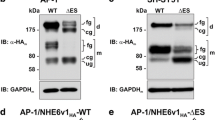
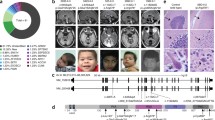
Abstract
Vesicular chloride/proton exchangers of the CLC family are critically involved in the function of the endosomal-lysosomal pathway. Their dysfunction leads to severe disorders including intellectual disability and epilepsy for ClC-4, Dent’s disease for ClC-5, and lysosomal storage disease and osteopetrosis for ClC-7. Here, we report a de novo variant p.Glu200Ala (p.E200A; c.599A>C) of the late endosomal ClC-6, encoded by CLCN6, in a patient with West syndrome (WS), severe developmental delay, autism, movement disorder, microcephaly, facial dysmorphism, and visual impairment. Mutation of this conserved glutamate uncouples chloride transport from proton antiport by ClC-6. This affects organellar ion homeostasis and was shown to be deleterious for other CLCs. In this study, we found that upon heterologous expression, the ClC-6 E200A variant caused autophagosome accumulation and impaired the clearance of autophagosomes by blocking autophagosome-lysosome fusion. Our study provides clinical and functional support for an association between CLCN6 variants and WS. Our findings also provide novel insights into the molecular mechanisms underlying the pathogenesis of WS, suggesting an involvement of autophagic-lysosomal dysfunction.





Similar content being viewed by others
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Wheless JW, Gibson PA, Rosbeck KL, Hardin M, O’Dell C, Whittemore V, Pellock JM (2012) Infantile spasms (West syndrome): update and resources for pediatricians and providers to share with parents. BMC Pediatr 12:108. https://doi.org/10.1186/1471-2431-12-108
McTague A, Howell KB, Cross JH, Kurian MA, Scheffer IE (2016) The genetic landscape of the epileptic encephalopathies of infancy and childhood. Lancet Neurol 15:304–316. https://doi.org/10.1016/S1474-4422(15)00250-1
Shbarou R, Mikati MA (2016) The expanding clinical spectrum of genetic pediatric epileptic encephalopathies. Semin Pediatr Neurol 23:134–142. https://doi.org/10.1016/j.spen.2016.06.002
Boutry-Kryza N, Labalme A, Ville D, de Bellescize J, Touraine R, Prieur F, Dimassi S, Poulat AL et al (2015) Molecular characterization of a cohort of 73 patients with infantile spasms syndrome. Eur J Med Genet 58:51–58. https://doi.org/10.1016/j.ejmg.2014.11.007
Holland KD, Hallinan BE (2010) What causes epileptic encephalopathy in infancy?: the answer may lie in our genes. Neurology 75:1132–1133. https://doi.org/10.1212/WNL.0b013e3181f6bc97
Olivetti PR, Noebels JL (2012) Interneuron, interrupted: molecular pathogenesis of ARX mutations and X-linked infantile spasms. Curr Opin Neurobiol 22:859–865. https://doi.org/10.1016/j.conb.2012.04.006
Jentsch TJ, Pusch M (2018) CLC chloride channels and transporters: structure, function, physiology, and disease. Physiol Rev 98:1493–1590. https://doi.org/10.1152/physrev.00047.2017
Neagoe I, Stauber T, Fidzinski P, Bergsdorf EY, Jentsch TJ (2010) The late endosomal ClC-6 mediates proton/chloride countertransport in heterologous plasma membrane expression. J Biol Chem 285:21689–21697. https://doi.org/10.1074/jbc.M110.125971
Poet M, Kornak U, Schweizer M, Zdebik AA, Scheel O, Hoelter S, Wurst W, Schmitt A et al (2006) Lysosomal storage disease upon disruption of the neuronal chloride transport protein ClC-6. Proc Natl Acad Sci U S A 103:13854–13859. https://doi.org/10.1073/pnas.0606137103
Stauber T, Jentsch TJ (2013) Chloride in vesicular trafficking and function. Annu Rev Physiol 75:453–477. https://doi.org/10.1146/annurev-physiol-030212-183702
