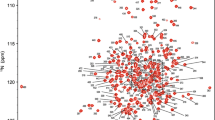
Abstract
Cyclophilins catalyze the reversible peptidyl-prolyl isomerization of their substrates and are present across all kingdoms of life from humans to bacteria. Although numerous biological roles have now been discovered for cyclophilins, their function was initially ascribed to their chaperone-like activity in protein folding where they catalyze the often rate-limiting step of proline isomerization. This chaperone-like activity may be especially important under extreme conditions where cyclophilins are often over expressed, such as in tumors for human cyclophilins (Lee Archiv Pharm Res 33(2): 181–187, 2010), but also in organisms that thrive under extreme conditions, such as theromophilic bacteria. Moreover, the reversible nature of the peptidyl-prolyl isomerization reaction catalyzed by cyclophilins has allowed these enzymes to serve as model systems for probing the role of conformational changes during catalytic turnover (Eisenmesser et al. Science 295(5559): 1520–1523, 2002; Eisenmesser et al. Nature 438(7064): 117–121, 2005). Thus, we present here the resonance assignments of a thermophilic cyclophilin from Geobacillus kaustophilus derived from deep-sea sediment (Takami et al. Extremophiles 8(5): 351–356, 2004). This thermophilic cyclophilin may now be studied at a variety of temperatures to provide insight into the comparative structure, dynamics, and catalytic mechanism of cyclophilins.


Similar content being viewed by others
References
Bahmed K, Henry C et al (2012). Extracellular cyclophilin-A stimulates ERK1/2 phosphorylation in a cell-dependent manner but broadly stimulates nuclear factor kappa B. Cancer Cell International 12
Berjanskii MV, Wishart DS (2005) A simple method to predict protein flexibility using secondary chemical shifts. J Am Chem Soc 127(43):14970–14971
Delaglio F, Grzesiek S et al (1995) Nmrpipe—a multidimensional spectral processing system based on unix pipes. J Biomol NMR 6(3):277–293
Doshi U, McGowan LC et al (2012). Resolving the complex role of enzyme conformational dynamics in catalytic function. Proceedings of the National Academy of Sciences of the United States of America 109(15): 5699–5704
Eisenmesser EZ, Bosco DA et al (2002) Enzyme dynamics during catalysis. Science 295(5559):1520–1523
Eisenmesser EZ, Millet O et al (2005) Intrinsic dynamics of an enzyme underlies catalysis. Nature 438(7064):117–121
Lee J (2010) Role of Cyclophilin A during oncogenesis. Archiv Pharm Res 33(2):181–187
Obchoei SS, Wongkhan S et al (2009) Cyclophilin A: potential functions and therapeutic target for human cancer. Med Sci Monit 15(11):221–232
Obi IR, Nordfelth R et al (2011) Varying dependency of periplasmic peptidylprolyl cis-trans isomerases in promoting Yersinia pseudotuberculosis stress tolerance and pathogenicity. Biochem J 439:321–332
Ottiger M, Zerbe O et al (1997) The NMR solution conformation of unligated human cyclophilin A. J Mol Biol 272(1):64–81
Otting G, Ruckert M et al (2000) NMR experiments for the sign determination of homonuclear scalar and residual dipolar couplings. J Biomol NMR 16(4):343–346
Sattler M, Schleucher J et al (1999) Heteronuclear multidimensional NMR experiments for the structure determination of proteins in solution employing pulsed field gradients. Prog Nucl Magn Reson Spectrosc 34(2):93–158
Schlegel J, Armstrong GS et al (2009a) Characterizing and controlling the inherent dynamics of cyclophilin-A. Protein Sci 18(4):811–824
Schlegel J, Redzic JS et al (2009b) Solution characterization of the extracellular region of CD147 and its interaction with its enzyme ligand cyclophilin-A. J Mol Biol 391:518–535
Takami H, Nishi S et al (2004) Genomic characterization of thermophilic Geobacillus species isolated from the deepest sea mud of the Mariana Trench. Extremophiles 8(5):351–356
Thai V, Renesto P et al (2008) Structural, biochemical, and in vivo characterization of the first virally encoded cyclophilin from the Mimivirus. J Mol Biol 378(1):71–86
Vranken WF, Boucher W et al (2005) The CCPN data model for NMR spectroscopy: development of a software pipeline. Proteins Struct Funct Bioinformatics 59(4):687–696
Acknowledgments
NMR experiments were collected at several facilities described herein. The High Magnetic Field Laboratory (NHMFL) that is supported by cooperative agreement DMR 0654118 between the National Science Foundation and the State of Florida. The Environmental Molecular Sciences Laboratory, which is a national scientific user facility sponsored by the Department of Energy’s Office of Biological and Environmental Research and located at Pacific Northwest National Laboratory. The Rocky Mountain 900 Facility, grant number NIHGM68928. MH is supported by NIH application number 5T32GM008730-13 and EZE is supported by NIH application number 1R01GM096019-01A1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Holliday, M.J., Zhang, F., Isern, N.G. et al. 1H, 13C, and 15N backbone and side chain resonance assignments of thermophilic Geobacillus kaustophilus cyclophilin-A. Biomol NMR Assign 8, 23–27 (2014). https://doi.org/10.1007/s12104-012-9445-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12104-012-9445-3