Abstract
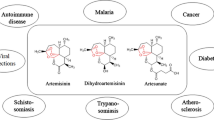
Artemisinins, as a class of bioactive molecules, are mainly derived from the extracts of Artemisia annua L. and are the mainstay for malaria treatment, including severe malaria, uncomplicated malaria and multi-drug resistant malaria. They are well-known for their good tolerability, safety and rapid onset of action. Their efficacy is not only limited to malaria but also extends to a variety of human diseases such as cancer, tuberculosis, viral diseases (e.g. Human cytomegalovirus), immune diseases and parasitic infections like schistosomiasis. Being a cheap and safe drug class, which saves millions of lives at risk from malaria around the globe, can also have significant potential in oncology as they have shown anti-cancer properties in both cell lines and animal models. Active derivatives (e.g. artesunate, artemether and arteether etc.) have also been synthesized which can be used for oral, rectal, intramuscular and intravenous administration. A comprehensive update on the non-malarial use of artemisinins and/or their derivatives and artemisinin-based drug development beyond anti-malarial is discussed in this review. With the collaborative efforts in the clinical pharmacology of artemisinins and novel synthesis of artemisinin analogues, it is likely that artemisinin-based drugs will become an important armamentarium impeding a number of diseases beyond malaria.


Similar content being viewed by others
References
Adenowo AF, Oyinloye BE, Ogunyinka BI, Kappo AP (2015) Impact of human schistosomiasis in sub-Saharan. Africa Brazilian Journal of Infectious Diseases 19:196–205
Ali Z, Mishra N, Baldi A (2016) Development and characterization of arteether-loaded nanostructured lipid carriers for the treatment of malaria. Artificial cells, nanomedicine, and biotechnology 44:545–549
Ali M, Abbasi BH, Ahmad N, Khan H, Ali GS (2017) Strategies to enhance biologically active-secondary metabolites in cell cultures of Artemisia–current trends. Crit Rev Biotechnol 37:833–851
Alonso PL, Brown G, Arevalo-Herrera M, Binka F, Chitnis C, Collins F, Doumbo OK, Greenwood B, Hall BF, Levine MM, Mendis K, Newman RD, Plowe CV, Rodríguez MH, Sinden R, Slutsker L, Tanner M (2011) A research agenda to underpin malaria eradication. PLoS Med 8:e1000406
Aregawi M, Cibulskis RE, Otten M, Williams R (2009) World malaria report 2009. World Health Organization
Bai Z, Guo X-H, Tang C, Yue S-T, Shi L, Qiang B (2018) Effects of Artesunate on the expressions of insulin-like growth Factor-1, Osteopontin and C-Telopeptides of type II collagen in a rat model of. Osteoarthritis Pharmacology 101:1–8
Banyai W, Kirdmanee C, Mii M, Supaibulwatana K (2010) Overexpression of farnesyl pyrophosphate synthase (FPS) gene affected artemisinin content and growth of Artemisia annua L plant cell. Tissue and Organ Culture (PCTOC) 103:255–265
Barger-Kamate B, Forman M, Sangare CO, Haidara ASA, Maiga H, Vaidya D, Djimde A, Arav-Boger R (2016) Effect of artemether-lumefantrine (Coartem) on cytomegalovirus urine viral load during and following treatment for malaria in children. J Clin Virol 77:40–45
Bassat Q (2011) The use of artemether-lumefantrine for the treatment of uncomplicated Plasmodium vivax malaria. PLoS Negl Trop Dis 5:e1325
Begue J-P, Bonnet-Delpon D, Crousse B, Ourevitch M, Chorki F, Grellepois F, Magueur G (2008) Artemisinine derivatives, and uses thereof for treating malaria. Google Patents
Bellone M (2010) Autoimmune Disease: Pathogenesis eLS
Blazquez AG, Fernandez-Dolon M, Sanchez-Vicente L, Maestre AD, Gomez-San Miguel AB, Alvarez M, Serrano MA, Jansen H, Efferth T, Marin JJG, Romero MR (2013) Novel artemisinin derivatives with potential usefulness against liver/colon cancer and viral hepatitis. Bioorg Med Chem 21:4432–4441
Bloom BR (1994) Tuberculosis: pathogenesis, protection, and control. ASM press
Carter R, Mendis KN (2003) Evolutionary and historical aspects of the burden of Malaria. Clin Microbiol Rev 16:173
