Abstract
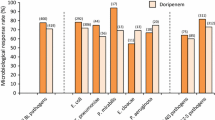
Antibiotic-resistant Escherichia coli are common causative agents of human urinary tract infections. Organotin compounds (OTCs) are man-made chemicals that may affect the renal function of exposed humans and rodents. OTCs are widely recognized as bactericides. However, many environmental and a few clinically relevant bacteria have been found resistant to high concentrations of some OTCs. We examined the susceptibility from 47 E. coli clinical isolates to 12 antibiotics and 5 OTCs. Minimum inhibitory concentrations were determined by the fully automated Sensititre™ ARIS™ 2X system, and E. coli strains were classified as resistant, intermediate resistant, or sensitive, according to the M07-A10 and M100-S26 criteria from the National Committee for Clinical Laboratory Standards. All 47 E. coli strains were susceptible to amikacin but resistant to imipenem and intermediate resistant to ampicillin, cefuroxime, and chloramphenicol. In addition, 26 strains were resistant and 21 intermediate resistant to aztreonam, 24 strains were resistant and 23 intermediate resistant to ceftazidime, 44 strains were intermediate resistant and 3 sensitive to cephalothin, and 43 strains were intermediate resistant and 4 sensitive to ciprofloxacin. Approximately half of the strains were susceptible to cefepime, cefotaxime, and gentamicin. E. coli strains were also found resistant to triphenyltin, tributyltin, dibutyltin, trimethyltin, or dimethyltin at final concentration between 10 μmol/L and 1 mmol/L, during 72-h in vitro culture. However, higher in vitro growth inhibition was induced by these OTCs in the presence of the efflux pump inhibitor carbonyl cyanide-m-chlorophenyl hydrazone, which suggests that efflux pumps contribute to making antibiotic-resistant E. coli also resistant to OTCs.


Similar content being viewed by others
References
Abubakar A, Mustafa MB, Johari WLW, Zulkifli SZ, Ismail A, Mohamat-Yusuff FB (2015) Klebsiella sp. FIRD 2, a TBT-resistant bacterium isolated from contaminated surface sediment along Strait of Johor Malaysia. Mar Pollut Bull 101:280–283. https://doi.org/10.1016/j.marpolbul.2015.09.041
Alibert S, N'gompaza Diarra J, Hernandez J, Stutzmann A, Fouad M, Boyer G, Pagès JM (2017) Multidrug efflux pumps and their role in antibiotic and antiseptic resistance: a pharmacodynamic perspective. Expert Opin Drug Metab Toxicol 13:301–309. https://doi.org/10.1080/17425255.2017.1251581
Barbosa CML, Ferrão FM, Graceli JB (2018) Organotin compounds toxicity: focus on kidney. Front Endocrinol (Lausanne) 9:256. https://doi.org/10.3389/fendo.2018.00256
Bhattacharyya T, Sharma A, Akhter J, Pathania R (2017) The small molecule IITR08027 restores the antibacterial activity of fluoroquinolones against multidrug-resistant Acinetobacter baumannii by efflux inhibition. Int J Antimicrob Agents 50:219–226. https://doi.org/10.1016/j.ijantimicag.2017.03.005
Clinical and Laboratory Standards Institute (ed) (2015) M07-A10. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved standard, 10th edn. Clinical and Laboratory Standards Institute, Wayne
Clinical and Laboratory Standards Institute (2016) M100-S26. Performance standards for antimicrobial susceptibility testing; 26th informational supplement. Clinical and Laboratory Standards Institute, Wayne
Cordoba G, Holm A, Hansen F, Hammerum AM, Bjerrum L (2017) Prevalence of antimicrobial resistant Escherichia coli from patients with suspected urinary tract infection in primary care, Denmark. BMC Infect Dis 17:670. https://doi.org/10.1186/s12879-017-2785-y
