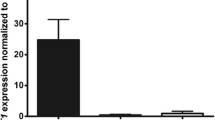
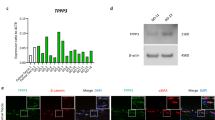
Abstract
Palmar fibromatosis, also known as Dupuytren’s disease (DD), is a common and heritable fibrosis of the hand. It is characterized by the formation of myofibroblastic nodules that can progress to palmar-digital contractures and permanent loss of dexterity. The presence of inflammatory cell infiltrate within these nodules has been interpreted to suggest a pathogenesis mediated by a proinflammatory microenvironment. However, the molecular mechanisms driving the formation of pro-fibrotic microenvironments in this and other fibroses remain unclear. To gain insights into this process, we have assessed the contributions of an alternatively spliced, multi-functional transcription factor, Wilms Tumor 1 (WT1), previously shown to be upregulated in primary myofibroblasts derived from DD tissues. Proinflammatory cytokine stimuli of DD myofibroblasts enhanced the expression of several distinct WT1 variants, the most sustained being a 5′ truncated version of WT1, alternative WT1 (AWT1). Constitutive adenoviral expression of AWT1 in myofibroblasts derived from phenotypically non-fibrotic palmar fascia significantly induced the expression and secretion of proinflammatory cytokines, including some with potential as novel therapeutic targets. In summary, these data implicate roles for sustained AWT1 expression in DD as a transcriptional driver of a proinflammatory fascial milieu.






Similar content being viewed by others
References
Amin EM, Oltean S, Hua J et al (2011) WT1 mutants reveal SRPK1 to be a downstream angiogenesis target by altering VEGF splicing. Cancer Cell 20:768–780. https://doi.org/10.1016/j.ccr.2011.10.016
Anuchapreeda S, Rungrojsakul M, Tima S et al (2019) Co-activation of WT1 and AP-1 proteins on WT1 gene promoter to induce WT1 gene expression in K562 cells. Cell Signal 53:339–347. https://doi.org/10.1016/j.cellsig.2018.11.001
Anwar MU, Al Ghazal SK, Boome RS (2007) Results of surgical treatment of Dupuytren’s disease in women: a review of 109 consecutive patients. J Hand Surg 32:1423–1428. https://doi.org/10.1016/j.jhsa.2007.06.015
Berasain C, Herrero J-I, García-Trevijano ER et al (2003) Expression of Wilms’ tumor suppressor in the liver with cirrhosis: relation to hepatocyte nuclear factor 4 and hepatocellular function. Hepatology (Baltimore, MD) 38:148–157. https://doi.org/10.1053/jhep.2003.50269
Bianchi E, Taurone S, Bardella L et al (2015) Involvement of pro-inflammatory cytokines and growth factors in the pathogenesis of Dupuytren’s contracture: a novel target for a possible future therapeutic strategy? Clin Sci 129:711–720. https://doi.org/10.1042/CS20150088
Borthwick LA, Wynn TA, Fisher AJ (2012) Cytokine mediated tissue fibrosis. Biochim Biophys Acta 1832:1049–1060. https://doi.org/10.1016/j.bbadis.2012.09.014
Braitsch CM, Kanisicak O, van Berlo JH et al (2013) Differential expression of embryonic epicardial progenitor markers and localization of cardiac fibrosis in adult ischemic injury and hypertensive heart disease. J Mol Cell Cardiol 65:108–119. https://doi.org/10.1016/j.yjmcc.2013.10.005
Bruening W, Pelletier J (1996) A non-AUG translational initiation event generates novel WT1 isoforms. J Biol Chem 271:8646–8654. https://doi.org/10.1074/jbc.271.15.8646
Chen P, Wang H, Zhao L et al (2021) Immune checkpoints OX40 and OX40L in small-cell lung cancer: predict prognosis and modulate immune microenvironment. Front Oncol 11:713853. https://doi.org/10.3389/fonc.2021.713853
Crawford J, Raykha C, Charles D et al (2015) WT1 expression is increased in primary fibroblasts derived from Dupuytren’s disease tissues. J Cell Commun Signal. https://doi.org/10.1007/s12079-015-0293-7
Crowley T, Buckley CD, Clark AR (2018) Stroma: the forgotten cells of innate immune memory. Clin Exp Immunol 193:24–36. https://doi.org/10.1111/cei.13149
Dallosso AR, Hancock AL, Brown KW et al (2004) Genomic imprinting at the WT1 gene involves a novel coding transcript (AWT1) that shows deregulation in Wilms’ tumours. Hum Mol Genet 13:405–415. https://doi.org/10.1093/hmg/ddh038