Chen J, Chen X, Wang F, Gao H, Hu W (2015) Dihydroartemisinin suppresses glioma proliferation and invasion via inhibition of the ADAM17 pathway. Neurol Sci 36:435–440
Chen X, Wong Y, Lim T, Lim W, Lin Q, Wang J, Hua Z (2017) Artesunate activates the intrinsic apoptosis of HCT116 cells through the suppression of fatty acid synthesis and the NF-κB pathway. Molecules 22:1272
Cheng C et al (2018) Induction of autophagy and autophagy-dependent apoptosis in diffuse large B-cell lymphoma by a new antimalarial artemisinin derivative. SM 1044 Cancer medicine 7:380–396
Choi W (2017a) Novel pharmacological activity of artesunate and artemisinin: their potential as anti-tubercular agents. J Clin Med 6(30)
Choi WH (2017b) Novel pharmacological activity of artesunate and artemisinin: their potential as anti-tubercular agents. J Clin Med 6(30)
Committee WMPA, Secretariat (2012) Malaria policy advisory committee to the WHO: conclusions and recommendations of September 2012 meeting. Springer,
Cox FE (2010) History of the discovery of the malaria parasites and their vectors. Parasit Vectors 3:5
Crellen T, Walker M, Lamberton PH, Kabatereine NB, Tukahebwa EM, Cotton JA, Webster JP (2016) Reduced efficacy of praziquantel against Schistosoma mansoni is associated with multiple rounds of mass drug administration. Clin Infect Dis 63:1151–1159
Cui X, Wang Y, Kokudo N, Fang D, Tang W (2010) Traditional Chinese medicine and related active compounds against hepatitis B virus infection bioscience trends 4
Czechowski T, Larson TR, Catania TM, Harvey D, Brown GD, Graham IA (2016) Artemisia annua mutant impaired in artemisinin synthesis demonstrates importance of nonenzymatic conversion in terpenoid metabolism. Proc Natl Acad Sci 113:15150–15155
Doenhoff MJ, Cioli D, Utzinger J (2008) Praziquantel: mechanisms of action, resistance and new derivatives for schistosomiasis. Curr Opin Infect Dis 21:659–667
Durantel D, Carrouée-Durantel S, Branza-Nichita N, Dwek RA, Zitzmann N (2004) Effects of interferon, ribavirin, and iminosugar derivatives on cells persistently infected with noncytopathic bovine viral diarrhea virus. Antimicrob Agents Chemother 48:497–504
Efferth T (2017) From ancient herb to modern drug: Artemisia annua and artemisinin for cancer therapy. In: Seminars in cancer biology, vol 46. Elsevier, pp 65–83
Efferth T, Romero MR, Wolf DG, Stamminger T, Marin JJ, Marschall M (2008) The antiviral activities of artemisinin and artesunate. Clin Infect Dis 47:804–811
El-Lakkany NM, el-Din SHS (2013) Haemin enhances the in vivo efficacy of artemether against juvenile and adult Schistosoma mansoni in mice parasitology research, vol 112, pp 2005–2015
Falzon D, Jaramillo E, Wares F, Zignol M, Floyd K, Raviglione MC (2013) Universal access to care for multidrug-resistant tuberculosis: an analysis of surveillance data. Lancet Infect Dis 13:690–697
Feng X, Chen W, Xiao L, Gu F, Huang J, Tsao BP, Sun L (2017) Artesunate inhibits type I interferon-induced production of macrophage migration inhibitory factor in patients with systemic lupus erythematosus. Lupus 26:62–72
French MD, Churcher TS, Gambhir M, Fenwick A, Webster JP, Kabatereine NB, Basáñez M-G (2010) Observed reductions in Schistosoma mansoni transmission from large-scale administration of praziquantel in Uganda: a mathematical modelling study. PLoS Negl Trop Dis 4:e897
Fröhlich T et al. (2017a) Synthesis of novel hybrids of quinazoline and artemisinin with high activities against Plasmodium falciparum, human cytomegalovirus, and leukemia cells ACS omega 2:2422
Fröhlich T, Reiter C, Ibrahim MM, Beutel J, Hutterer C, Zeitträger I, Bahsi H, Leidenberger M, Friedrich O, Kappes B, Efferth T, Marschall M, Tsogoeva SB (2017b) Synthesis of novel hybrids of Quinazoline and artemisinin with high activities against Plasmodium falciparum. Human Cytomegalovirus, and Leukemia Cells ACS Omega 2:2422–2431
Greenshields AL, Shepherd TG, Hoskin DW (2017) Contribution of reactive oxygen species to ovarian cancer cell growth arrest and killing by the anti-malarial drug artesunate. Mol Carcinog 56:75–93
Guo Z (2016) Artemisinin anti-malarial drugs in China. Acta Pharm Sin B 6:115–124
