Abstract
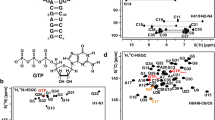
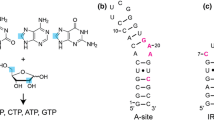
The structures of RNA-aptamer-ligand complexes solved in the last two decades were instrumental in realizing the amazing potential of RNA for forming complex tertiary structures and for molecular recognition of small molecules. For GTP as ligand the sequences and secondary structures for multiple families of aptamers were reported which differ widely in their structural complexity, ligand affinity and ligand functional groups involved in RNA-binding. However, for only one of these families the structure of the GTP-RNA complex was solved. In order to gain further insights into the variability of ligand recognition modes we are currently determining the structure of another GTP-aptamer—the so-called class II aptamer—bound to GTP using NMR-spectroscopy in solution. As a prerequisite for a full structure determination, we report here 1H, 13C, 15N and partial 31P-NMR resonance assignments for the class II GTP-aptamer bound to GTP.

Similar content being viewed by others
References
Aquino-Jarquin G, Toscano-Garibay J (2011) RNA aptamer evolution: two decades of SELEction. Int J Mol Sci 12:9155–9171
Carothers JM, Oestreich SC, Davis JH, Szostak JW (2004) Informational complexity and functional activity of RNA structures. J Am Chem Soc 126:5130–5137
Carothers JM, Oestreich SC, Szostak JW (2006a) Aptamers selected for higher-affinity binding are not more specific for the target ligand. J Am Chem Soc 128:7929–7937
Carothers JM, Davis JH, Chou JJ, Szostak JW (2006b) Solution structure of an informationally complex high-affinity RNA aptamer to GTP. RNA 12:567–579
Davis JH, Szostak JW (2002) Isolation of high-affinity GTP aptamers from partially structured RNA libraries. Proc Natl Acad Sci USA 99:11616–11621
Dieckmann T, Suzuki E, Nakamura GK, Feigon J (1996) Solution structure of an ATP-binding RNA aptamer reveals a novel fold. RNA 2:628–640
Dingley AJ, Grzesiek S (1998) Direct observation of hydrogen bonds in nucleic acid base pairs by internucleotide 2JNN couplings. J Am Chem Soc 120:8293–8297
Duchardt-Ferner E, Weigand JE, Ohlenschläger O, Schmidtke SR, Suess B, Wöhnert J (2010) Highly modular structure and ligand binding by conformational capture in a minimalistic riboswitch. Angew Chem Int Ed 49:6216–6219
Ebrahimi M, Rossi P, Rogers C, Harbison GS (2001) Dependence of 13C NMR chemical shifts on conformations of RNA nucleosides and nucleotides. J Magn Reson 150:1–9
Fürtig B, Richter C, Wöhnert J, Schwalbe H (2003) NMR spectroscopy of RNA. ChemBioChem 4:936–962
Grosshans CA, Cech TR (1991) A hammerhead ribozyme allows synthesis of a new form of the Tetrahymena ribozyme homogenous in length with a 3′ end blocked for transesterification. Nucleic Acids Res 19:3875–3880
Grzesiek S, Bax A (1993) The importance of not saturating H2O in protein NMR: application to sensitivity enhancement and NOE measurements. J Am Chem Soc 115:12593–12594
Hermann T, Patel DJ (2000) Adaptive recognition by nucleic acid aptamers. Science 287:820–825
Jiang F, Kumar RA, Jones RA, Patel DJ (1996) Structural basis of RNA folding and recognition in an AMP-RNA aptamer complex. Nature 382:183–186
Keller R (2004) The computer aided resonance assignment tutorial. CANTINA Verlag, Goldau
Legault P, Farmer BT, Mueller L, Pardi A (1994) Through-bond correlation of adenine protons in a 13C-labeled ribozyme. J Am Chem Soc 116:2203–2204