Dallosso AR, Hancock AL, Malik S et al (2007) Alternately spliced WT1 antisense transcripts interact with WT1 sense RNA and show epigenetic and splicing defects in cancer. RNA (new York, NY) 13:2287–2299. https://doi.org/10.1261/rna.562907
Ding L, Tang S, Tang W et al (2022) Perfluorocarbon nanoemulsions enhance therapeutic siRNA delivery in the treatment of pulmonary fibrosis. Adv Sci. https://doi.org/10.1002/advs.202103676
Elhai M, Avouac J, Hoffmann-Vold AM et al (2016) OX40L blockade protects against inflammation-driven fibrosis. Proc Natl Acad Sci USA 113:E3901–E3910. https://doi.org/10.1073/pnas.1523512113
Gonga-Cavé BC, Pena Diaz AM, O’Gorman DB (2021) Biomimetic analyses of interactions between macrophages and palmar fascia myofibroblasts derived from Dupuytren’s disease reveal distinct inflammatory cytokine responses. Wound Repair Regen 29:627–636. https://doi.org/10.1111/wrr.12928
Hamilton TB, Barilla KC, Romaniuk PJ (1995) High affinity binding sites for the Wilms’ tumour suppressor protein WT1. Nucleic Acids Res 23:277–284
Hancock AL, Brown KW, Moorwood K et al (2007) A CTCF-binding silencer regulates the imprinted genes AWT1 and WT1-AS and exhibits sequential epigenetic defects during Wilms’ tumourigenesis. Hum Mol Genet 16:343–354. https://doi.org/10.1093/hmg/ddl478
Hastie ND (2017) Wilms’ tumour 1 (WT1) in development, homeostasis and disease. Development (Cambridge, England) 144:2862–2872. https://doi.org/10.1242/dev.153163
Hewitt SM, Fraizer GC, Wu YJ et al (1996) Differential function of Wilms’ tumor gene WT1 splice isoforms in transcriptional regulation. J Biol Chem 271:8588–8592. https://doi.org/10.1074/jbc.271.15.8588
Hossain A, Nixon M, Kuo MT, Saunders GF (2006) N-terminally truncated WT1 protein with oncogenic properties overexpressed in leukemia. J Biol Chem 281:28122–28130. https://doi.org/10.1074/jbc.M512391200
Karki S, Surolia R, Hock TD et al (2015) Wilms’ tumor 1 (Wt1) regulates pleural mesothelial cell plasticity and transition into myofibroblasts in idiopathic pulmonary fibrosis. Faseb J 28:1122–1131
Kennedy D, Ramsdale T, Mattick J, Little M (1996) An RNA recognition motif in Wilms’ tumour protein (WT1) revealed by structural modelling. Nat Genet 12:329–331
Kramarzova K, Stuchly J, Willasch A et al (2012) Real-time PCR quantification of major Wilms’ tumor gene 1 (WT1) isoforms in acute myeloid leukemia, their characteristic expression patterns and possible functional consequences. Leukemia 26:2086–2095. https://doi.org/10.1038/leu.2012.76
Langerak AW, Williamson KA, Miyagawa K et al (1995) Expression of the Wilms’ tumor gene WT1 in human malignant mesothelioma cell lines and relationship to platelet-derived growth factor A and insulin-like growth factor 2 expression. Genes Chromosom Cancer 12:87–96
Lee KY, Jeon YJ, Kim HG et al (2017) The CUG-translated WT1, not AUG-WT1, is an oncogene. Carcinogenesis 38:1228–1240. https://doi.org/10.1093/carcin/bgx108
Li Y, Chen Y, Miao L et al (2021) Stress-induced upregulation of TNFSF4 in cancer-associated fibroblast facilitates chemoresistance of lung adenocarcinoma through inhibiting apoptosis of tumor cells. Cancer Lett 497:212–220. https://doi.org/10.1016/j.canlet.2020.10.032
Lv L, Chen G, Zhou J et al (2015) WT1-AS promotes cell apoptosis in hepatocellular carcinoma through down-regulating of WT1. J Exp Clin Cancer Res 34:119. https://doi.org/10.1186/s13046-015-0233-7
Mao L-L, Chen W-Y, Ma A-J et al (2020) High serum OX40 ligand correlates with severity and mortality in patients with massive cerebral infarction. Medicine 99:e20883. https://doi.org/10.1097/MD.0000000000020883
Mata R, Yao Y, Cao W et al (2021) The dynamic inflammatory tissue microenvironment: signality and disease therapy by biomaterials. Research 2021:4189516. https://doi.org/10.34133/2021/4189516
McCarty G, Loeb DM (2015) Hypoxia-sensitive epigenetic regulation of an antisense-oriented lncRNA controls WT1 expression in myeloid leukemia cells. PLoS ONE 10:e0119837. https://doi.org/10.1371/journal.pone.0119837
Mella JR, Guo L, Hung V (2018) Dupuytren’s contracture: an evidence based review. Ann Plast Surg 81:S97–S101. https://doi.org/10.1097/SAP.0000000000001607
Miądlikowska E, Rzepka-Wrona P, Miłkowska-Dymanowska J et al (2021) Review: serum biomarkers of lung fibrosis in interstitial pneumonia with autoimmune features-what do we already know? J Clin Med. https://doi.org/10.3390/jcm11010079