von Hagens C, Walter-Sack I, Goeckenjan M, Osburg J, Storch-Hagenlocher B, Sertel S, Elsässer M, Remppis BA, Edler L, Munzinger J, Efferth T, Schneeweiss A, Strowitzki T (2017) Prospective open uncontrolled phase I study to define a well-tolerated dose of oral artesunate as add-on therapy in patients with metastatic breast cancer (ARTIC M33/2). Breast Cancer Res Treat 164:359–369
Hahn F, Fröhlich T, Frank T, Bertzbach LD, Kohrt S, Kaufer BB, Stamminger T, Tsogoeva SB, Marschall M (2018) Artesunate-derived monomeric, dimeric and trimeric experimental drugs–their unique mechanistic basis and pronounced antiherpesviral activity. Antivir Res 152:104–110
Hakacova N, Klingel K, Kandolf R, Engdahl E, Fogdell-Hahn A, Higgins T (2013) First therapeutic use of Artesunate in treatment of human herpesvirus 6B myocarditis in a child. J Clin Virol 57:157–160
Han J, Wang H, Kanagarajan S, Hao M, Lundgren A, Brodelius PE (2016) Promoting artemisinin biosynthesis in Artemisia annua plants by substrate channeling. Mol Plant 9:946–948
Harper C (2007) Tuberculosis, a neglected opportunity. Nat Med 13:309–312
He Q, Fu X, Shi P, Liu M, Shen Q, Tang K (2017) Glandular trichome-specific expression of alcohol dehydrogenase 1 (ADH1) using a promoter-GUS fusion in Artemisia annua L plant cell. Tissue and Organ Culture (PCTOC) 130:61–72
Ho WE, Cheng C, Peh HY, Xu F, Tannenbaum SR, Ong CN, Wong WF (2012) Anti-malarial drug artesunate ameliorates oxidative lung damage in experimental allergic asthma. Free Radic Biol Med 53:498–507
Ho WE, Peh HY, Chan TK, Wong WF (2014) Artemisinins: pharmacological actions beyond anti-malarial. Pharmacol Ther 142:126–139
Hou L, Huang H (2016) Immune suppressive properties of artemisinin family drugs. Pharmacol Ther 166:123–127
Hurtley S, Ash C, Roberts L (2010) Landscapes of Infection. In: Landscapes of infection. American Association for the Advancement of Science
Ikram NK, Simonsen HT (2017) A review of biotechnological artemisinin production in plants. Front Plant Sci 8:1966
Ikram K, Binti NK, Beyraghdar Kashkooli A, Peramuna AV, van der Krol AR, Bouwmeester H, Simonsen HT (2017) Stable production of the antimalarial drug artemisinin in the moss Physcomitrella patens. Frontiers in bioengineering and biotechnology 5:47
Jana S, Iram S, Thomas J, Liekens S, Dehaen W (2017) Synthesis and anticancer activity of novel aza-artemisinin derivatives. Bioorg Med Chem 25:3671–3676
Ji Y, Zhang Y-C, Pei L-B, Shi L-L, Yan J-L, Ma X-H (2011) Anti-tumor effects of dihydroartemisinin on human osteosarcoma. Mol Cell Biochem 351:99–108
Jiao Y, Preston S, Koehler AV, Stroehlein AJ, Chang BCH, Simpson KJ, Cowley KJ, Palmer MJ, Laleu B, Wells TNC, Jabbar A, Gasser RB (2017) Screening of the ‘stasis Box’identifies two kinase inhibitors under pharmaceutical development with activity against Haemonchus contortus. Parasit Vectors 10:323
Jones I, Lund A, Riveau G, Jouanard N, Ndione RA, Sokolow SH, De Leo GA (2018) Ecological control of schistosomiasis in sub-Saharan Africa: restoration of predator-prey dynamics to reduce transmission ecology and evolution of infectious disease: pathogen control and public health management in low-income countries:236
Ju R-J et al (2018) Octreotide-modified liposomes containing daunorubicin and dihydroartemisinin for treatment of invasive breast cancer artificial cells, nanomedicine, and biotechnology, pp 1–13
Kadambari S, Atkinson C, Luck S, Macartney M, Conibear T, Harrison I, Booth C, Sharland M, Griffiths PD (2017) Characterising variation in five genetic loci of cytomegalovirus during treatment for congenital infection. J Med Virol 89:502–507
Kaptein SJ et al (2006) The anti-malaria drug artesunate inhibits replication of cytomegalovirus in vitro and in vivo. Antivir Res 69:60–69
Kar Han Lau P, Woods ML, Kanneyalal Ratanjee S, Tharayil John G (2011) Artesunate is ineffective in controlling valganciclovir-resistant cytomegalovirus infection. Clinical Infectious Diseases 52:279–279
