Abstract
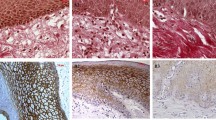
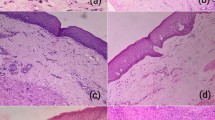
The aim was to evaluate and compare the presence of myofibroblasts in oral squamous cell carcinoma (OSCC), verrucous carcinoma (VC), high-risk epithelial dysplasia (HRED), low-risk epithelial dysplasia (LRED), and normal oral mucosa (NOM). The study consisted of 37 OSCC, 15 VC, 15 HRED, 15 LRED and 15 NOM. α-smooth muscle actin (α-SMA) antibody was used to identify myofibroblasts. The α-SMA expression was not observed in NOM and LRED. The α-SMA was expressed in 97.29% of OSCC, 86.66% of VC, 46.66 % of HRED. The α-SMA expression was significantly higher in OSCC than VC (p = 0.023) and HRED (p < 0.000). The α-SMA expression was significantly higher in VC than HRED (p = 0.043). Myofibroblastic expression, as highlighted by α-SMA, is undetectable in NOM and LRED but increases as the disease progresses from potentially malignant disorders, as HRED to VC to invasive OSCC. Thus, proliferation of myofibroblasts may be used as a stromal marker of oral premalignancy and malignancy.








Similar content being viewed by others
References
Desmouliere A, Guyot CH, Gabbiani G. The stroma reaction myofibroblast: a key player in the control tumor cell behavior. Int J Dev Biol. 2004;48:509–17.
Walter-Yohrling J, Pratt BM, Ledbetter S, Teicher BA. Myofibroblasts enable invasion of endothelial cells into three dimensional tumor cell clusters; a novel in vitro tumor model. Cancer Chemother Pharmacol. 2003;52:263–9.
De wever O, Mareel M. Role of tissue stroma in cancer cell invasion. J Pathol. 2003;200:429–47.
Seemayer TA, Lagace R, Schurch W, Tremblay G. Myofibroblasts in the stroma of invasive and metastatic carcinoma. A possible host response to neoplasia. Am J Surg Pathol. 1979;3:525–33.
Nakayama H, Enzan H, Miyazaki E, Naruse K, Kiyoku H, Hiroi M. The role of myofibroblasts at the tumor border of invasive colorectal adenocarcinomas. Jpn J Clin Oncol. 1998;28(10):615–20.
Baglole CJ, Ray DM, Bernstein SH, Feldon SE, Smith TJ, Sime PJ, Phipps RP. More than structural cells, fibroblasts create and orchestrate the tumor microenvironment. Immunol Invest. 2006;35(3–4):297–325.
De Wever O, Mareel M. Role of myofibroblasts at the invasion front. Biol Chem. 2002;383(1):55–67.
Desmoulière A, Guyot C, Gabbiani G. The stroma reaction myofibroblast: a key player in the control of tumor cell behavior. Int J Dev Biol. 2004;48:509–17.
Zidar N, Gale N, Kambic V, Fischinger J. Proliferation of myofibroblasts in the stroma of epithelial hyperplastic lesions and squamous carcinoma of the larynx. Oncology. 2002;62:381–5.
Adegboyega PA, Mifflin RC, Dimari JF, Saada JI, Powell DW. Immunohistochemical study of myofibroblasts in normal colonic mucosa, hyperplastic polyps, and adenomatous colorectal polyps. Arch Pathol Lab Med. 2002;126:829–36.
Surowiak P, Suchocki S, Györffy B, Gansukh T, Wojnar A, Maciejczyk A, et al. Stromal myofibroblasts in breast cancer: relations between their occurrence, tumor grade and expression of some tumour markers. Folia Histochem Cytobiol. 2006;44(2):111–6.
Warnakulasuriya S, Johnson NW, van der Waal I. Nomenclature and classification of potentially malignant disorders of the oral mucosa. J Oral Pathol Med. 2007;36(10):575–80.
