Abstract
Purpose
Baseline LDH, derived neutrophil–lymphocyte ratio (dNLR) and immune-related adverse events (irAEs) are associated with outcomes of patients with metastatic melanoma (MM). We hypothesized whether dynamic shifts in LDH, dNLR and incidence of irAEs may impact the prognosis of MM patients treated with anti-CTLA4 or anti-PD1 as single agents.
Methods
Retrospective analysis of medical charts from MM patients with prospective monitoring of dNLR, LDH values and irAE incidence. Primary endpoint was overall survival (OS).
Results
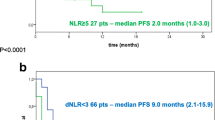
Patients switching from either high dNLR (≥2.5) to low dNLR (HR: 0.14; 0.03–0.74; p = 0.02) or high LDH (≥1.5 × ULN) to low LDH levels (HR: 0.08; 0.01–0.68; p = 0.02) had significantly better OS than those with high dNLR or LDH scores at the end of cycle 2. Longer OS was also observed in patients developing irAEs ≥ grade 2 as compared to no irAEs (HR: 0.2; 0.05–0.89; p = 0.03).
Conclusions
We found that major shifts in dNLR and LDH measures from baseline to cycle 2 measures and shifts from baseline to cycle 2 are significantly associated with OS in MM patients receiving single agent anti-PD1 therapy. Laboratory changes and clinical variables may help optimize prognostic estimates.



Similar content being viewed by others
References
Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711–23.
Boutros C, Tarhini A, Routier E, Lambotte O, Ladurie FL, Carbonnel F, et al. Safety profiles of anti-CTLA-4 and anti-PD-1 antibodies alone and in combination. Nat Rev Clin Oncol. 2016;13(8):473–86.
Hodi FS, Kluger H, Sznol M, Carvajal R, Lawrence D, Atkins M et al. Abstract CT001: durable, long-term survival in previously treated patients with advanced melanoma (MEL) who received nivolumab (NIVO) monotherapy in a phase I trial. Cancer Res. 2016;76(14 Supplement).
Michielin O, van Akkooi A, Ascierto P, Dummer R, Keilholz U. Cutaneous melanoma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2019;30:1884–901.
Diem S, Kasenda B, Spain L, Martin-Liberal J, Marconcini R, Gore M, et al. Serum lactate dehydrogenase as an early marker for outcome in patients treated with anti-PD-1 therapy in metastatic melanoma. Br J Cancer. 2016;114(3):256–61.
Rochet NM, Markovic SN, Porrata LF. The role of complete blood cell count in prognosis. Oncol Hematol Rev. 2012;8(1):76–82.
Hawinkels LJAC, Zuidwijk K, Verspaget HW, de Jonge-Muller ES, van Duijn W, Ferreira V, et al. VEGF release by MMP-9 mediated heparan sulphate cleavage induces colorectal cancer angiogenesis. Eur J Cancer. 2008;44(13):1904–13.
Ray-Coquard I, Cropet C, Van Glabbeke M, Sebban C, Le Cesne A, Judson I, et al. Lymphopenia as a prognostic factor for overall survival in advanced carcinomas, sarcomas, and lymphomas. Cancer Res. 2009;69(13):5383–91.
Capone M, Giannarelli D, Mallardo D, Madonna G, Festino L, Grimaldi AM, et al. Baseline neutrophil-to-lymphocyte ratio (NLR) and derived NLR could predict overall survival in patients with advanced melanoma treated with nivolumab. J Immuno Ther Cancer. 2018;6:74.
Mezquita L, Auclin E, Ferrara R, Charrier M, Remon J, Planchard D, et al. Association of the lung immune prognostic index with immune checkpoint inhibitor outcomes in patients with advanced non-small cell lung cancer. JAMA Oncol. 2018;4(3):351–7.
Hua C, Boussemart L, Mateus C, Routier E, Boutros C, Cazenave H, et al. Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol. 2016;152(1):45–51.
Indini A, Di Guardo L, Cimminiello C, Prisciandaro M, Randon G, De Braud F, et al. Immune-related adverse events correlate with improved survival in patients undergoing anti-PD1 immunotherapy for metastatic melanoma. J Cancer Res Clin Oncol. 2018;145(2):511–21.
Karim ME, Gustafson P, Petkau J, Tremlett J. Comparison of statistical approaches for dealing with immortal time bias in drug effectiveness studies. Am J Epidemiol. 2016;184(4):325–35.
Cristescu R, Mogg R, Ayers M, Albright A, Murphy E, Yearley J. et al Pan-tumor genomic biomarkers for PD-1 checkpoint blockade-based immunotherapy. Science. 2018;362(6411).