Kayser O, Ryden A, Bouwmeester H, Spira CR, Osada H, Muranaka T (2011) The molecular cloning of dihydroartemisinic aldehyde reductase and its implication in artemisinin biosynthesis in Artemisia annua. Planta Med 77:SL28
Keiser J, N’Guessan NA, Adoubryn KD, Silué KD, Vounatsou P, Hatz C, Utzinger J, N’Goran EK (2010) Efficacy and safety of mefloquine, artesunate, mefloquine-artesunate, and praziquantel against Schistosoma haematobium: randomized, exploratory open-label trial. Clin Infect Dis 50:1205–1213
Kim SH, Kang SH, Kang BS (2016) Therapeutic effects of dihydroartemisinin and transferrin against glioblastoma. Nutr Res Pract 10:393–397
Konstat-Korzenny E, Ascencio-Aragón JA, Niezen-Lugo S, Vázquez-López R (2018) Artemisinin and its synthetic derivatives as a. Possible Therapy for Cancer Medical Sciences 6:19
Krishna S, Ganapathi S, Ster IC, Saeed MEM, Cowan M, Finlayson C, Kovacsevics H, Jansen H, Kremsner PG, Efferth T, Kumar D (2015) A randomised, double blind, placebo-controlled pilot study of oral artesunate therapy for colorectal cancer. EBioMedicine 2:82–90
Krungkrai J, Krungkrai SR (2016) Antimalarial qinghaosu/artemisinin: the therapy worthy of a Nobel prize. Asian Pac J Trop Biomed 6:371–375
Kumari K, Keshari S, Sengupta D, Sabat SC, Mishra SK (2017) Transcriptome analysis of genes associated with breast cancer cell motility in response to artemisinin treatment. BMC Cancer 17:858
del Villar LP, Burguillo FJ, López-Abán J, Muro A (2012) Systematic review and meta-analysis of artemisinin based therapies for the treatment and prevention of schistosomiasis. PLoS One 7:e45867
Lai HC, Singh NP, Sasaki T (2013) Development of artemisinin compounds for cancer treatment. Investig New Drugs 31:230–246
Lalloo DG, Shingadia D, Bell DJ, Beeching NJ, Whitty CJ, Chiodini PL (2016) UK malaria treatment guidelines 2016. J Infect 72:635–649
Laxminarayan R, Over M, Smith DL (2006) Will a global subsidy of new antimalarials delay the emergence of resistance and save lives? Health Aff 25:325–336
Leistikow RL, Morton RA, Bartek IL, Frimpong I, Wagner K, Voskuil MI (2010) The mycobacterium tuberculosis DosR regulon assists in metabolic homeostasis and enables rapid recovery from nonrespiring dormancy. J Bacteriol 192:1662–1670
Leto I, Coronnello M, Righeschi C, Bergonzi MC, Mini E, Bilia AR (2016) Enhanced efficacy of artemisinin loaded in transferrin-conjugated liposomes versus stealth liposomes against HCT-8 Colon Cancer cells. ChemMedChem 11:1745–1751
Li Y (2012) Qinghaosu (artemisinin): chemistry and pharmacology. Acta Pharmacol Sin 33:1141–1146
Li X, Li TT, Zhang XH, Hou LF, Yang XQ, Zhu FH, Tang W, Zuo JP (2013a) Artemisinin analogue SM934 ameliorates murine experimental autoimmune encephalomyelitis through enhancing the expansion and functions of regulatory T cell. PLoS One 8:e74108
Li Y, Wang S, Wang Y, Zhou C, Chen G, Shen W, Li C, Lin W, Lin S, Huang H, Liu P, Shen X (2013b) Inhibitory effect of the antimalarial agent artesunate on collagen-induced arthritis in rats through nuclear factor kappa B and mitogen-activated protein kinase signaling pathway. Transl Res 161:89–98
Li Z, Li Q, Wu J, Wang M, Yu J (2016) Artemisinin and its derivatives as a repurposing anticancer agent: what else do we need to do. Molecules 21:1331
Li X, Gu S, Sun D, Dai H, Chen H, Zhang Z (2018) The selectivity of artemisinin-based drugs on human lung normal and cancer cells. Environ Toxicol Pharmacol 57:86–94
Liang Y-J, Luo J, Yuan Q, Zheng D, Liu YP, Shi L, Zhou Y, Chen AL, Ren YY, Sun KY, Sun Y, Wang Y, Zhang ZS (2011) New insight into the antifibrotic effects of praziquantel on mice in infection with Schistosoma japonicum. PLoS One 6:e20247
Liu C-x (2017) Discovery and development of artemisinin and related compounds. Chinese Herbal Medicines 9:101–114
Liu L, Zuo LF, Zuo J, Wang J (2015) Artesunate induces apoptosis and inhibits growth of Eca109 and Ec9706 human esophageal cancer cell lines in vitro and in vivo. Mol Med Rep 12:1465–1472
Liu M, Shi P, Fu X, Brodelius PE, Shen Q, Jiang W, He Q, Tang K (2016) Characterization of a trichome-specific promoter of the aldehyde dehydrogenase 1 (ALDH1) gene in Artemisia annua. Plant Cell, Tissue and Organ Culture (PCTOC) 126:469–480
