Abstract
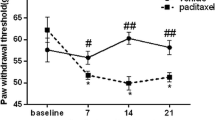
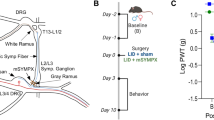
Chemotherapy-induced peripheral neuropathy (CIPN) adversely impacts quality of life and a challenge to treat with existing drugs used for neuropathic pain. Losartan, an angiotensin II type 1 receptor (AT1R) antagonist widely used to treat hypertension, has been reported to have analgesic effects in several pain models. In this study, we assessed losartan’s analgesic effect on paclitaxel-induced neuropathic pain (PINP) in rats and its mechanism of action in dorsal root ganglion (DRG). Rats received intraperitoneal injections of 2 mg/kg paclitaxel on days 0, 2, 4, and 6 and received single or multiple intraperitoneal injections of losartan potassium dissolved in phosphate-buffered saline at various times. The mechanical thresholds, protein levels of inflammatory cytokines, and cellular location of AT1R and interleukin 1β (IL-1β) in the DRG were assessed with behavioral testing, Western blotting, and immunohistochemistry, respectively. Data were analyzed by two-way repeated-measures analysis of variance for the behavioral test or the Mann-Whitney U test for the Western blot analysis and immunohistochemistry. Single and multiple injections of losartan ameliorated PINP, and losartan delayed the development of PINP. Paclitaxel significantly increased, and losartan subsequently decreased, the expression levels of inflammatory cytokines, including IL-1β and tumor necrosis factor α (TNF-α), in the lumbar DRG. AT1R and IL-1β were expressed in both neurons and satellite cells and losartan decreased the intensity of IL-1β in the DRG. Losartan ameliorates PINP by decreasing inflammatory cytokines including IL-1β and TNF-α in the DRG. Our findings provide a new or add-on therapy for CIPN patients.






Similar content being viewed by others
Abbreviations
- CINP:
-
chemotherapy-induced peripheral neuropathy
- AT1R:
-
angiotensin II type 1 receptor
- PINP:
-
paclitaxel-induced neuropathic pain
- DRG:
-
dorsal root ganglion
- IL-1β:
-
interleukin 1β
- TNF-α:
-
tumor necrosis factor α
- DMSO:
-
dimethyl sulfoxide
- PBS:
-
phosphate-buffered saline
- MCP-1:
-
monocyte chemoattractant protein 1
- GAPDH:
-
glyceraldehydes-3-phosphate dehydrogenase
- GFAP:
-
glial fibrillary acidic protein
- DAPI:
-
4′,6-diamidino-2-phenylindole
- p-NFκB:
-
phosphorylated nuclear factor kappa B
- NFκB:
-
nuclear factor kappa B
References
Seretny M, Currie GL, Sena ES, Ramnarine S, Grant R, MacLeod MR, Colvin LA, Fallon M (2014) Incidence, prevalence, and predictors of chemotherapy-induced peripheral neuropathy: a systematic review and meta-analysis. Pain 155(12):2461–2470. https://doi.org/10.1016/j.pain.2014.09.020
Yazdani S, Abdi S (2014) Brief review: pain management for cancer survivors: challenges and opportunities. Can J Anaesth 61(8):745–753. https://doi.org/10.1007/s12630-014-0170-5
Mols F, Beijers T, Vreugdenhil G, van de Poll-Franse L (2014) Chemotherapy-induced peripheral neuropathy and its association with quality of life: a systematic review. Support Care Cancer 22 (8):2261–2269. doi:https://doi.org/10.1007/s00520-014-2255-7
Vyas DM, Kadow JF (1995) Paclitaxel: a unique tubulin interacting anticancer agent. Prog Med Chem 32:289–337
Balayssac D, Ferrier J, Descoeur J, Ling B, Pezet D, Eschalier A, Authier N (2011) Chemotherapy-induced peripheral neuropathies: from clinical relevance to preclinical evidence. Expert Opin Drug Saf 10(3):407–417. https://doi.org/10.1517/14740338.2011.543417
Sisignano M, Baron R, Scholich K, Geisslinger G (2014) Mechanism-based treatment for chemotherapy-induced peripheral neuropathic pain. Nat Rev Neurol 10(12):694–707. https://doi.org/10.1038/nrneurol.2014.211
Rao RD, Michalak JC, Sloan JA, Loprinzi CL, Soori GS, Nikcevich DA, Warner DO, Novotny P et al (2007) Efficacy of gabapentin in the management of chemotherapy-induced peripheral neuropathy: a phase 3 randomized, double-blind, placebo-controlled, crossover trial (N00C3). Cancer 110(9):2110–2118. https://doi.org/10.1002/cncr.23008
Kautio AL, Haanpaa M, Leminen A, Kalso E, Kautiainen H, Saarto T (2009) Amitriptyline in the prevention of chemotherapy-induced neuropathic symptoms. Anticancer Res 29(7):2601–2606
Kobori H, Nangaku M, Navar LG, Nishiyama A (2007) The intrarenal renin-angiotensin system: from physiology to the pathobiology of hypertension and kidney disease. Pharmacol Rev 59(3):251–287
Paul M, Mehr AP, Kreutz R (2006) Physiology of local renin-angiotensin systems. Physiol Rev 86(3):747–803