Zaretsky JM, Garcia-Diaz A, Shin DS, Escuin-Ordinas H, Hugo W, Hu-Lieskovan S, et al. Mutations associated with acquired resistance to PD-1 blockade in melanoma. N Engl J Med. 2016;375(9):819–29.
Nakamura Y. Biomarkers for immune checkpoint inhibitor-mediated tumor response and adverse events. Front Med. 2019;6:119.
Tarhini A, Kudchadkar RR. Predictive and on-treatment monitoring biomarkers in advanced melanoma: moving toward personalized medicine. Cancer Treat Rev. 2018;71:8–18.
Fässler M, Diem S, Mangana J, Hasan Ali O, Berner F, et al. Antibodies as biomarker candidates for response and survival to checkpoint inhibitors in melanoma patients. J Immuno Ther Cancer. 2019;7:50.
Allavena P, Sica A, Garlanda C, Mantovani A. The Yin-Yang of tumor-associated macrophages in neoplastic progression and immune surveillance. Immunol Rev. 2008;222(1):155–61.
Arlauckas SP, Garris CS, Kohler RH, Kitaoka M, Cuccarese MF, et al. In vivo imaging reveals a tumor-associated macrophage-mediated resistance pathway in anti-PD-1 therapy. Sci Transl Med. 2017;9(389).
Magnuson AM, Kiner E, Ergun A, Park JS, Asinovski N, et al. Identification and validation of a tumor-infiltrating Treg transcriptional signature conserved across species and tumor types. Proc Natl Acad Sci USA. 2018;115(45):E10672–E1068110681.
Freeman ZT, Nirschl TR, Hovelson DH, Johnston RJ, Engelhardt JJ, et al. A conserved intratumoral regulatory T cell signature identifies 4-1BB as a pan-cancer target. J Clin Invest. 2020;130(3):1405–16.
Chakravarthy A, Furness A, Joshi K, et al. Pan-cancer deconvolution of tumour composition using DNA methylation. Nat Commun. 2018;9(1).
Ferrucci PF, Ascierto PA, Pigozzo J, Del Vecchio M, Maio M, Antonini Cappellini GC, et al. Baseline neutrophils and derived neutrophil-to-lymphocyte ratio: prognostic relevance in metastatic melanoma patients receiving ipilimumab. Ann Oncol. 2016;27(4):732–8.
Tavakkoli M, Wilkins CR, Mones JV, Mauro MJ. A novel paradigm between leukocytosis, G-CSF secretion, neutrophil-to-lymphocyte ratio, myeloid-derived suppressor cells, and prognosis in non-small cell lung cancer. Front Oncol. 2019;9:295.
Soda H, Ogawara D, Fukuda Y, Tomono H, Okuno D, et al. Dynamics of blood neutrophil-related indices during nivolumab treatment may be associated with response to salvage chemotherapy for non-small cell lung cancer: a hypothesis-generating study. Thorac Cancer. 2019;10:341–6.
Bilen MA, Martini DJ, Liu Y, Lewis C, Collins HH, et al. The prognostic and predictive impact of inflammatory biomarkers in patients who have advanced-stage cancer treated with immunotherapy. Cancer. 2019;125(1):127–34.
Teulings HE, Limpens J, Jansen SN, Zwinderman AH, Reitsma JB, et al. Vitiligo-like depigmentation in patients with stage III–IV melanoma receiving immunotherapy and its association with survival: a systematic review and meta-analysis. J Clin Oncol. 2015;33:773–8.
Maher VE, Fernandes LL, Weinstock C, Tang S, Agarwal S, et al. Analysis of the association between adverse events and outcome in patients receiving a programmed death protein 1 or programmed death ligand 1 antibody. J Clin Oncol. 2019;37(30):2730–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
RD reported receiving honoraria for speaker activities from Roche, Ipsen, Amgen, Sanofi, Servier Laboratories, Merck Sharp & Dohme; advisory role from Roche and Boehringer Ingelheim; and research grants from Merck and Pierre Fabre. GV reported receiving honoraria for speaker activities from Merck Sharp & Dohme. The rest of the authors declare that they have no conflict of interest.
Ethical approval
All the patients provided consent to participate in secondary research when they provided informed consent to take part of clinical trials in our institution, because of the observational nature of this research manuscript no additional consent was required.
Informed consent
All the patients provided consent to participate in secondary research when they provided informed consent to take part of clinical trials in our institution.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hernando-Calvo, A., García-Alvarez, A., Villacampa, G. et al. Dynamics of clinical biomarkers as predictors of immunotherapy benefit in metastatic melanoma patients. Clin Transl Oncol 23, 311–317 (2021). https://doi.org/10.1007/s12094-020-02420-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02420-9