Liu L-x, Li-li J, Qiong C, Xiao-lin F (2017) Recent advances in the synthesis of Antischistosomal drugs and. Agents Mini reviews in medicinal chemistry 17:467–484
Liu R, Miller PA, Vakulenko SB, Stewart NK, Boggess WC, Miller MJ (2018) A synthetic dual drug Sideromycin induces gram-negative Bacteria to commit suicide with a gram-positive antibiotic. J Med Chem 61:3845–3854
Loop T, Pahl H (2003) Activators and target genes of Rel/NF-кB transcription factors. In: Nuclear factor кB. Springer, pp 1–48
Lovis L, Mak TK, Phongluxa K, Ayé Soukhathammavong P, Vonghachack Y, Keiser J, Vounatsou P, Tanner M, Hatz C, Utzinger J, Odermatt P, Akkhavong K (2012) Efficacy of praziquantel against Schistosoma mekongi and Opisthorchis viverrini: a randomized, single-blinded dose-comparison trial. PLoS Negl Trop Dis 6:e1726
Luo J, Zhu W, Tang Y, Cao H, Zhou Y, Ji R, Zhou X, Lu Z, Yang H, Zhang S, Cao J (2014) Artemisinin derivative artesunate induces radiosensitivity in cervical cancer cells in vitro and in vivo. Radiat Oncol 9:84
Makarov V et al. (2009) Benzothiazinones kill Mycobacterium tuberculosis by blocking arabinan synthesis science
Mandara CI et al (2018) High efficacy of artemether–lumefantrine and dihydroartemisinin–piperaquine for the treatment of uncomplicated falciparum malaria in Muheza and Kigoma districts. Tanzania Malaria journal 17:261
Maxmen A (2016) Busting the billion-dollar myth: how to slash the cost of drug development. Nature 536:388–390
McCoy A, Lissenden N, Morton A, Worrall E (2017) Towards an economics policy framework to combat malaria, in an era of insecticide resistance
Meskill JMM (2017) A Chinese Pioneer family: the Lins of Wu-feng, Taiwan, 1729–1895 vol 4941. Princeton University Press,
Miller LH, Su X (2011) Artemisinin: discovery from the Chinese herbal garden. Cell 146:855–858
Miller MJ, Walz AJ, Zhu H, Wu C, Moraski G, Möllmann U, Tristani EM, Crumbliss AL, Ferdig MT, Checkley L, Edwards RL, Boshoff HI (2011) Design, synthesis, and study of a mycobactin− artemisinin conjugate that has selective and potent activity against tuberculosis and malaria. J Am Chem Soc 133:2076–2079
Morere L et al (2015) Ex vivo model of congenital cytomegalovirus infection and new combination therapies. Placenta 36:41–47
Morris CA, Duparc S, Borghini-Fuhrer I, Jung D, Shin C-S, Fleckenstein L (2011) Review of the clinical pharmacokinetics of artesunate and its active metabolite dihydroartemisinin following intravenous, intramuscular, oral or rectal administration. Malar J 10:263
Morrissey C, Gallis B, Solazzi JW, Kim BJ, Gulati R, Vakar-Lopez F, Goodlett DR, Vessella RL, Sasaki T (2010) Effect of artemisinin derivatives on apoptosis and cell cycle in prostate cancer cells. Anti-Cancer Drugs 21:423–432
Nakase I, Lai H, Singh NP, Sasaki T (2008) Anticancer properties of artemisinin derivatives and their targeted delivery by transferrin conjugation. Int J Pharm 354:28–33
Nakase I, Gallis B, Takatani-Nakase T, Oh S, Lacoste E, Singh NP, Goodlett DR, Tanaka S, Futaki S, Lai H, Sasaki T (2009) Transferrin receptor-dependent cytotoxicity of artemisinin–transferrin conjugates on prostate cancer cells and induction of apoptosis. Cancer Lett 274:290–298
Nondo RSO, Moshi MJ, Erasto P, Masimba PJ, Machumi F, Kidukuli AW, Heydenreich M, Zofou D (2017) Anti-plasmodial activity of Norcaesalpin D and extracts of four medicinal plants used traditionally for treatment of malaria. BMC Complement Altern Med 17:167
Olofsson L, Engström A, Lundgren A, Brodelius PE (2011) Relative expression of genes of terpene metabolism in different tissues of Artemisia annua L. BMC Plant Biol 11:45
Organization WH (2011) Global tuberculosis control: WHO report 2011
Organization WH (2016) World health statistics 2016: monitoring health for the SDGs sustainable development goals. World Health Organization
Pagan JD, Kitaoka M, Anthony RM (2018) Engineered sialylation of pathogenic antibodies in vivo attenuates autoimmune disease. Cell 172:564–577 e513
