KM, Hudson TJ, McPherson JD (2011) Unraveling the genetics of cancer: genome sequencing and beyond. Annu Rev Genomics Hum Genet 12:407–430
Meyerson M, Gabriel S, Getz G (2010) Advances in understanding cancer genomes through second-generation sequencing. Nat Rev Genet 11:685–696
Eifert C, Powers RS (2012) From cancer genomes to oncogenic drivers, tumour dependencies and therapeutic targets. Nat Rev Cancer 12:572–578
Lovly CM, McDonald NT, Chen H, Ortiz-Cuaran S, Heukamp LC, Yan Y, Florin A, Ozretić L, Lim D, Wang L, Chen Z, Chen X, Lu P, Paik PK, Shen R, Jin H, Buettner R, Ansén S, Perner S, Brockmann M, Bos M, Wolf J, Gardizi M, Wright GM, Solomon B, Rusell PA, Rogers TM, Suehara Y, Red-Brewer M, Tieu R, de Stanchina E, Wang Q, Zhao Z, Johonson DH, Horn L, Wong KK, Thomas RK, Ladanyi M, Pao W (2014) Rationale for co-targeting IGF-1R and ALK in ALK fusion-positive lung cancer. Nat Med 20:1027–1034
Xia J, Jia P, Hutchinson KE, Dahlman KB, Johnson D, Sosman J, Pao W, Zhao Z (2014) A meta-analysis of somatic mutations from next generation sequencing of 241 melanomas: a road map for the study of genes with potential clinical relevance. Mol Cancer Ther 13:1918–1928
Jia P, Jin H, Meador CB, Xia J, Ohashi K, Liu L, Pirazzoli V, Dahmal KB, Politi K, Michor F, Zhao Z, Pao W (2013) Next-generation sequencing of paired tyrosine kinase inhibitor-sensitive and –resistant EGFR mutant lung cancer cell lines identifies spectrum of DNA changes associated with drug resistance. Genome Res 23:1434–1445
Dahlman KB, Xia J, Hutchinson K, Ng C, Hucks D, Jia P, Atefi M, Su Z, Branch S, Lyle PL, Hicks DJ, Bozon V, Glaspy JA, Rosen N, Solit DB, Netterville JL, Vnencak-Jones c, Sosman JA, Ribas A, Zhao Z, Pao W (2012) BRAF(L597) mutations in melanoma are associated with sensitivity to MEK inhibitors. Cancer Discov 2:791–797
Cheng F, Zhao J, Zhao Z (2016) Advances in computational approaches for prioritizing driver mutations and significantly mutated genes in cancer genomes. Brief Bioinform 17:642–656
Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA Jr, Kinzler KW (2013) Cancer genome landscapes. Science 339:1546–1558
Khan J, Ezan F, Crémet J, Fautrel A, Gilot D, Lambert M, Benaud C, Troadec MB, Prigent C (2011) Overexpression of active aurora-C kinase results in cell transformation and tumour formation. PLoS One 6:e26512
Lim SK, Gopalan G (2007) Aurora-a kinase interacting protein 1 (AURKAIP1) promotes aurora-a degradation through an alternative ubiquitin-independent pathway. Biochem J 403:119–127
Fu J, Bian M, Jiang Q, Zhang C (2007) Roles of aurora kinases in mitosis and tumorigenesis. Mol Cancer Res 5:1–10
Sun T, Miao X, Wang J, Tan W, Zhou Y, Yu C, Lin D (2004) Functional Phe31Ile polymorphism in aurora a and risk of breast carcinoma. Carcinogenesis 25:2225–2230
Xu J, Wu X, Zhou WH, Liu AW, Wu JB, Deng JY, Yue CF, Yang SB, Wang J, Yuan ZY, Liu Q (2013) Aurora-a identifies early recurrence and poor prognosis and promises a potential therapeutic target in triple negative breast cancer. PLoS One 8:e56919
Mendiola M, Barriuso J, Mariño-Enríquez A, Redondo A, Domínguez-Cáceres A, Hernández-Cortés G, Pérez-Fernández E, Sánchez-Navarro I, Vera JA, Suárez A, Espinosa E, González-Barón M, Palacios J, Hardisson D (2009) Aurora kinases as prognostic biomarkers in ovarian carcinoma. Hum Pathol 40:631–638
Dai ZJ, Kang HF, Wang XJ, Shao YP, Lin S, Zhao Y, Ren HT, Min WL, Wang M, Liu XX (2014) Association between genetic polymorphisms in AURKA (rs2273535 and rs1047972) and breast cancer risk: a meta-analysis involving 37,221 subjects. Cancer Cell Int 14:91