Chen TT, Klassen TL, Goldman AM, Marini C, Guerrini R, Noebels JL (2013) Novel brain expression of ClC-1 chloride channels and enrichment of CLCN1 variants in epilepsy. Neurology 80:1078–1085. https://doi.org/10.1212/WNL.0b013e31828868e7
D'Agostino D, Bertelli M, Gallo S, Cecchin S, Albiero E, Garofalo PG, Gambardella A, St Hilaire JM et al (2004) Mutations and polymorphisms of the CLCN2 gene in idiopathic epilepsy. Neurology 63:1500–1502. https://doi.org/10.1212/01.wnl.0000142093.94998.1a
Kleefuss-Lie A, Friedl W, Cichon S, Haug K, Warnstedt M, Alekov A, Sander T, Ramirez A et al (2009) CLCN2 variants in idiopathic generalized epilepsy. Nat Genet 41:954–955. https://doi.org/10.1038/ng0909-954
Palmer EE, Stuhlmann T, Weinert S, Haan E, Van Esch H, Holvoet M, Boyle J, Leffler M et al (2018) De novo and inherited mutations in the X-linked gene CLCN4 are associated with syndromic intellectual disability and behavior and seizure disorders in males and females. Mol Psychiatry 23:222–230. https://doi.org/10.1038/mp.2016.135
Veeramah KR, Johnstone L, Karafet TM, Wolf D, Sprissler R, Salogiannis J, Barth-Maron A, Greenberg ME et al (2013) Exome sequencing reveals new causal mutations in children with epileptic encephalopathies. Epilepsia 54:1270–1281. https://doi.org/10.1111/epi.12201
Peng J, Wang Y, He F, Chen C, Wu LW, Yang LF, Ma YP, Zhang W et al (2018) Novel West syndrome candidate genes in a Chinese cohort. CNS Neurosci Ther 24:1196–1206. https://doi.org/10.1111/cns.12860
Wang Y, Du X, Bin R, Yu S, Xia Z, Zheng G, Zhong J, Zhang Y et al (2017) Genetic variants identified from epilepsy of unknown etiology in Chinese children by targeted exome sequencing. Sci Rep 7:40319. https://doi.org/10.1038/srep40319
Yamamoto T, Shimojima K, Sangu N, Komoike Y, Ishii A, Abe S, Yamashita S, Imai K et al (2015) Single nucleotide variations in CLCN6 identified in patients with benign partial epilepsies in infancy and/or febrile seizures. PLoS One 10:e0118946. https://doi.org/10.1371/journal.pone.0118946
Yu B, Pulit SL, Hwang SJ, Brody JA, Amin N, Auer PL, Bis JC, Boerwinkle E et al (2016) Rare exome sequence variants in CLCN6 reduce blood pressure levels and hypertension risk. Circ Cardiovasc Genet 9:64–70. https://doi.org/10.1161/CIRCGENETICS.115.001215
Zhang L, Zhang T, Xiang Z, Lu S (2015) The rs3737964 single-nucleotide polymorphism of the chloride channel-6 gene as a risk factor for coronary heart disease. Mol Genet Genomic Med 3:537–542. https://doi.org/10.1002/mgg3.163
Genomes Project C, Auton A, Brooks LD, Durbin RM, Garrison EP, Kang HM, Korbel JO, Marchini JL et al (2015) A global reference for human genetic variation. Nature 526:68–74. https://doi.org/10.1038/nature15393
Lek M, Karczewski KJ, Minikel EV, Samocha KE, Banks E, Fennell T, O’Donnell-Luria AH, Ware JS et al (2016) Analysis of protein-coding genetic variation in 60,706 humans. Nature 536:285–291. https://doi.org/10.1038/nature19057
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, Hegde M et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–424. https://doi.org/10.1038/gim.2015.30
Kimura S, Noda T, Yoshimori T (2007) Dissection of the autophagosome maturation process by a novel reporter protein, tandem fluorescent-tagged LC3. Autophagy 3:452–460. https://doi.org/10.4161/auto.4451
Ignoul S, Simaels J, Hermans D, Annaert W, Eggermont J (2007) Human ClC-6 is a late endosomal glycoprotein that associates with detergent-resistant lipid domains. PLoS One 2:e474. https://doi.org/10.1371/journal.pone.0000474
