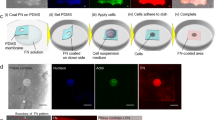
Abstract
Introduction
Directed fibroblast migration is central to highly proliferative processes in regenerative medicine and developmental biology. However, the mechanisms by which single fibroblasts affect each other’s directional decisions, while chemotaxing in microscopic pores, are not well understood.
Methods
We explored effects of cell sequence and mitosis on fibroblast platelet-derived growth factor-BB (PDGF-BB)-induced migration in microfluidic mazes with two possible through paths: short and long. Additionally, image-based modeling of the chemoattractant’s diffusion, consumption and decay, was used to explain the experimental observations.
Results
It both cases, the cells displayed behavior that is contradictory to expectation based on the global chemoattractant gradient pre-established in the maze. In case of the sequence, the cells tend to alternate when faced with a bifurcation: if a leading cell takes the shorter (steeper gradient) path, the cell following it chooses the longer (weaker gradient) path, and vice versa. Image-based modeling of the process showed that the local PDGF-BB consumption by the individual fibroblasts may be responsible for this phenomenon. Additionally, it was found that when a mother cell divides, its two daughters go in opposite directions (even if it means migrating against the chemoattractant gradient and overcoming on-going cell traffic).
Conclusions
It is apparent that micro-confined fibroblasts modify each other’s directional decisions in a manner that is counter-intuitive to what is expected from classical chemotaxis theory. Consequently, accounting for these effects could lead to a better understanding of tissue generation in vivo, and result in more advanced engineered tissue products in vitro.






Similar content being viewed by others
References
Abercrombie, M., and J. E. Heaysman. Observations on the social behaviour of cells in tissue culture. I. Speed of movement of chick heart fibroblasts in relation to their mutual contacts. Exp. Cell. Res. 5(1):111–131, 1953.
Abercrombie, M., and J. E. Heaysman. Observations on the social behaviour of cells in tissue culture. Ii. Monolayering of fibroblasts. Exp. Cell. Res. 6(2):293–306, 1954.
Akar, B., B. Jiang, S. I. Somo, A. A. Appel, J. C. Larson, K. M. Tichauer, and E. M. Brey. Biomaterials with persistent growth factor gradients in vivo accelerate vascularized tissue formation. Biomaterials 72:61–73, 2015.
Albini, A., B. C. Adelmann-Grill, and P. K. Muller. Fibroblast chemotaxis. Coll. Relat. Res. 5(3):283–296, 1985.
Albrecht-Buehler, G. The phagokinetic tracks of 3t3 cells. Cell 11(2):395–404, 1977.
Albrecht-Buehler, G. Daughter 3t3 Cells. Are They Mirror Images of Each Other? J. Cell. Biol. 72(3):595–603, 1977.
Ambravaneswaran, V., I. Y. Wong, A. J. Aranyosi, M. Toner, and D. Irimia. Directional decisions during neutrophil chemotaxis inside bifurcating channels. Integr. Biol. 2(11–12):639–647, 2010.
Beacham, D. A., and E. Cukierman. Stromagenesis: the changing face of fibroblastic microenvironments during tumor progression. Semin. Cancer. Biol. 15(5):329–341, 2005.
Castello-Cros, R., and E. Cukierman. Stromagenesis during tumorigenesis: characterization of tumor-associated fibroblasts and stroma-derived 3d matrices. Methods Mol. Biol. 522:275–305, 2009.
Christensen, S. T., I. R. Veland, A. Schwab, M. Cammer, and P. Satir. Analysis of primary cilia in directional cell migration in fibroblasts. Methods Enzymol. 525:45–58, 2013.
Costa, G., K. I. Harrington, H. E. Lovegrove, D. J. Page, S. Chakravartula, K. Bentley, and S. P. Herbert. Asymmetric division coordinates collective cell migration in angiogenesis. Nat. Cell. Biol. 18(12):1292–1301, 2016.
De Donatis, A., G. Comito, F. Buricchi, M. C. Vinci, A. Parenti, A. Caselli, G. Camici, G. Manao, G. Ramponi, and P. Cirri. Proliferation versus migration in platelet-derived growth factor signaling: the key role of endocytosis. J. Biol. Chem. 283(29):19948–19956, 2008.