López-Cortés A, Echeverría C, Oña-Cisneros F, Sánchez ME, Herrera C, Cabrera-Andrade A, Rosales F, Ortiz M, Paz-y-Miño C (2015) Breast cancer risk associated with gene expression and genotype polymorphisms of thefolate-metabolizing MTHFR gene: a case-control study in a high altitude Ecuadorian mestizo population. Tumor Biol 36:6451–6461
Rodriguez S, Gaunt TR, Day IN (2009) Hardy-Weinberg equilibrium testing of biological ascertainment for Mendelian randomization studies. Am J Epidemiol 169:505–514
Engle LJ, Simpson CL, Landers JE (2006) Using high-throughput SNP technologies to study cancer. Oncogene 25:1594–1601
Fejerman L, Ahmadiyeh N, Hu D, Huntsman S, Beckman KB, Caswell JL, Tsung K, John EM, Torres-Mejia G, Carvajal-Carmona L, Echeverry MM, Tuazon AM, Ramirez C, COLUMBUS Consortium, Gognoux CR, Eng C, Gonzalez-Burchard E, Henderson B, Le Marchard L, Kooperberg C, Hou L, Agalliu I, Kraft P, Lindström S, Perez-Stable EJ, Haiman CA, Ziv E (2014) Genome-wide association study of breast cancer in Latinas identifies novel protective variants on 6q25. Nat Commun 5:5260
Ruan Y, Song AP, Wang H, Xie YT, Han JY, Sajdik C, Tian XX, Fang WG (2011) Genetic polymorphisms in AURKA and BRCA1 are associated with breast cancer susceptibility in a Chinese Han population. J Pathol 225:535–543
Webb EL, Rudd MF, Houlston RS (2006) Case-control, kin-cohort and meta-analyses provide no support for STK15 F31I as a low penetrance colorectal cancer allele. Br J Cancer 95:1047–1049
Miao X, Sun T, Wang Y, Zhang X, Tan W, Lin D (2004) Functional STK15 Phe31Ile polymorphism is associated with the occurrence and advanced disease status of esophageal squamous cell carcinoma. Cancer Res 64:2680–2683
Tang W, Qiu H, Ding H, Sun B, Wang L, Yin J, Gu H (2013) Association between the STK15 F31I polymorphism and cancer susceptibility: a meta-analysis involving 43,626 subjects. PLoS One 8:e82790
Sun T, Miao X, Wang J, Tan W, Zhou Y, Yu C, Lin D (2004) Functional Phe31Ile polymorphism in aurora a and risk of breast carcinoma. Carcinogenesis 25:2225–2230
Dai Q, Cai QY, Shu XO, Ewart-Toland A, Wen WQ, Balmain A, Gao YT, Zheng W (2004) Synergistic effects of STK15 gene polymorphisms and endogenous estrogen exposure in the risk of breast cancer. Cancer Epidemiol Biomark Prev 13:2065–2070
Lo YL, Yu JC, Chen ST, Yang HC, Fann CS, Mau YC, Shen CY (2005) Breast cancer risk associated with genotypic polymorphism of the mitosis-regulating gene aurora-STK15/BTAK. Int J Cancer 115:276–283
MARIE-GENICA Consortium on Genetic Susceptibility for Menopausal Hormone Therapy Related Breast Cancer Risk (2010) Polymorphisms in the BRCA1 and ABCB1 genes modulate menopausal hormone therapy associated breast cancer risk in postmenopausal women. Breast Cancer Res Treat 120:727–736
Tchatchou S, Wirtenberger M, Hemminki K, Sutter C, Meindl A, Wappenschmidt B, Kiechle M, Bugert P, Schmutzler RK, Bartram CR, Burweinkel B (2007) Aurora kinases a and B and familial breast cancer risk. Cancer Lett 247:266–272
Vidarsdottir L, Bodvarsdottir SK, Hilmarsdottir H, Tryggvadottir L, Eyfjord JE (2007) Breast cancer risk associated with AURKA 91T -->a polymorphism in relation to BRCA mutations. Cancer Lett 250:206–212
Fletcher O, Johnson N, Palles C, dos Santos Silva I, McCormack V, Whittaker J, Ashworth A, Peto J (2006) Inconsistent association between the STK15 F31I genetic polymorphism and breast cancer risk. J Natl Cancer Inst 98:1014–1018
Cox DG, Hankinson SE, Hunter DJ (2006) Polymorphisms of the AURKA (STK15/aurora kinase) gene and breast cancer risk (United States). Cancer Causes Control 17:81–83