Stauber T, Jentsch TJ (2010) Sorting motifs of the endosomal/lysosomal CLC chloride transporters. J Biol Chem 285:34537–34548. https://doi.org/10.1074/jbc.M110.162545
Wartosch L, Fuhrmann JC, Schweizer M, Stauber T, Jentsch TJ (2009) Lysosomal degradation of endocytosed proteins depends on the chloride transport protein ClC-7. FASEB J 23:4056–4068. https://doi.org/10.1096/fj.09-130880
Kang R, Zeh HJ, Lotze MT, Tang D (2011) The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 18:571–580. https://doi.org/10.1038/cdd.2010.191
Pankiv S, Clausen TH, Lamark T, Brech A, Bruun JA, Outzen H, Overvatn A, Bjorkoy G et al (2007) p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J Biol Chem 282:24131–24145. https://doi.org/10.1074/jbc.M702824200
Niemeyer MI, Cid LP, Sepulveda FV, Blanz J, Auberson M, Jentsch TJ (2010) No evidence for a role of CLCN2 variants in idiopathic generalized epilepsy. Nat Genet 42:3. https://doi.org/10.1038/ng0110-3
Feng Y, He D, Yao Z, Klionsky DJ (2014) The machinery of macroautophagy. Cell Res 24:24–41. https://doi.org/10.1038/cr.2013.168
Saha S, Panigrahi DP, Patil S, Bhutia SK (2018) Autophagy in health and disease: a comprehensive review. Biomed Pharmacother 104:485–495. https://doi.org/10.1016/j.biopha.2018.05.007
Rubinsztein DC, Codogno P, Levine B (2012) Autophagy modulation as a potential therapeutic target for diverse diseases. Nat Rev Drug Discov 11:709–730. https://doi.org/10.1038/nrd3802
Hara T, Nakamura K, Matsui M, Yamamoto A, Nakahara Y, Suzuki-Migishima R, Yokoyama M, Mishima K et al (2006) Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441:885–889. https://doi.org/10.1038/nature04724
Komatsu M, Waguri S, Chiba T, Murata S, Iwata J, Tanida I, Ueno T, Koike M et al (2006) Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441:880–884. https://doi.org/10.1038/nature04723
Shen W, Ganetzky B (2009) Autophagy promotes synapse development in Drosophila. J Cell Biol 187:71–79. https://doi.org/10.1083/jcb.200907109
Chen H, Qian Y, Yu S, Xiao D, Guo X, Wang Q, Hao L, Yan K et al (2019) Early onset developmental delay and epilepsy in pediatric patients with WDR45 variants. Eur J Med Genet 62:149–160. https://doi.org/10.1016/j.ejmg.2018.07.002
Giorgi FS, Biagioni F, Lenzi P, Frati A, Fornai F (2015) The role of autophagy in epileptogenesis and in epilepsy-induced neuronal alterations. J Neural Transm (Vienna) 122:849–862. https://doi.org/10.1007/s00702-014-1312-1
Li Q, Han Y, Du J, Jin H, Zhang J, Niu M, Qin J (2018) Alterations of apoptosis and autophagy in developing brain of rats with epilepsy: changes in LC3, P62, Beclin-1 and Bcl-2 levels. Neurosci Res 130:47–55. https://doi.org/10.1016/j.neures.2017.08.004
Nixon RA (2017) Amyloid precursor protein and endosomal-lysosomal dysfunction in Alzheimer’s disease: inseparable partners in a multifactorial disease. FASEB J 31:2729–2743. https://doi.org/10.1096/fj.201700359
Accardi A, Miller C (2004) Secondary active transport mediated by a prokaryotic homologue of ClC Cl- channels. Nature 427:803–807. https://doi.org/10.1038/nature02314
Picollo A, Pusch M (2005) Chloride/proton antiporter activity of mammalian CLC proteins ClC-4 and ClC-5. Nature 436:420–423. https://doi.org/10.1038/nature03720
Scheel O, Zdebik AA, Lourdel S, Jentsch TJ (2005) Voltage-dependent electrogenic chloride/proton exchange by endosomal CLC proteins. Nature 436:424–427. https://doi.org/10.1038/nature03860