Dona, E., J. D. Barry, G. Valentin, C. Quirin, A. Khmelinskii, A. Kunze, S. Durdu, L. R. Newton, A. Fernandez-Minan, W. Huber, M. Knop, and D. Gilmour. Directional tissue migration through a self-generated chemokine gradient. Nature 503(7475):285–289, 2013.
Ellison, D., A. Mugler, M. D. Brennan, S. H. Lee, R. J. Huebner, E. R. Shamir, L. A. Woo, J. Kim, P. Amar, I. Nemenman, A. J. Ewald, and A. Levchenko. Cell-cell communication enhances the capacity of cell ensembles to sense shallow gradients during morphogenesis. Proc. Natl. Acad. Sci. USA 113(6):E679–E688, 2016.
Franz, C. M., G. E. Jones, and A. J. Ridley. Cell migration in development and disease. Dev. Cell. 2(2):153–158, 2002.
Haugh, J. M. Deterministic model of dermal wound invasion incorporating receptor-mediated signal transduction and spatial gradient sensing. Biophys. J. 90(7):2297–2308, 2006.
Kirfel, G., A. Rigort, B. Borm, and V. Herzog. Cell Migration: mechanisms of rear detachment and the formation of migration tracks. Eur. J. Cell Biol. 83(11–12):717–724, 2004.
Kisseleva, T., and D. A. Brenner. Mechanisms of fibrogenesis. Exp. Biol. Med. 233(2):109–122, 2008.
Kisseleva, T., and D. A. Brenner. Fibrogenesis of parenchymal organs. Proc. Am. Thorac. Soc. 5(3):338–342, 2008.
Kurosaka, S., and A. Kashina. Cell biology of embryonic migration. Birth Defects Res. C 84(2):102–122, 2008.
Leong, M. C., S. R. K. Vedula, C. T. Lim, and B. Ladoux. Geometrical constraints and physical crowding direct collective migration of fibroblasts. Commun. Integr. Biol. 6(2):e23197, 2013.
Lepisto, J., M. Laato, J. Niinikoski, C. Lundberg, B. Gerdin, and C. H. Heldin. Effects of homodimeric isoforms of platelet-derived growth factor (Pdgf-Aa and Pdgf-Bb) on wound healing in rat. J. Surg. Res. 53(6):596–601, 1992.
Lepisto, J., J. Peltonen, M. Vaha-Kreula, J. Niinikoski, and M. Laato. Platelet-derived growth factor isoforms Pdgf-Aa, -Ab and -Bb exert specific effects on collagen gene expression and mitotic activity of cultured human wound fibroblasts. Biochem. Biophys. Res. Commun. 209(2):393–399, 1995.
Lerman, O. Z., R. D. Galiano, M. Armour, J. P. Levine, and G. C. Gurtner. Cellular dysfunction in the diabetic fibroblast: impairment in migration, vascular endothelial growth factor production, and response to hypoxia. Am. J. Pathol. 162(1):303–312, 2003.
Levinstone, D., M. Eden, and E. Bell. Similarity of sister-cell trajectories in fibroblast clones. J. Cell. Sci. 59:105–119, 1983.
Mak, M., and D. Erickson. Mechanical Decision trees for investigating and modulating single-cell cancer invasion dynamics. Lab Chip 14(5):964–971, 2014.
Menon, S. N., J. A. Flegg, S. W. McCue, R. C. Schugart, R. A. Dawson, and D. L. McElwain. Modelling the interaction of keratinocytes and fibroblasts during normal and abnormal wound healing processes. Proc. Biol. Sci. 279(1741):3329–3338, 2012.
Miron-Mendoza, M., X. Lin, L. Ma, P. Ririe, and W. M. Petroll. Individual versus collective fibroblast spreading and migration: regulation by matrix composition in 3-D culture. Exp. Eye. Res. 99:36–44, 2012.
Paul, C. D., P. Mistriotis, and K. Konstantopoulos. Cancer cell motility: lessons from migration in confined spaces. Nat. Rev. Cancer 17(2):131–140, 2017.
Paul, C. D., D. J. Shea, M. R. Mahoney, A. Chai, V. Laney, W. C. Hung, and K. Konstantopoulos. Interplay of the physical microenvironment, contact guidance, and intracellular signaling in cell decision making. FASEB J. 30(6):2161–2170, 2016.