Marino JP, Schwalbe H, Anklin C, Bermel W, Crothers DM, Griesinger C (1994) A three-dimensional triple-resonance 1H,13C,31P experiment: sequential through-bond correlation of ribose protons and intervening phosphorus along the RNA oligonucleotide backbone. J Am Chem Soc 116:6472–6473
Milligan JF, Uhlenbeck OC (1989) Synthesis of small RNAs using T7 RNA polymerase. Methods Enzymol 180:51–62
Ohlenschläger O, Wöhnert J, Bucci E, Seitz S, Zell R, Häfner S, Ramachandran R, Görlach M (2004) The structure of the stemloop D subdomain of coxsackievirus B3 cloverleaf RNA and its interaction with the proteinase 3C. Structure 12:237–248
Ohlenschläger O, Haumann S, Ramachandran R, Görlach M (2008) Conformational signatures of 13C chemical shifts in RNA ribose. J Biomol NMR 42:139–142
Pardi A, Nikonowicz EP (1992) Simple procedure for resonance assignment of sugar protons in 13C-labeled RNAs. J Am Chem Soc 114:9202–9203
Roth A, Breaker RR (2009) The structural and functional diversity of metabolite-binding riboswitches. Annu Rev Biochem 78:305–334
Sassanfar M, Szostak JW (1993) An RNA motif that binds ATP. Nature 364:550–553
Serganov A, Patel DJ (2012) Metabolite recognition principles and molecular mechanisms underlying riboswitch function. Annu Rev Biophys 41:343–370
Sklenar V, Peterson RD, Rejante MR, Feigon J (1994) Correlation of nucleotide base and sugar protons in a 15N-labeled HIV-1 RNA oligonucleotide by 1H-15N HSQC experiments. J Biomol NMR 4:117–122
Sklenar V, Dieckmann T, Feigon J (1996) Through-bond correlation of imino and aromatic resonances in 13C-, 15N-labeled RNA via heteronuclear TOCSY. J Biomol NMR 7:83–87
Sklenar V, Dieckmann T, Butcher SE, Feigon J (1998) Optimization of triple-resonance HCN experiments for application to larger RNA oligonucleotides. J Mag Res 130:119–124
Wöhnert J, Dingley AJ, Stoldt M, Görlach M, Grzesiek S, Brown LR (1999a) Direct identification of NH···N hydrogen bonds in non-canonical base pairs of RNA by NMR spectroscopy. Nucleic Acids Res 27:3104–3110
Wöhnert J, Ramachandran R, Görlach M, Brown LR (1999b) Triple-resonance experiments for correlation of H5 and exchangeable pyrimidine base hydrogens in 13C,15N-labeled RNA. J Magn Reson 139:430–432
Wöhnert J, Görlach M, Schwalbe H (2003) Triple resonance experiments for the simultaneous correlation of H5/H6 and exchangeable protons of pyrimidine nucleotides in 13C,15N labeled RNA applicable to larger RNA molecules. J Biomol NMR 26:79–83
Wunderlich CH, Spitzer R, Santner T, Fauster K, Tollinger M, Kreutz C (2012) Synthesis of (6-(13)C)pyrimidine nucleotides as spin-labels for RNA dynamics. J Am Chem Soc 134:7558–7569
Acknowledgments
We are grateful to Christian Richter and Manfred Strupf for maintenance of the NMR facility and to Heiko Keller and Kerstin Yacoub for providing the T7-RNA-polymerase. This work was supported by the Center for Biomolecular Magnetic Resonance (BMRZ) of the Johann-Wolfgang-Goethe-University Frankfurt and the Deutsche Forschungsgemeinschaft (DFG) (Grant WO 901/1-1).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wolter, A.C., Duchardt-Ferner, E., Nasiri, A.H. et al. NMR resonance assignments for the class II GTP binding RNA aptamer in complex with GTP. Biomol NMR Assign 10, 101–105 (2016). https://doi.org/10.1007/s12104-015-9646-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12104-015-9646-7