Abstract
Objective
To evaluate the relationship between microscopic cancerous involvement of surgical margin and recurrence in patients with differentiated papillary thyroid cancer (PTC) who underwent total thyroidectomy followed by high-dose radioactive iodine ablation (HDRIA).
Methods
Consecutive 197 PTC patients (184 women; mean age 44.9 years) who underwent total thyroidectomy without gross residual tumor followed by HDRIA were retrospectively reviewed. Resection margin involvement was evaluated and recurrence of the disease was assessed with clinicopathologically. Recurrence detected within 12 months after HDRIA were defined as early recurrence, detected after 12 months were defined as late recurrence.
Results
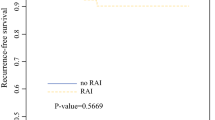
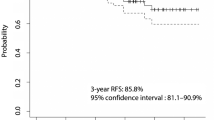
The mean follow-up was 85.9 ± 16.6 months. Twelve patients (6.1%) had microscopic cancerous involvement of surgical margin [margin (+) group], and 185 patients had negative surgical resection margins [margin (−) group]. Three patients (25.0%) in the margin (+) group and 11 patients (5.9%) in the margin (−) group had early recurrence. Margin (+) group showed higher incidence of early recurrence and lower incidence of disease free compared to margin (−) group (25.0 vs. 5.9%, p < 0.01; 66.7 vs. 81.1%, p < 0.01, respectively); however, there was no difference in incidence of late recurrence between the two groups (p = 1.00). There were no significant differences in the disease-free survival between the margin (+) and margin (−) groups after exclusion of early recurrence (p = 0.78).
Conclusions
After high-dose radioactive iodine ablation, PTC patients with microscopic cancerous surgical margin involvement had a higher incidence of early recurrence and no different late recurrence rate compared to patients without microscopic cancerous surgical margin involvement.




Similar content being viewed by others
References
Enewold L, Zhu K, Ron E, Marrogi AJ, Stojadinovic A, Peoples GE, et al. Rising thyroid cancer incidence in the United States by demographic and tumor characteristics, 1980–2005. Cancer Epidemiol Biomarkers Prev. 2009;18:784–91.
Wang TS, Roman SA, Sosa JA. Differentiated thyroid cancer: an update. Curr Opin Oncol. 2011;23:7–12.
Engroff SL, Coletti D. Bisphosphonate related osteonecrosis of the palate: report of a case managed with free tissue transfer. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105:580–2.
Hundahl SA, Fleming ID, Fremgen AM, Menck HR. A National Cancer Data Base report on 53,856 cases of thyroid carcinoma treated in the US, 1985–1995. Cancer. 1998;83:2638–48.
Sipos JA, Mazzaferri EL. Thyroid cancer epidemiology and prognostic variables. Clin Oncol (R Coll Radiol). 2010;22:395–404.
Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, Mandel SJ, et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19:1167–214.
Farahati J, Reiners C, Stuschke M, Muller SP, Stuben G, Sauerwein W, et al. Differentiated thyroid cancer. Impact of adjuvant external radiotherapy in patients with perithyroidal tumor infiltration (stage pT4). Cancer. 1996;77:172–80.
Kim TH, Chung KW, Lee YJ, Park CS, Lee EK, Kim TS, et al. The effect of external beam radiotherapy volume on locoregional control in patients with locoregionally advanced or recurrent nonanaplastic thyroid cancer. Radiat Oncol. 2010;5:69.
Tsang RW, Brierley JD, Simpson WJ, Panzarella T, Gospodarowicz MK, Sutcliffe SB. The effects of surgery, radioiodine, and external radiation therapy on the clinical outcome of patients with differentiated thyroid carcinoma. Cancer. 1998;82:375–88.
Edge SB, Greene FL, Trotti A, Fritz AG, Compton CC, Byrd DR, et al. AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer; 2010.
Tuttle RM, Leboeuf R, Shaha AR. Medical management of thyroid cancer: a risk adapted approach. J Surg Oncol. 2008;97:712–6.