Park J, Lai HC, Singh M, Sasaki T, Singh NP (2014) Development of a dihydroartemisinin-resistant Molt-4 leukemia cell line. Anticancer Res 34:2807–2810
Pérez dVL, Burguillo F, Lopez-Aban J, Muro A (2012) Systematic review and meta-analysis of artemisinin based therapies for the treatment and prevention of schistosomiasis
Poespoprodjo JR, Fobia W, Kenangalem E, Lampah DA, Sugiarto P, Tjitra E, Anstey NM, Price RN (2014) Dihydroartemisinin-piperaquine treatment of multidrug resistant falciparum and vivax malaria in pregnancy. PLoS One 9:e84976
Poravuth Y, Socheat D, Rueangweerayut R, Uthaisin C, Pyae Phyo A, Valecha N, Rao BHK, Tjitra E, Purnama A, Borghini-Fuhrer I, Duparc S, Shin CS, Fleckenstein L (2011) Pyronaridine-artesunate versus chloroquine in patients with acute Plasmodium vivax malaria: a randomized, double-blind, non-inferiority trial. PLoS One 6:e14501
Premji ZG (2009) Coartem®: the journey to the clinic Malaria journal, vol 8, p S3
Qi F et al (2013) Traditional Chinese medicine and related active compounds: a review of their role on hepatitis B virus infection. Drug discoveries & therapeutics 7:212–224
Ramacher M, Umansky V, Efferth T (2009) Effect of artesunate on immune cells in ret-transgenic mouse melanoma model. Anti-Cancer Drugs 20:910–917
Rojas Vargas JA, López AG, Froeyen M (2016) Molecular Docking Studies of 1, 2, 4, 5-tetrasubstituted Imidazoles with Different Protein Targets of Mycobacterium tuberculosis Biomirror 7
Rosales-Mendoza S, Nieto-Gómez R, Angulo C (2017) A perspective on the development of plant-made vaccines in the fight against Ebola virus. Front Immunol 8:252
Ross AG, Chau TN, Inobaya MT, Olveda RM, Li Y, Harn DA (2017) A new global strategy for the elimination of schistosomiasis. In: A new global strategy for the elimination of schistosomiasis. Elsevier
Sayce AC, Alonzi DS, Killingbeck SS, Tyrrell BE, Hill ML, Caputo AT, Iwaki R, Kinami K, Ide D, Kiappes JL, Beatty PR, Kato A, Harris E, Dwek RA, Miller JL, Zitzmann N (2016) Iminosugars inhibit dengue virus production via inhibition of ER alpha-glucosidases—not glycolipid processing enzymes. PLoS Negl Trop Dis 10:e0004524
Schnepf N, Corvo J, Sanson-Le Pors M-J, Mazeron M-C (2011) Antiviral activity of ganciclovir and artesunate towards human cytomegalovirus in astrocytoma cells. Antivir Res 89:186–188
Schreiber A, Härter G, Schubert A, Bunjes D, Mertens T, Michel D (2009) Antiviral treatment of cytomegalovirus infection and resistant strains. Expert Opin Pharmacother 10:191–209
Schwartz DM, Bonelli M, Gadina M, O'shea JJ (2016) Type I/II cytokines, JAKs, and new strategies for treating autoimmune diseases. Nat Rev Rheumatol 12:25–36
Shayo A, Buza J, Ishengoma DS (2015) Monitoring of efficacy and safety of artemisinin-based anti-malarials for treatment of uncomplicated malaria: a review of evidence of implementation of anti-malarial therapeutic efficacy trials in Tanzania. Malar J 14:135
Shi JQ et al. (2013) Antimalarial Drug Artemisinin Extenuates Amyloidogenesis and Neuroinflammation in APP swe/PS 1dE9 Transgenic Mice via Inhibition of Nuclear Factor-κ B and NLRP 3 Inflammasome Activation CNS neuroscience & therapeutics 19:262–268
Shuhua X, Tanner M, N’Goran EK, Utzinger J, Chollet J, Bergquist R, Minggang C, Jiang Z (2002) Recent investigations of artemether, a novel agent for the prevention of schistosomiasis japonica, mansoni and haematobia. Acta Trop 82:175–181
Simonsen HT, Weitzel C, Christensen SB (2013) Guaianolide sesquiterpenoids: pharmacology and biosynthesis. In: Natural products. Springer, pp 3069–3098
Sinka ME, Bangs MJ, Manguin S, Rubio-Palis Y, Chareonviriyaphap T, Coetzee M, Mbogo CM, Hemingway J, Patil AP, Temperley WH, Gething PW, Kabaria CW, Burkot TR, Harbach RE, Hay SI (2012) A global map of dominant malaria vectors. Parasit Vectors 5:69
Skrahina A et al (2011) Alarming levels of drug-resistant tuberculosis in Belarus: results of a survey in Minsk. Eur Respir J:erj01454–erj02011
Stebbins RC, Emch M, Meshnick SR (2018) The effectiveness of community bed net use on Malaria Parasitemia among children less than 5 years old in Liberia