Prosser R, Conolly WB (1996) Complications following surgical treatment for Dupuytren’s contracture. J Hand Ther 9:344–348
Qiu L, Gong G, Wu W et al (2021) A novel prognostic signature for idiopathic pulmonary fibrosis based on five-immune-related genes. Ann Transl Med 9:1570–1570. https://doi.org/10.21037/atm-21-4545
Rajesh KR, Rex C, Mehdi H et al (2000) Severe Dupuytren’s contracture of the proximal interphalangeal joint: treatment by two-stage technique. J Hand Surg [Br] 25:442–444
Rius-Pérez S, Pérez S, Martí-Andrés P et al (2020) Nuclear factor kappa B signaling complexes in acute inflammation. Antioxid Redox Signal 33:145–165. https://doi.org/10.1089/ars.2019.7975
Sani AI, Rubab ZE, Usman S et al (2021) Serum levels of OX40 in early and late-stage oral squamous cell carcinoma. Cureus 13:e14597. https://doi.org/10.7759/cureus.14597
Satish L, LaFramboise WA, O’Gorman DB et al (2008) Identification of differentially expressed genes in fibroblasts derived from patients with Dupuytren’s Contracture. BMC Med Genom 1:10. https://doi.org/10.1186/1755-8794-1-10
Satish L, LaFramboise WAWA, Johnson S et al (2012) Fibroblasts from phenotypically normal palmar fascia exhibit molecular profiles highly similar to fibroblasts from active disease in Dupuytren’s Contracture. BMC Med Genom 5:15. https://doi.org/10.1186/1755-8794-5-15
Scotton CJ, Chambers RC (2007) Molecular targets in pulmonary fibrosis: the myofibroblast in focus. Chest 132:1311–1321
Shao Y, Lu J, Cheng C et al (2007) Reversible histone acetylation involved in transcriptional regulation of WT1 gene. Acta Biochim Biophys Sin 39:931–938. https://doi.org/10.1111/j.1745-7270.2007.00363.x
Singh A, Ramesh S, Cibi DM et al (2016) Hippo signaling mediators Yap and Taz are required in the epicardium for coronary vasculature development. Cell Rep 15:1384–1393. https://doi.org/10.1016/j.celrep.2016.04.027
Sontake V, Shanmukhappa SK, DiPasquale BA et al (2015) Fibrocytes regulate wilms tumor 1-positive cell accumulation in severe fibrotic lung disease. J Immunol (Baltimore, Md: 1950) 195:3978–91. https://doi.org/10.4049/jimmunol.1500963
Taniguchi K, Karin M (2018) NF-κB, inflammation, immunity and cancer: coming of age. Nat Rev Immunol 18:309–324. https://doi.org/10.1038/nri.2017.142
Verjee LS, Verhoekx JSN, Chan JKK et al (2013) Unraveling the signaling pathways promoting fibrosis in Dupuytren’s disease reveals TNF as a therapeutic target. Proc Natl Acad Sci USA 110:E928–E937. https://doi.org/10.1073/pnas.1301100110
White SM, Murakami S, Yi C (2019) The complex entanglement of Hippo-Yap/Taz signaling in tumor immunity. Oncogene 38:2899–2909. https://doi.org/10.1038/s41388-018-0649-6
Xia H, Bodempudi V, Benyumov A et al (2014) Identification of a cell-of-origin for fibroblasts comprising the fibrotic reticulum in idiopathic pulmonary fibrosis. Am J Pathol 184:1369–1383. https://doi.org/10.1016/j.ajpath.2014.01.012
Xie K, Xu C, Zhang M et al (2019) Yes-associated protein regulates podocyte cell cycle re-entry and dedifferentiation in adriamycin-induced nephropathy. Cell Death Dis 10:915. https://doi.org/10.1038/s41419-019-2139-3
Ye Y, Raychaudhuri B, Gurney A et al (1996) Regulation of WT1 by phosphorylation: inhibition of DNA binding, alteration of transcriptional activity and cellular translocation. EMBO J 15:5606–5615
Zhang S-Y, Fan Q, Moktefi A et al (2021) CMIP interacts with WT1 and targets it on the proteasome degradation pathway. Clin Transl Med 11:e460. https://doi.org/10.1002/ctm2.460
Acknowledgements
We would like to acknowledge the invaluable contributions of Drs. Bing Siang Gan, Ruby Grewal and Nina Suh from the Roth McFarlane Hand and Upper Limb Centre, London Ontario, for surgically resected palmar fascia tissues. This study was supported by a Canadian Institutes of Health Research (CIHR) Operating Grant (#362714) to DBO.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Luo, J., Tugade, T., Sun, E. et al. Sustained AWT1 expression by Dupuytren’s disease myofibroblasts promotes a proinflammatory milieu. J. Cell Commun. Signal. 16, 677–690 (2022). https://doi.org/10.1007/s12079-022-00677-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-022-00677-z