Leisle L, Ludwig CF, Wagner FA, Jentsch TJ, Stauber T (2011) ClC-7 is a slowly voltage-gated 2Cl(-)/1H(+)-exchanger and requires Ostm1 for transport activity. EMBO J 30:2140–2152. https://doi.org/10.1038/emboj.2011.137
Nicoli ER, Weston MR, Hackbarth M, Becerril A, Larson A, Zein WM, Baker PR 2nd, Burke JD et al (2019) Lysosomal storage and albinism due to effects of a de novo CLCN7 variant on lysosomal acidification. Am J Hum Genet 104:1127–1138. https://doi.org/10.1016/j.ajhg.2019.04.008
Sartelet A, Stauber T, Coppieters W, Ludwig CF, Fasquelle C, Druet T, Zhang Z, Ahariz N et al (2014) A missense mutation accelerating the gating of the lysosomal Cl-/H+-exchanger ClC-7/Ostm1 causes osteopetrosis with gingival hamartomas in cattle. Dis Model Mech 7:119–128. https://doi.org/10.1242/dmm.012500
Novarino G, Weinert S, Rickheit G, Jentsch TJ (2010) Endosomal chloride-proton exchange rather than chloride conductance is crucial for renal endocytosis. Science 328:1398–1401. https://doi.org/10.1126/science.1188070
Weinert S, Gimber N, Deuschel D, Stuhlmann T, Puchkov D, Farsi Z, Ludwig CF, Novarino G et al (2020) Uncoupling endosomal CLC chloride/proton exchange causes severe neurodegeneration. EMBO J 39:e103358. https://doi.org/10.15252/embj.2019103358
Weinert S, Jabs S, Supanchart C, Schweizer M, Gimber N, Richter M, Rademann J, Stauber T et al (2010) Lysosomal pathology and osteopetrosis upon loss of H+-driven lysosomal Cl- accumulation. Science 328:1401–1403. https://doi.org/10.1126/science.1188072
Satoh N, Yamada H, Yamazaki O, Suzuki M, Nakamura M, Suzuki A, Ashida A, Yamamoto D et al (2016) A pure chloride channel mutant of CLC-5 causes Dent’s disease via insufficient V-ATPase activation. Pflugers Arch 468:1183–1196. https://doi.org/10.1007/s00424-016-1808-7
Sekine T, Komoda F, Miura K, Takita J, Shimadzu M, Matsuyama T, Ashida A, Igarashi T (2014) Japanese Dent disease has a wider clinical spectrum than Dent disease in Europe/USA: genetic and clinical studies of 86 unrelated patients with low-molecular-weight proteinuria. Nephrol Dial Transplant 29:376–384. https://doi.org/10.1093/ndt/gft394
Acknowledgements
We are grateful to the patient and his parents for their participation in this study. We thank Prof. Jiada Li for providing cell lines and Dengfeng Li for technical assistance.
Funding
This research was funded by the National Natural Science Foundation of China (grant numbers 81771409 and 81771408), the National Key Research and Development Program of China (grant numbers 2016YFC1306202 and 2016YFC0904400), the German Research Foundation (DFG, within FOR 2625), and by the China Scholarship Council fellowship.
Author information
Authors and Affiliations
Contributions
Conceptualization: H.H. and J.P.; investigation: H.H., X.C., F.Y., and T.W; resources: J.P.; writing—original draft preparation: H.H.; writing—review and editing: T.S. and J.P.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
This study was approved by the Ethics Committee of Xiangya Hospital of Central South University, China (Human study/protocol #201603205) and performed in accordance with the ethical standards laid down in the Declaration of Helsinki. Written informed consent was obtained for the patient from his parents.
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
He, H., Cao, X., Yin, F. et al. West Syndrome Caused By a Chloride/Proton Exchange-Uncoupling CLCN6 Mutation Related to Autophagic-Lysosomal Dysfunction. Mol Neurobiol 58, 2990–2999 (2021). https://doi.org/10.1007/s12035-021-02291-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-021-02291-3