Tambo E, Ai L, Zhou X, Chen JH, Hu W, Bergquist R, Guo JG, Utzinger J, Tanner M, Zhou XN (2014) Surveillance-response systems: the key to elimination of tropical diseases. Infectious diseases of poverty 3:17
Teoh KH, Polichuk DR, Reed DW, Covello PS (2009) Molecular cloning of an aldehyde dehydrogenase implicated in artemisinin biosynthesis in Artemisia annua. Botany 87:635–642
Thanaketpaisarn O, Waiwut P, Sakurai H, Saiki I (2011) Artesunate enhances TRAIL-induced apoptosis in human cervical carcinoma cells through inhibition of the NF-κB and PI3K/Akt signaling pathways. Int J Oncol 39:279–285
Tilaoui M, Mouse HA, Jaafari A, Zyad A (2014) Differential effect of artemisinin against cancer cell lines. Natural products and bioprospecting 4:189–196
Tu Y (2011) The discovery of artemisinin (qinghaosu) and gifts from Chinese medicine. Nat Med 17:1217–1220
Tu Y (2016) Artemisinin—a gift from traditional Chinese medicine to the world (nobel lecture). Angew Chem Int Ed 55:10210–10226
Utzinger J et al (2009) Schistosomiasis and neglected tropical diseases: towards integrated and sustainable control and a word of caution. Parasitology 136:1859–1874
Vale N, Gouveia MJ, Rinaldi G, Brindley PJ, Gärtner F, da Costa JMC (2017) Praziquantel for schistosomiasis, single drug revisited metabolism, mode of action and resistance antimicrobial agents and chemotherapy:AAC, pp 02582–02516
Van Huijsduijnen RH et al (2013) Anticancer properties of distinct antimalarial drug classes. PLoS One 8:e82962
Wang JX et al (2008) The new water-soluble artemisinin derivative SM905 ameliorates collagen-induced arthritis by suppression of inflammatory and Th17 responses. Br J Pharmacol 153:1303–1310
Wang J-x, L-f H, YAng Y, TAng W, Li Y, J-p Z (2009) SM905, an artemisinin derivative, inhibited NO and pro-inflammatory cytokine production by suppressing MAPK and NF-κB pathways in RAW 264.7 macrophages. Acta Pharmacol Sin 30:1428
Wang X-Q, Liu HL, Wang GB, Wu PF, Yan T, Xie J, Tang Y, Sun LK, Li C (2011a) Effect of artesunate on endotoxin-induced uveitis in rats. Invest Ophthalmol Vis Sci 52:916–919
Wang Y, Yang K, Jing F, Li M, Deng T, Huang R, Wang B, Wang G, Sun X, Tang KX (2011b) Cloning and characterization of trichome-specific promoter of cpr71av1 gene involved in artemisinin biosynthesis in Artemisia annua L. Mol Biol 45:751–758
Wang J, Zhang CJ, Chia WN, Loh CCY, Li Z, Lee YM, He Y, Yuan LX, Lim TK, Liu M, Liew CX, Lee YQ, Zhang J, Lu N, Lim CT, Hua ZC, Liu B, Shen HM, Tan KSW, Lin Q (2015) Haem-activated promiscuous targeting of artemisinin in Plasmodium falciparum. Nat Commun 6:10111
Wang B, Kashkooli AB, Sallets A, Ting HM, de Ruijter NCA, Olofsson L, Brodelius P, Pottier M, Boutry M, Bouwmeester H, van der Krol AR (2016) Transient production of artemisinin in Nicotiana benthamiana is boosted by a specific lipid transfer protein from a. annua. Metab Eng 38:159–169
Wang J, Xu C, Lun Z-R, Meshnick SR (2017a) Unpacking ‘artemisinin resistance. Trends Pharmacol Sci 38:506–511
Wang J, Zhang J, Shi Y, Xu C, Zhang C, Wong YK, Lee YM, Krishna S, He Y, Lim TK, Sim W, Hua ZC, Shen HM, Lin Q (2017b) Mechanistic investigation of the specific anticancer property of artemisinin and its combination with aminolevulinic acid for enhanced anticolorectal cancer activity. ACS central science 3:743–750
Wei J, Zhang L, Ren L, Zhang J, Liu J, Duan J, Yu Y, Li Y, Peng C, Zhou X, Sun Z (2017) Endosulfan induces cell dysfunction through cycle arrest resulting from DNA damage and DNA damage response signaling pathways. Sci Total Environ 589:97–106
Weifeng T, Feng S, Xiangji L, Changqing S, Zhiquan Q, Huazhong Z, Peining Y, Yong Y, Mengchao W, Xiaoqing J, Wan-yee L (2011) Artemisinin inhibits in vitro and in vivo invasion and metastasis of human hepatocellular carcinoma cells. Phytomedicine 18:158–162
Wen W, Yu R (2011) Artemisinin biosynthesis and its regulatory enzymes: Progress and perspective. Pharmacogn Rev 5:189
Wencewicz TA, Miller MJ (2017) Sideromycins as pathogen-targeted antibiotics