Saito T, Sugiura C, Hirai A, et al. Development of squamous cell carcinoma from pre-existent oral leukoplakia: with respect to treatment modality. Int J Oral Maxillofac Surg. 2001;30(1):49–53.
Reibel J. Prognosis of oral pre-malignant lesions: significance of clinical, histopathological, andmolecular biological characteristics. Crit Rev Oral Biol Med. 2003;14(1):47–62.
Lodi G, Porter S. Management of potentially malignant disorders: evidence and critique. J Oral Pathol Med. 2008;37(2):63–9.
Warnakulasuriya S, Reibel J, Bouquot J, Dabelsteen E. Oral epithelial dysplasia classification systems: predictive value, utility, weaknesses and scope for improvement. J Oral Pathol Med. 2008;37:127–33.
Addante RR, McKenna SJ. Verrucous carcinoma. Oral Maxillofac Surg Clin N Am. 2006;18:513–9.
Mareel M, Madani I. Tumour-Associated Host Cells Participating at Invasion and Metastasis: Targets for Therapy? Acta Chir Belg. 2006;106:635–40.
Beacham DA, Cukierman E. Stromagenesis: the changing face of fibroblastic microenvironments during tumor progression. Semin Cancer Biol. 2005;15:329–41.
Amatangelo MD, Bassi DE, Klein-Szanto AJ, Cukierman E. Stroma-derived three-dimensional matrices are necessary and sufficient to promote desmoplastic differentiation of normal fibroblasts. Am J Pathol. 2005;167:475–88.
Barth PJ, Schenck zu Schweinsberg T, Ramaswamy A, Moll R. CD34+ fibrocytes, alpha-smooth muscle antigen positive myofibroblasts and CD 117 expression in the stroma of invasive squamous cell carcinoma of the oral cavity, Pharynx, and larynx. Virchows Arch. 2004;444(3):231–4.
Yazhou C, Wenlv S, Weidong Z, Licun W. Clinicopathological significance of myofibroblasts in invasive ductal carcinoma of breast. Tumour Biol. 2004;25(5–6):290–5.
Cimpean Am, Raica M, Marita D. Diagnostic significance of immunoexpression of CD34 and smooth muscle cell action in benign and malignant tumors of the breast. Rom J Morphol Embryol. 2005;46:123–9.
Kellermann MG, Sobral LM, da Silva SD, Zecchin KG, Graner E, Lopes MA, et al. Mutual paracrine effects of oral squamous cell carcinoma cells and normal oral fibroblasts: induction of fibroblast to myofibroblast transdifferentiation and modulation of tumor cell proliferation. Oral Oncol. 2008;44(5):509–17.
Kawashiri S, Tanaka A, Noguchi N, Hase T, Nakaya H, Ohara T, et al. Significance of stromal desmoplasia and myofibroblast appearance at the invasive front in squamous cell carcinoma of the oral cavity. Head Neck. 2009;31(10):1346–53.
Etemad-Moghadam S, Khalili M, Tirgary F, Alaeddini M. Evaluation of myofibroblasts in oral epithelial dysplasia and squamous cell carcinoma. J Oral Pathol Med. 2009;38:639–43.
Seifi S, Shafaei S, Shafigh E, Sahabi SM, Ghasemi H. Myofibroblast stromal presence and distribution in squamous epithelial carcinomas, oral dysplasia and hyperkeratosis. Asian Pacific J Cancer Prev. 2011;11:359–64.
Marsh D, Suchak K, Moutasim KA, Vallath S, Hopper C, Jerjes W, et al. Stromal features are predictive of disease mortality in oral cancer patients. J Pathol. 2011;223(4):470–81. doi:10.1002/path.2830. Epub.
Tuxhorn JA, Ayala GE, Smith MJ, Smith VC, Dang TD, Rowley DR. Reactive stroma in human prostate cancer: induction of myofibroblast phenotype and extracellular matrix remodeling. Clin Cancer Res. 2002;8:2912–23.