Coutinho JV, Freitas-Lima LC, Freitas F, Freitas FP, Podratz PL, Magnago RP, Porto ML, Meyrelles SS, Vasquez EC, Brandão PA, Carneiro MT, Paiva-Melo FD, Miranda-Alves L, Silva IV, Gava AL, Graceli JB (2016) Tributyltin chloride induces renal dysfunction by inflammation and oxidative stress in female rats. Toxicol Lett 260:52–69. https://doi.org/10.1016/j.toxlet.2016.08.007
Cruz A, Micaelo N, Felix V, Song JY, Kitamura S, Suzuki S, Mendo S (2013) sugE: a gene involved in tributyltin (TBT) resistance of Aeromonas molluscorum Av27. J Gen Appl Microbiol 59:39–47
De Santiago A, Aguilar-Santelises M (1999) Organotin compounds decrease in vitro survival, proliferation and differentiation of normal human B lymphocytes. Human Exp Toxicol 18:619–624. https://doi.org/10.1191/096032799678839437
EFSA (2004) Opinion of the scientific panel on contaminants in the food chain on a request from the commission to assess the health risks to consumers associated with exposure to organotins in foodstuffs. Available from http://www.efsa.europa.eu/sites/default/files/scientific_output/files/main_documents/102.pdf
Fang L, Xu C, Li J, Borggaard OK, Wang D (2017) The importance of environmental factors and matrices in the adsorption, desorption, and toxicity of butyltins: a review. Environ Sci Pollut Res Int 24:9159–9173. https://doi.org/10.1007/s11356-017-8449-z
Fonseca-Salazar MA, Diaz-Avalos C, Castañon-Martinez MT, Tapia-Palacios MA, Mazari-Hiriart M (2016) Microbial indicators, opportunistic bacteria, and pathogenic protozoa for monitoring urban wastewater reused for irrigation in the proximity of a megacity. Ecohealth 13:672–686. https://doi.org/10.1007/s10393-016-1172-2
Graceli JB, Sena GC, Lopes PF, Zamprogno GC, da Costa MB, Godoi AF, Dos Santos DM, de Marchi MR, Dos Santos Fernandez MA (2013) Organotins: a review of their reproductive toxicity, biochemistry, and environmental fate. Reprod Toxicol 36:40–52. https://doi.org/10.1016/j.reprotox.2012.11.008
Hassan HA, Dawah SE, El-Sheekh MM (2018) Monitoring the degradation capability of novel haloalkaliphilic tributyltin chloride (TBTCl) resistant bacteria from butyltin-polluted site. Rev Argent Microbiol 51:39–46. https://doi.org/10.1016/j.ram.2017.12.002
Jamborova I, Johnston BD, Papousek I, Kachlikova K, Micenkova L, Clabots C, Skalova A, Chudejova K, Dolejska M, Literak I, Johnson JR. (2018) Extensive genetic commonality among wildlife, wastewater, community, and nosocomial isolates of Escherichia coli sequence type 131 (H30R1 and H30Rx sub clones) that carry blaCTX-M-27 or blaCTX-M-15. Antimicrob Agents Chemother pii: AAC.00519-18. https://doi.org/10.1128/AAC.00519-18
Jana B, Panja S, Saha S, Basu T (2009) Mechanism of protonophores-mediated induction of heat-shock response in Escherichia coli. BMC Microbiol 9:20. https://doi.org/10.1186/1471-2180-9-20
Jude F, Arpin C, Brachet-Castang C, Capdepuy M, Caumette P, Quentin C (2004) TbtABM, a multidrug efflux pump associated with tributyltin resistance in Pseudomonas stutzeri. FEMS Microbiol Lett 232:7–14. https://doi.org/10.1016/S0378-1097(04)00012-6
Karkman A, Do TT, Walsh F, Virta MPJ (2018) Antibiotic-resistance genes in wastewater. Trends Microbiol 26:220–228. https://doi.org/10.1016/j.tim.2017.09.005
Lagunas-Rangel FA (2018) Antimicrobial susceptibility profiles of bacteria causing urinary tract infection in Mexico - single center experience with 10 years results. J Glob Antimicrob Resist. https://doi.org/10.1016/j.jgar.2018.03.004