White N, Pukrittayakamee S, Hien T, Faiz M, O a M, Dondorp AM (2013) Malaria Lancet 6736:13
White N, Pukrittayakamee S, Hien T, Faiz M, Mokuolu O, Malaria DA (2014) Malaria. Lancet [Internet] 383(9918):723–735
Wilson ME, Kantele A, Jokiranta TS (2011) Review of cases with the emerging fifth human malaria parasite. Plasmodium knowlesi Clinical infectious diseases 52:1356–1362
Wohlfarth C, Efferth T (2009) Natural products as promising drug candidates for the treatment of hepatitis B and C. Acta Pharmacol Sin 30:25–30
Wong YK, Xu C, Kalesh KA, He Y, Lin Q, Wong WSF, Shen HM, Wang J (2017) Artemisinin as an anticancer drug: recent advances in target profiling and mechanisms of action. Med Res Rev 37:1492–1517
Wu X, Zhang W, Shi X, An P, Sun W, Wang Z (2010) Therapeutic effect of artemisinin on lupus nephritis mice and its mechanisms. Acta Biochim Biophys Sin 42:916–923
Wu H, Hu B, Zhou X, Zhou C, Meng J, Yang Y, Zhao X, Shi Z, Yan S (2018) Artemether attenuates LPS-induced inflammatory bone loss by inhibiting osteoclastogenesis and bone resorption via suppression of MAPK signaling pathway. Cell Death Dis 9
Yakasai AM et al. (2015) Adherence to artemisinin-based combination therapy for the treatment of uncomplicated malaria: a systematic review and meta-analysis Journal of tropical medicine 2015
YANG L, ZHANG D (2017) Summary of dihydroartemisinin and its application for the treatment of lupus erythematosus. Chin Sci Bull 62:2007–2012
Yang Z, Ding J, Yang C, Gao Y, Li X, Chen X, Peng Y, Fang J, Xiao S (2012) Immunomodulatory and anti-inflammatory properties of artesunate in experimental colitis. Curr Med Chem 19:4541–4551
Yi P, Park J-S, Melton DA, Huang KC (2017) NF-κB was discovered 30 years ago as a rapidly inducible transcription factor. Since that time, it has been found to have a broad role in gene induction in diverse cellular responses, particularly throughout the immune system. Here, we summarize elaborate regulatory pathways involving this transcription factor and use recent discoveries in human genetic diseases to place specific proteins within their. Cell 168:1–2
Zhang CJ, Wang J, Zhang J, Lee YM, Feng G, Lim TK, Shen HM, Lin Q, Liu B (2016) Mechanism-guided design and synthesis of a mitochondria-targeting artemisinin analogue with enhanced anticancer activity. Angew Chem Int Ed 55:13770–13774
Zhang S, Shi L, Ma H, Li H, Li Y, Lu Y, Wang Q, Li W (2017) Dihydroartemisinin induces apoptosis in human gastric cancer cell line BGC-823 through activation of JNK1/2 and p38 MAPK signaling pathways. Journal of Receptors and Signal Transduction 37:174–180
Zhang J, Sun X, Wang L, Wong YK, Lee YM, Zhou C, Wu G, Zhao T, Yang L, Lu L, Zhong J, Huang D, Wang J (2018) Artesunate-induced mitophagy alters cellular redox status. Redox Biol 19:263–273
Zheng H, Colvin CJ, Johnson BK, Kirchhoff PD, Wilson M, Jorgensen-Muga K, Larsen SD, Abramovitch RB (2017) Inhibitors of mycobacterium tuberculosis DosRST signaling and persistence. Nat Chem Biol 13:218–225
Zhou H-J, Zhang J-L, Li A, Wang Z, Lou X-E (2010) Dihydroartemisinin improves the efficiency of chemotherapeutics in lung carcinomas in vivo and inhibits murine Lewis lung carcinoma cell line growth in vitro. Cancer Chemother Pharmacol 66:21–29
Acknowledgements
The authors are thankful to the Shanghai Jiao Tong University, Shanghai 200240, China, for providing literature facilities. We are also thankful to the anonymous reviewers for their valuable comments. We would also like to thank Dr. Shujaul Mulk Khan of Quaid-i-Azam University Islamabad, Pakistan for his guidance and checking plagiarism in the first draft of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that there is no conflict of interest to claim.
Additional information
Communicated by: Yuval Cohen
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saeed-ur-Rahman, Khalid, M., Kayani, SI. et al. Biological Activities of Artemisinins Beyond Anti-Malarial: a Review. Tropical Plant Biol. 12, 231–243 (2019). https://doi.org/10.1007/s12042-019-09228-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12042-019-09228-0