Egan KM, Newcomb PA, Ambrosone CB, Trentham-Dietz A, Titus-Ernstoff L, Hampton JM, Kimura MT, Nagase H (2004) STK15 polymorphism and breast cancer risk in a population-based study. Carcinogenesis 25:2149–2153
Royce ME, Xia W, Sahin AA, Katayama H, Johnston DA, Hortobagyi G, Hung MC (2004) STK15/ aurora-a expression in primary breast tumors is correlated with nuclear grade but not with prognosis. Cancer 100:12–19
Loman N, Johannson O, Kristoffersson U, Olsson H, Borg A (2001) Family history of breast and ovarian cancers and BRCA1 and BRCA2 mutations in a population-based series of early-onset breast cancer. J Natl Cancer Inst 93:1215–1223
Lindström L, Karlsson E, Wilking UM, Johansson U, Hartman J, Lidbrink EK, Hatschek T, Skoog L, Bergh J (2012) Clinically used breast cancer markers such as estrogen receptor, progesterone receptor and human epidermal growth factor receptor 2 are unstable throughout tumor progression. J Clin Oncol 30:2601–2608
Colditz G, Rosner BA, Chen WY, Holmes MD, Hankinson SE (2004) Risk factors for breast cancer according to estrogen and progesterone receptor status. J Natl Cancer Inst 96:218–228
López-Cortés A, Jaramillo-Koupermann G, Muñoz MJ, Cabrera A, Echeverría C, Rosales F, Vivar N, Paz-y-Miño C (2013) Genetic polymorphisms in MTHFR (C677T, A1298C), MTR (A2756G) and MTRR (A66G) genes associated with pathological characteristics of prostate cancer in the Ecuadorian population. Am J Med Sci 346:447–454
Paz-y-Miño C, Robles P, Salazar C, Leone PE, García Cárdenas JM, Naranjo M, López-Cortés A (2016) Positive association of the androgen receptor CAG repeat length polymorphism with the risk of prostate cancer. Mol Med Rep 14:1791–1798
Paz-y-Miño C, López-Cortés A, Muñoz MJ, Cabrera A, Castro B, Sánchez ME (2010) Incidence of the L858R and G719S mutations of the epidermal growth factor receptor oncogene in an Ecuadorian population with lung cancer. Cancer Genet Cytogenet 196:201–203
Paz-y-Miño C, Muñoz MJ, López-Cortés A, Cabrera A, Palacios A, Castro B, Paz-y-Miño N, Sánchez ME (2010) Frequency of polymorphisms pro198leu in GPX-1 gene and ile58thr in MnSOD gene in the altitude Ecuadorian population with bladder cancer. Oncol Res 18:395–400
Paz-y-Miño C, López-Cortés A, Muñoz MJ, Castro B, Cabrera A, Sánchez ME (2010) Relationship of an hRAD54 gene polymorphism (2290 C/T) in an Ecuadorian population with chronic myelogenous leukemia. Genet Mol Biol 33:646–649
Quiñones LA, Lavanderos MA, Cayun JP, Garcia-Martin E, Agundez JA, Caceres DD, Roco AM, Morales JE, Herrera L, Encina G, Isaza CA, Redal MA, Larovere L, Soria NW, Eslava-Schmalbach J, Castaneda-Hernandez G, López-Cortés A, Magno LA, Lopez M, Chiurillo MA, Rodeiro I, Castro de Guerra D, Teran E, Estevez-Carrizo F, Lares Assef I (2014) Perception of the usefulness of drug/gene pairs and barriers for pharmacogenomics in Latin America. Curr Drug Metab 15:202–208
López-Cortés A, Guerrero S, Redal M, Alvarado A, Quiñones L (2017) State of Art of Cancer Pharmacogenomics in Latin American Populations. Int J Mol Sci 18(6):639
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
None.
Rights and permissions
About this article
Cite this article
López-Cortés, A., Cabrera-Andrade, A., Oña-Cisneros, F. et al. Breast Cancer Risk Associated with Genotype Polymorphisms of the Aurora Kinase a Gene (AURKA): a Case-Control Study in a High Altitude Ecuadorian Mestizo Population. Pathol. Oncol. Res. 24, 457–465 (2018). https://doi.org/10.1007/s12253-017-0267-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-017-0267-6