Prentice-Mott, H. V., C. H. Chang, L. Mahadevan, T. J. Mitchison, D. Irimia, and J. V. Shah. Biased migration of confined neutrophil-like cells in asymmetric hydraulic environments. Proc. Natl. Acad. Sci. USA 110(52):21006–21011, 2013.
Rao, S., U. Tata, V. Lin, and J.-C. Chiao. The migration of cancer cells in gradually varying chemical gradients and mechanical constraints. Micromachines 5(1):13, 2014.
Rappel, W.-J. Cell-cell communication during collective migration. Proc. Am. Thorac. Soc. 113(6):1471–1473, 2016.
Reig, G., E. Pulgar, and M. L. Concha. Cell migration: from tissue culture to embryos. Development 141(10):1999–2013, 2014.
Rhee, S. Fibroblasts in three dimensional matrices: cell migration and matrix remodeling. Exp. Mol. Med. 41(12):858–865, 2009.
Rouillard, A. D., and J. W. Holmes. Mechanical regulation of fibroblast migration and collagen remodelling in healing myocardial infarcts. J. Physiol. 590(18):4585–4602, 2012.
Scarpa, E., and R. Mayor. Collective cell migration in development. J. Cell. Biol. 212(2):143–155, 2016.
Scherber, C., A. J. Aranyosi, B. Kulemann, S. P. Thayer, M. Toner, O. Iliopoulos, and D. Irimia. Epithelial cell guidance by self-generated Egf gradients. Integr. Biol. 4(3):259–269, 2012.
Schneider, C. A., W. S. Rasband, and K. W. Eliceiri. Nih Image to Imagej: 25 years of image analysis. Nat. Methods 9(7):671–675, 2012.
Seppä, H., G. Grotendorst, S. Seppä, E. Schiffmann, and G. R. Martin. Platelet-derived growth factor in chemotactic for fibroblasts. J. Cell. Biol. 92(2):584–588, 1982.
Shreiber, D. I., P. A. Enever, and R. T. Tranquillo. Effects of Pdgf-Bb on rat dermal fibroblast behavior in mechanically stressed and unstressed collagen and fibrin gels. Exp. Cell. Res. 266(1):155–166, 2001.
Siegbahn, A., A. Hammacher, B. Westermark, and C. H. Heldin. Differential effects of the various isoforms of platelet-derived growth factor on chemotaxis of fibroblasts, monocytes, and granulocytes. J. Clin. Invest. 85(3):916–920, 1990.
Sriram, G., P. L. Bigliardi, and M. Bigliardi-Qi. Fibroblast heterogeneity and its implications for engineering organotypic skin models in vitro. Eur. J. Cell Biol. 94(11):483–512, 2015.
Trepat, X., Z. Chen, and K. Jacobson. Cell migration. Compr. Physiol. 2(4):2369–2392, 2012.
Tse, H. T. K., W. M. Weaver, and D. Di Carlo. Increased asymmetric and multi-daughter cell division in mechanically confined microenvironments. PLoS One 7(6):e38986, 2012.
Vedel, S., S. Tay, D. M. Johnston, H. Bruus, and S. R. Quake. Migration of cells in a social context. Proc. Am. Thorac. Soc. 110(1):129–134, 2013.
Wong, T., J. A. McGrath, and H. Navsaria. The role of fibroblasts in tissue engineering and regeneration. Br. J. Dermatol. 156(6):1149–1155, 2007.
Yan, J., and D. Irimia. Stochastic Variations of migration speed between cells in clonal populations. Technology (Singap World Sci) 2(3):185–188, 2014.
Acknowledgments
The authors also thank Gustavus and Louise Pfeiffer Research Foundation for their gracious funding of our work. Additionally, the authors would like to thank New Jersey Institute of Technology (NJIT)’s McNair Achievement and Provost Summer Research Programs for providing student labor for this project. A fibroblast donation from Prof. Xiaoyang Xu’s laboratory at NJIT’s Department of Chemical, Biological and Pharmaceutical Engineering is greatly appreciated. Lastly, we would like to thank the anonymous reviewer who provided the order of magnitude estimate of the PDGF uptake rate by a cell in our model, which we have included into the Online Appendix.
Funding
This study was funded by the Gustavus and Louise Pfeiffer Research Foundation’s Major Investment Grant, while the custom mask aligner was in part funded by NSF I-Corps Site Award #: 1450182.