David LS, Mark JL, Ang KK, William HM, David IR, Anesa A, et al. Postoperative external beam radiotherapy for differentiated thyroid cancer: outcomes and morbidity with conformal treatment. Int J Radiat Oncol Biol Phys. 2009;74:1083–91.
Passler C, Scheuba C, Prager G, Kaczirek K, Kaserer K, Zettinig G, et al. Prognostic factors of papillary and follicular thyroid cancer: differences in an iodine-replete endemic goiter region. Endocr Relat Cancer. 2004;11:131–9.
Brierley J, Tsang R, Panzarella T, Bana N. Prognostic factors and the effect of treatment with radioactive iodine and external beam radiation on patients with differentiated thyroid cancer seen at a single institution over 40 years. Clin Endocrinol. 2005;63:418–27.
Gonzalez HE, Cruz F, O’Brien A, Goni I, Leon A, Claure R, et al. Impact of preoperative ultrasonographic staging of the neck in papillary thyroid carcinoma. Arch Otolaryngol Head Neck Surg. 2007;133:1258–62.
Kouvaraki MA, Shapiro SE, Fornage BD, Edeiken-Monro BS, Sherman SI, Vassilopoulou-Sellin R, et al. Role of preoperative ultrasonography in the surgical management of patients with thyroid cancer. Surgery. 2003;134:946–54.
Choi JS, Kim J, Kwak JY, Kim MJ, Chang HS, Kim E-K. Preoperative staging of papillary thyroid carcinoma: comparison of ultrasound imaging and CT. Am J Roentgenol. 2009;193:871–8.
Mazzaferri EL, Kloos RT. Current approaches to primary therapy for papillary and follicular thyroid cancer. J Clin Endocrinol Metab. 2001;86:1447–63.
Wilson PC, Millar BM, Brierley JD. The management of advanced thyroid cancer. Clin Oncol (R Coll Radiol). 2004;16:561–8.
Pineda JD, Lee T, Ain K, Reynolds JC, Robbins J. Iodine-131 therapy for thyroid cancer patients with elevated thyroglobulin and negative diagnostic scan. J Clin Endocrinol Metab. 1995;80:1488–92.
Roelants V, Nayer PD, Bouckaert A, Beckers C. The predictive value of serum thyroglobulin in the follow-up of differentiated thyroid cancer. Eur J Nucl Med. 1997;24:722–7.
Duren M, Siperstein AE, Shen W, Duh QY, Morita E, Clark OH. Value of stimulated serum thyroglobulin levels for detecting persistent or recurrent differentiated thyroid cancer in high- and low-risk patients. Surgery. 1999;126:13–9.
Kim TY, Kim WB, Kim ES, Ryu JS, Yeo JS, Kim SC, et al. Serum thyroglobulin levels at the time of 131I remnant ablation just after thyroidectomy are useful for early prediction of clinical recurrence in low-risk patients with differentiated thyroid carcinoma. J Clin Endocrinol Metab. 2005;90:1440–5.
Pacini F, Lippi F, Formica N, Elisei R, Anelli S, Ceccarelli C, et al. Therapeutic doses of iodine-131 reveal undiagnosed metastases in thyroid cancer patients with detectable serum thyroglobulin levels. J Nucl Med. 1987;28:1888–91.
Toubeau M, Touzery C, Arveux P, Chaplain G, Vaillant G, Berriolo A, et al. Predictive value for disease progression of serum thyroglobulin levels measured in the postoperative period and after (131)I ablation therapy in patients with differentiated thyroid cancer. J Nucl Med. 2004;45:988–94.
Acknowledgments
This work was supported by the Grants of the Korean Ministry of Education, Science and Technology (The Regional Core Research Program/Anti-aging and Well-being Research Center) and Nuclear Research & Development Program of National Research Foundation of Korea (NRF) funded by Ministry of Education, Science & Technology (MEST; grant code: 2010-0017515) and the grant (A102132) of the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hong, C.M., Ahn, BC., Park, J.Y. et al. Prognostic implications of microscopic involvement of surgical resection margin in patients with differentiated papillary thyroid cancer after high-dose radioactive iodine ablation. Ann Nucl Med 26, 311–318 (2012). https://doi.org/10.1007/s12149-012-0574-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-012-0574-7