Vered N, Allon I, Buchner A, et al. Stromal myofibroblasts accompany modifications in the epithelial phenotype of tongue dysplastic and malignant lesions. Cancer Microenviron. 2009;2:49–57.
Shimasaki N, Kuroda N, Miyazaki E, Hayashi Y, Toi M, Hiroi M, et al. The distribution pattern of myofibroblasts in the stroma of human bladder carcinoma depends on their invasiveness. Histol Histopathol. 2006;21(4):349–53.
Chauhan H, Abraham A, Phillips JR, Pringle JH, Walker RA, Jones JL. There is more than one kind of myofibroblast: analysis of CD34 expression in benign, in situ, and invasive breast lesions. J Clin Pathol. 2003;56(4):271–6.
Beacham DA, Cukierman E. Stromagenesis: the changing face of fibroblastic microenvironments during tumor progression. Semin Cancer Biol. 2005;15:329–41.
Weaver VM, Gilbert P. Watch thy neighbor: cancer is a communal affair. J Cell Sci. 2004;117:1287–90.
Varayoud J, Ramos JG, Joazeiro PP, Montes GS, Muñoz De Toro MM, Luque EH. Characterization of fibroblastic cell plasticity in the lamina properia of the rat uterine cervix at term. Biol Reprod. 2001;65(2):375–83.
Lewis MP, Lygoe KA, Nystrom ML, Anderson WP, Speight PM, Marshall JF, et al. Tumour-derived TGF-beta1 modulates myofibroblast differentiation and promotes HGF/SF-dependent invasion of squamous carcinoma cells. Br J Cancer. 2004;90(4):822–32.
Solorzano CC, Jones SC, Pettitjean M, O’Daniel TG, Auffenberg PG, Woost PG, et al. Inhibition of transforming growth factor alpha stimulation of human squamous cell carcinoma of the head and neck with anti-TGF-alpha antibodies and tyrphostin. Ann Surg Oncol. 1997;4(8):670–84.
Jackson-Bernitsas DG, Ichikawa H, Takada Y, Myers JN, Lin XL, Darnay BG, et al. Evidence that TNF-TNFR1-TRADD-TRAF2-RIPTAK1-IKK pathway mediates constitutive NF-kappaB activation and proliferation in human head and neck squamous cell carcinoma. Oncogene. 2006;26(10):1385–97.
Hagedorn H, Elbertzhagen A, Ruoss I, Sauer U, Nerlich AG. Immunohistochemical analysis of major TGF-beta isoforms and their receptors in laryngeal carcinomas. Virchows Arch. 2001;439(4):531–9.
Sobral LM, Montan PF, Martelli-Junior H, Graner E, Coletta RD. Opposite effects of TGF-beta1 and IFN-gamma on transdifferentiation of myofibroblast in human gingival cell cultures. J Clin Periodontol. 2007;34(5):397–406.
Sobral LM, Zecchin KG, Nascimento de Aquino S, Lopes MA, Graner E, Coletta RD. Isolation and characterization of myofibroblast cell lines from oral squamous cell carcinoma. Oncol Rep. 2011;25(4):1013–20. doi:10.3892/or.2011.1161. Epub.
Acknowledgments
The authors extend their sincere thanks to Mr. Rupesh Maladhary, Lab Technician, Dept of Oral and Maxillofacial Pathology, Sharad Pawar Dental College and Hospital, Datta Meghe Institute of Medical Sciences, Sawangi (M), Wardha, Maharashtra, India.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaudhary, M., Gadbail, A.R., Vidhale, G. et al. Comparison of Myofibroblasts Expression in Oral Squamous Cell Carcinoma, Verrucous Carcinoma, High Risk Epithelial Dysplasia, Low Risk Epithelial Dysplasia and Normal Oral Mucosa. Head and Neck Pathol 6, 305–313 (2012). https://doi.org/10.1007/s12105-012-0335-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-012-0335-x