Conflict of interest
Authors Quang Long Pham, Lydia N. Rodrigues, Max A. Maximov, Vishnu Deep Chandran, Cheng Bi, David Chege, Timothy Dijamco, Elisabeth Stein, Nhat Anh Nguyen Tong, Sagnik Basuray, and Roman S. Voronov declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Michael R. King oversaw the review of this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12195_2018_551_MOESM2_ESM.tif
Supplementary material 2 (TIFF 1046 kb) Supplemental Figure 1: Detailed breakdown of the directional decision sequences displayed by the fibroblasts in the maze. (A) Directional choices of the first two cells reaching the maze bifurcation, when the cell sequence is taken into account. (B) Directional choices of any two consecutive cells to reach the maze bifurcation, when the cell sequence is taken into account. N indicates the number of sequences being counted.
12195_2018_551_MOESM3_ESM.tif
Supplementary material 3 (TIFF 132 kb) Supplemental Figure 2: Spatial distribution of mitosis events in different maze segments, overlaid on the PDGF-BB concentration profile from COMSOL. (A) Divisions in a maze segment per total number of divisions in the whole maze. (B) Divisions in a maze segment per total number of cell visits into the same maze segment. Black lines indicate boundaries between the maze segments considered.
12195_2018_551_MOESM4_ESM.avi
Supplementary material 4 (AVI 9708 kb)Supplemental Video 1: Negative control experiment, showing that the cells do not go into the maze in the absence of a PDGF gradient.
Supplementary material 5 (AVI 8015 kb) Supplemental Video 2: Control experiment, showing a correspondence between the simulated PDGF diffusion kinetics (top row) in an empty maze, and those of experimentally-diffused fluorescent dextran (bottom row) with a similar molecular weight. Left column shows the absolute concentration values, in the case of the simulated PDGF, and the fluorescence intensity, in the case of the dextran experiment; right column shows a % difference relative to the steady state values for the same.
12195_2018_551_MOESM6_ESM.avi
Supplementary material 6 (AVI 231104 kb)Supplemental Video 3: Control experiment, showing that cells do not obstruct dextran diffusion in the maze. Left – fluorescence microscopy of the dextran; Right – phase contrast microscopy of the cells. Circular markers highlight instances of the cells spreading across the maze channels.
12195_2018_551_MOESM7_ESM.avi
Supplementary material 7 (AVI 6894 kb)Video 1: Alternating patterns of cell decision making with the first cell selecting the short path
12195_2018_551_MOESM8_ESM.avi
Supplementary material 8 (AVI 8565 kb) Video 2: Alternating patterns of cell decision making with the first cell selecting the long path
12195_2018_551_MOESM9_ESM.avi
Supplementary material 9 (AVI 3423 kb) Video 3: Image-based model of fibroblasts consuming the chemoattractant in the maze. The PDGF-BB concentration scaled by the exit boundary condition concentration. Scale bar is 100 μm. Although the simulation is performed with a Δtsimulation = 1min, the frames shown in this video correspond to frequency at which the images are captured by the microscope, Δtmicroscope = 15 minutes (while the intermediate frames are omitted for clarity). The video frames correspond to the acquisition Δtmicroscope = 15 minutes, while the simulation
12195_2018_551_MOESM10_ESM.avi
Supplementary material 10 (AVI 4854 kb) Video 4: Image-based model of fibroblasts modifying the chemoattractant gradient in the maze. Scale bar is 100 μm. Although the simulation is performed with a Δtsimulation = 1min, the frames shown in this video correspond to the frequency at which the images are captured by the microscope, Δtmicroscope = 15 minutes (while the intermediate frames are omitted for clarity).
12195_2018_551_MOESM11_ESM.avi
Supplementary material 11 (AVI 1633 kb) Video 5: Daughter cells following each other in the same direction after division.
12195_2018_551_MOESM12_ESM.avi
Supplementary material 12 (AVI 2127 kb)Video 6: Daughter cells moving in the opposite directions following a cell division.
Rights and permissions
About this article
Cite this article
Pham, Q.L., Rodrigues, L.N., Maximov, M.A. et al. Cell Sequence and Mitosis Affect Fibroblast Directional Decision-Making During Chemotaxis in Microfluidic Mazes. Cel. Mol. Bioeng. 11, 483–494 (2018). https://doi.org/10.1007/s12195-018-0551-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12195-018-0551-x