Landrigan PJ, Sly JL, Ruchirawat M, Silva ER, Huo X, Diaz-Barriga F, Zar HJ, King M, Ha EH, Asante KA, Ahanchian H, Sly PD (2016) Health consequences of environmental exposures: changing global patterns of exposure and disease. Ann Glob Health 82:10–19. https://doi.org/10.1016/j.aogh.2016.01.005
Lehn N, Stöwer-Hoffmann J, Kott T, Strassner C, Wagner H, Krönke M, Schneider-Brachert W (1996) Characterization of clinical isolates of Escherichia coli showing high levels of fluoroquinolone resistance. J Clin Microbiol 34:597–602
Li D, Zeng S, He M, Gu AZ (2016) Water disinfection by-products induce antibiotic resistance. Role of environmental pollutants in resistance phenomena. Environ Sci Technol 50:3193–3201. https://doi.org/10.1021/acs.est.5b05113
Lin JL, Hsueh S (1993) Acute nephropathy of organotin compounds. Am J Nephrol 13:124–128. https://doi.org/10.1159/000168601
Mahanty S, Raghav D, Rathinasamy K (2017) In vitro evaluation of the cytotoxic and bactericidal mechanism of the commonly used pesticide triphenyltin hydroxide. Chemosphere 183:339–352. https://doi.org/10.1016/j.chemosphere.2017.05.117
Mahfouz N, Caucci S, Achatz E, Semmler T, Guenther S, Berendonk TU, Schroeder M (2018) High genomic diversity of multi-drug resistant wastewater Escherichia coli. Sci Rep 8:8928. https://doi.org/10.1038/s41598-018-27292-6
Maillard JY (2018) Resistance of bacteria to biocides. Microbiol Spectr 6. https://doi.org/10.1128/microbiolspec.ARBA-0006-2017
Mann R, Mediati DG, Duggin IG, Harry EJ, Bottomley AL (2017) Metabolic adaptations of uropathogenic E. coli in the urinary tract. Front Cell Infect Microbiol 7:241. https://doi.org/10.3389/fcimb.2017.00241 eCollection 2017
Mazzariol A, Bazaj A, Cornaglia G (2017) Multi-drug-resistant Gram-negative bacteria causing urinary tract infections: a review. J Chemother 29(S1):2–9. https://doi.org/10.1080/1120009X.2017.1380395
Mimura H, Yagi M, Yoshida K (2017) Environmental impact of tributyltin-resistant marine bacteria in the indigenous microbial population of tributyltin-polluted surface sediments. Biocontrol Sci 22:89–96. https://doi.org/10.4265/bio.22.89
Moges F, Endris M, Belyhun Y, Worku W (2014) Isolation and characterization of multiple drug resistance bacterial pathogens from waste water in hospital and non-hospital environments, Northwest Ethiopia. BMC Res Notes 7:215. https://doi.org/10.1186/1756-0500-7-215
Pal C, Bengtsson-Palme J, Kristiansson E, Larsson DG (2015) Co-occurrence of resistance genes to antibiotics, biocides and metals reveals novel insights into their co-selection potential. BMC Genomics 16:964. https://doi.org/10.1186/s12864-015-2153-5
Paredes-Cervantes V, Diaz-Cedillo F, Aguilar-Santelises M (2013) Higher concentrations of organotin compounds are required to decrease bacterial growth than to kill human cells. In: Méndez-Vilas A (ed) Microbial pathogens and strategies for combating them: science, technology and education, vol 3. Formatex Research Center, pp 1675–1679
Paredes-Cervantes V, Castillo-Vera J, Gomez-Reynoso F, Diaz-Cedillo F, Aguilar-Santelises M (2017) Wastewater from Mexico City contains organotin compounds and organotin-resistant bacteria. Cogent Environ Sci 3:1347996. https://doi.org/10.1080/23311843.2017.1347996
Rantakokko P, Main KM, Wohlfart-Veje C, Kiviranta H, Airaksinen R, Vartiainen T, Skakkebæk NE, Toppari J, Virtanen HE (2014) Association of placenta organotin concentrations with growth and ponderal index in 110 newborn boys from Finland during the first 18 months of life: a cohort study. Environ Health 13:45. https://doi.org/10.1186/1476-069X-13-45
Ren X, Wu X, Sui G, Gong Z, Yawson E, Wu B, Lai G, Ruan X, Gao H, Zhou F, Su B, Olson JR, Tang X (2015) Chronic trimethyltin chloride exposure and the development of kidney stones in rats. J Appl Toxicol 35:500–507. https://doi.org/10.1002/jat.3054
Richmond GE, Chua KL, Piddock LJ (2013) Efflux in Acinetobacter baumannii can be determined by measuring accumulation of H33342 (bis-benzamide). J Antimicrob Chemother 68:1594–1600. https://doi.org/10.1093/jac/dkt052
Tang X, Li N, Kang L, Dubois AM, Gong Z, Wu B, Lai G, Yang A, Ruan X, Gao H, Zhu G, Ge Y, Zhang J, Lin Z, Olson JR, Ren X (2013a) Chronic low level trimethyltin exposure and the risk of developing nephrolithiasis. Occup Environ Med 70:561–567. https://doi.org/10.1136/oemed-2012-101261
Tang X, Wu X, Dubois AM, Sui G, Wu B, Lai G, Gong Z, Gao H, Liu S, Zhong Z, Lin Z, Olson J, Ren X (2013b) Toxicity of trimethyltin and dimethyltin in rats and mice. Bull Environ Contam Toxicol 90:626–633. https://doi.org/10.1007/s00128-013-0975-x
Tomova A, Ivanova L, Buschmann AH, Rioseco ML, Kalsi RK, Godfrey HP, Cabello FC (2015) Antimicrobial resistance genes in marine bacteria and human uropathogenic Escherichia coli from a region of intensive aquaculture. Environ Microbiol Rep 7:803–809. https://doi.org/10.1111/1758-2229.12327
Tomova A, Ivanova L, Buschmann AH, Godfrey HP, Cabello FC (2018) Plasmid-mediated quinolone resistance (PMQR) genes and class 1 integrons in quinolone-resistant marine bacteria and clinical isolates of Escherichia coli from an aquacultural area. Microb Ecol 75:104–112. https://doi.org/10.1007/s00248-017-1016-9
Ukah UV, Glass M, Avery B, Daignault D, Mulvey MR, Reid-Smith RJ, Parmley EJ, Portt A, Boerlin P, Manges AR (2018) Risk factors for acquisition of multidrug-resistant Escherichia coli and development of community-acquired urinary tract infections. Epidemiol Infect 146:46–57. https://doi.org/10.1017/S0950268817002680
Yang B, Yang F, Wang S, Wang Q, Liu Z, Feng W, Sun F, Xia P (2018) Analysis of the spectrum and antibiotic resistance of uropathogens in outpatients at a tertiary hospital. J Chemother 30:145–149. https://doi.org/10.1080/1120009X.2017.1418646
Zhang Y, Gu AZ, He M, Li D, Chen J (2017) Sub inhibitory concentrations of disinfectants promote the horizontal transfer of multidrug resistance genes within and across genera. Environ Sci Technol 51:570–580. https://doi.org/10.1021/acs.est.6b03132
Acknowledgments
Support from the National Polytechnic Institute (20144249, 2016-279-1-156) is gratefully acknowledged. The IPN, UNAM, and IMSS had no involvement in the decision to submit this manuscript.
Extent of each individual’s contribution
MAS conceived the study, performed research, and wrote the paper. JCV performed research and analyzed data. RGM, AGV, MCM, and LAS performed research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal studies
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 175 kb)
Rights and permissions
About this article
Cite this article
Aguilar-Santelises, M., Castillo-Vera, J., Gonzalez-Molina, R. et al. Clinical isolates of Escherichia coli are resistant both to antibiotics and organotin compounds. Folia Microbiol 65, 87–94 (2020). https://doi.org/10.1007/s12223-019-00707-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-019-00707-1