Irvine RJ, White JM (1997) The effects of central and peripheral angiotensin on hypertension and nociception in rats. Pharmacol Biochem Behav 57(1–2):37–41
Nemoto W, Nakagawasai O, Yaoita F, Kanno SI, Yomogida S, Ishikawa M, Tadano T, Tan-No K (2013) Angiotensin II produces nociceptive behavior through spinal AT1 receptor-mediated p38 mitogen-activated protein kinase activation in mice. Mol Pain 9(1):1744-8069-9-38. https://doi.org/10.1186/1744-8069-9-38
Pavel J, Oroszova Z, Hricova L, Lukacova N (2013) Effect of subpressor dose of angiotensin II on pain-related behavior in relation with neuronal injury and activation of satellite glial cells in the rat dorsal root ganglia. Cell Mol Neurobiol 33(5):681–688. https://doi.org/10.1007/s10571-013-9934-7
Nemoto W, Ogata Y, Nakagawasai O, Yaoita F, Tanado T, Tan-No K (2015) The intrathecal administration of losartan, an AT1 receptor antagonist, produces an antinociceptive effect through the inhibiton of p38 MAPK phosphorylation in the mouse formalin test. Neurosci Lett 585:17–22. https://doi.org/10.1016/j.neulet.2014.11.018
Costa AC, Romero TR, Pacheco DF, Perez AC, Savernini A, Santos RR, Duarte ID (2014) Participation of AT1 and mas receptors in the modulation of inflammatory pain. Peptides 61:17–22. https://doi.org/10.1016/j.peptides.2014.08.010
Ogata Y, Nemoto W, Nakagawasai O, Yamagata R, Tadano T, Tan-No K (2016) Involvement of spinal angiotensin II system in streptozotocin-induced diabetic neuropathic pain in mice. Mol Pharmacol 90(3):205–213. https://doi.org/10.1124/mol.116.104133
Kim HK, Zhang YP, Gwak YS, Abdi S (2010) Phenyl N-tert-butylnitrone, a free radical scavenger, reduces mechanical allodynia in chemotherapy-induced neuropathic pain in rats. Anesthesiology 112(2):432–439. https://doi.org/10.1097/ALN.0b013e3181ca31bd
Chaplan SR, Bach F, Pogrel J, Chung J, Yaksh T (1994) Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 53(1):55–63
Dixon WJ (1980) Efficient analysis of experimental observations. Annu Rev Pharmacol Toxicol 20(1):441–462
Kim HK, Park SK, Zhou J-L, Taglialatela G, Chung K, Coggeshall RE, Chung JM (2004) Reactive oxygen species (ROS) play an important role in a rat model of neuropathic pain. Pain 111 (1):116–124
Kim HK, Hwang SH, Oh E, Abdi S (2017) Rolipram, a selective phosphodiesterase 4 inhibitor, ameliorates mechanical hyperalgesia in a rat model of chemotherapy-induced neuropathic pain through inhibition of inflammatory cytokines in the dorsal root ganglion. Front Pharmacol 8(885). https://doi.org/10.3389/fphar.2017.00885
Kim HK, Hwang SH, Abdi S (2016) Tempol ameliorates and prevents mechanical hyperalgesia in a rat model of chemotherapy-induced neuropathic pain. Front Pharmacol 7:532. https://doi.org/10.3389/fphar.2016.00532
Kim HK, Hwang SH, Lee SO, Kim SH, Abdi S (2016) Pentoxifylline ameliorates mechanical hyperalgesia in a rat model of chemotherapy-induced neuropathic pain. Pain Physician 19(4):E589–E600
Duraikannu A, Martinez JA, Chandrasekhar A, Zochodne DW (2018) Expression and manipulation of the APC-beta-catenin pathway during peripheral neuron regeneration. Sci Rep 8(1):13197. https://doi.org/10.1038/s41598-018-31167-1
Yu Z, Xu N, Zhang N, Xiong Y, Wang Z, Liang S, Zhao D, Huang F et al (2019) Repair of peripheral nerve sensory impairments via the transplantation of bone marrow neural tissue-committed stem cell-derived sensory neurons. Cell Mol Neurobiol 39:341–353. https://doi.org/10.1007/s10571-019-00650-2
Peach MJ (1977) Renin-angiotensin system: biochemistry and mechanisms of action. Physiol Rev 57(2):313–370
Patil J, Schwab A, Nussberger J, Schaffner T, Saavedra JM, Imboden H (2010) Intraneuronal angiotensinergic system in rat and human dorsal root ganglia. Regul Pept 162(1–3):90–98. https://doi.org/10.1016/j.regpep.2010.03.004
Oroszova Z, Hricova L, Stropkovska A, Lukacova N, Pavel J (2017) The characterization of AT1 expression in the dorsal root ganglia after chronic constriction injury. Cell Mol Neurobiol 37(3):545–554. https://doi.org/10.1007/s10571-016-0396-6
Kalynovska N, Diallo M, Palecek J (2019) Losartan treatment attenuates the development of neuropathic thermal hyperalgesia induced by peripheral nerve injury in rats. Life Sci 220:147–155. https://doi.org/10.1016/j.lfs.2019.02.008
Oono Y, Baad-Hansen L, Wang K, Arendt-Nielsen L, Svensson P (2013) Effect of conditioned pain modulation on trigeminal somatosensory function evaluated by quantitative sensory testing. Pain 154(12):2684–2690. https://doi.org/10.1016/j.pain.2013.07.049
Hou S, Huh B, