Abstract
Objective
To investigate the changes in cerebral glucose metabolism in patients with posttraumatic cognitive impairment after memantine therapy.
Methods
We performed serial F-18 fluorodeoxyglucose positron emission tomography studies before and after memantine therapy (20 mg per day) on 17 patients with posttraumatic cognitive impairment using statistical parametric mapping analysis. In addition, covariance analysis was performed to identify regions, where changes in regional cerebral glucose metabolism correlated significantly with increased Mini-Mental Status Examination scores.
Results
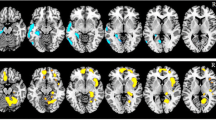
Statistical parametric mapping analysis demonstrated that, compared with baseline, significantly increased cerebral glucose metabolism occurred in both inferior, middle and superior frontal gyri, both angular gyri, both precuneus, the right middle cingulum, the left inferior parietal lobule, the left fusiform gyrus, the left precentral gyrus, the left paracentral lobule, and the left lingual gyrus after memantine therapy (P uncorrected < 0.005). In contrast, cerebral glucose metabolism was significantly decreased in both cerebellum, the left thalamus, the left olfactory, the right middle temporal gyrus, the right amygdala, and the right insula (P uncorrected < 0.005). In the correlation analysis, improvements in Mini-Mental Status Examination scores after memantine therapy were significantly associated with increased cerebral glucose metabolism in the inferior and middle frontal gyri and the inferior parietal lobule of the left hemisphere (P corrected < 0.0001).
Conclusions
Our findings indicate that the prefrontal and the parietal association cortices may be the relevant structures for the pharmacological response to memantine therapy in patients with posttraumatic cognitive impairment.



Similar content being viewed by others
References
Waxweiler RJ, Thurman D, Sniezek J, Sosin D, O’Neil J. Monitoring the impact of traumatic brain injury: a review and update. J Neurotrauma. 1995;12:509–16.
Arciniegas D, Adler L, Topkoff J, Cawthra E, Filley CM, Reite M. Attention and memory dysfunction after traumatic brain injury: cholinergic mechanisms, sensory gating, and a hypothesis for further investigation. Brain Inj. 1999;13:1–13.
Arciniegas DB, Held K, Wagner P. Cognitive impairment following traumatic brain injury. Curr Treat Options Neurol. 2002;4:43–57.
Arciniegas DB, Silver JM. Pharmacotherapy of posttraumatic cognitive impairments. Behav Neurol. 2006;17:25–42.
Phillips JP, Devier DJ, Feeney DM. Rehabilitation pharmacology: bridging laboratory work to clinical application. J Head Trauma Rehabil. 2003;18:342–56.
Challman TD, Lipsky JJ. Methylphenidate: its pharmacology and uses. Mayo Clinic Proc. 2000;75:711–21.
Masanic CA, Bayley MT, VanReekum R, Simard M. Open-label study of donepezil in traumatic brain injury. Arch Phys Med Rehabil. 2001;82:896–901.
Ben Smal D, Samuel C, Rouy-Thenaisy K, Rgnault J, Azouvi P. Bromocriptine in traumatic brain injury. Brain Inj. 2006;20:111–5.
Silver JM, McAllister TW, Arciniegas DB. Depression and cognitive complaints following mild traumatic brain injury. Am J Psychiatry. 2009;166:653–61.
Areosa SA, Sherriff F, McShane R. Memantine for dementia. Cochrane Database Syst Rev 2005:CD003154.
Parsons CG, Danysz W, Quack G. Memantine is a clinically well tolerated N-methyl-d-aspartate (NMDA) receptor antagonist—a review of preclinical data. Neuropharmacology. 1999;38:735–67.
Rao VL, Dogan A, Todd KG, Bowen KK, Dempsey RJ. Neuroprotection by memantine, a non-competitive NMDA receptor antagonist after traumatic brain injury in rats. Brain Res. 2001;911:96–100.
Muir KW. Glutamate-based therapeutic approaches: clinical trials with NMDA antagonists. Curr Opin Pharmacol. 2006;6:53–60.
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage. 2002;15:273–89.
Orgogozo J, Rigaud A, Stffler A, Mbius H, Forette F. Efficacy and safety of memantine in patients with mild to moderate vascular dementia: a randomized, placebo-controlled trial (MMM 300). Stroke. 2002;33:1834–9.
Mukhin A, Fan L, Faden AI. Activation of metabotropic glutamate receptor subtype mGluR1 contributes to post-traumatic neuronal injury. J Neurosci. 1996;16:6012–20.
Zipfel GJ, Babcock DJ, Lee JM, Choi DW. Neuronal apoptosis after CNS injury: the roles of glutamate and calcium. J Neurotrauma. 2000;17:857–69.
Regner A, Alves LB, Chemale I, Costa MS, Friedman G, Achaval M, et al. Neurochemical characterization of traumatic brain injury in humans. J Neurotrauma. 2001;18:783–92.
Pomara N, Ott BR, Peskind E, Resnick EM. Memantine treatment of cognitive symptoms in mild to moderate Alzheimer disease: secondary analyses from a placebo-controlled randomized trial. Alzheimer Dis Assoc Disord. 2007;21:60–4.
Berthier ML, Green C, Lara JP, Higueras C, Barbancho MA, Dvila G, et al. Memantine and constraint-induced aphasia therapy in chronic poststroke aphasia. Ann Neurol. 2009;65:577–85.
Rogers M, Rasheed A, Moradimehr A, Baumrucker SJ. Memantine (Namenda) for neuropathic pain. Am J Hosp Palliat Care. 2009;26:57–9.
Cerullo MA, Adler CM, Strakowski SM, Eliassen JC, Nasrallah HA, Nasrallah AT. Memantine normalizes brain activity in the inferior frontal gyrus: a controlled pilot fMRI study. Schizophr Res. 2007;97:294–6.
Sencer A, Arica O, Kirin T, Grgl A, Aktan D. Effects of memantine and MK-801 on ischemia in an experimental model of acute subdural hematoma. Neurol Res. 2008;30:497–503.
Liu C, Lin N, Wu B, Qiu Y. Neuroprotective effect of memantine combined with topiramate in hypoxic–ischemic brain injury. Brain Res. 2009;1282:173–82.
Riepe MW, Adler G, Ibach B, Weinkauf B, Tracik F, Gunay I. Domain-specific improvement of cognition on memantine in patients with Alzheimer’s disease treated with rivastigmine. Dement Geriatr Cogn Disord. 2007;23:301–6.
Nakayama N, Okumura A, Shinoda J, Nakashima T, Iwama T. Relationship between regional cerebral metabolism and consciousness disturbance in traumatic diffuse brain injury without large focal lesions: an FDG-PET study with statistical parametric mapping analysis. J Neurol Neurosurg Psychiatry. 2006;77:856–62.
Shiga T, Ikoma K, Katoh C, Isoyama H, Matsuyama T, Kuge Y, et al. Loss of neuronal integrity: a cause of hypometabolism in patients with traumatic brain injury without MRI abnormality in the chronic stage. Eur J Nucl Med Mol Imaging. 2006;33:817–22.
Preuss UW, Bahlmann M, Bartenstein P, Schtz CG, Soyka M. Memantine treatment in alcohol dementia: rapid PET changes and clinical course. Eur Neurol. 2001;45:57–8.
Groc L, Bard L, Choquet D. Surface trafficking of N-methyl-d-aspartate receptors: physiological and pathological perspectives. Neuroscience. 2009;158:4–18.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, Y.W., Shin, JC. & An, Ys. Changes in cerebral glucose metabolism in patients with posttraumatic cognitive impairment after memantine therapy: a preliminary study. Ann Nucl Med 24, 363–369 (2010). https://doi.org/10.1007/s12149-010-0360-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-010-0360-3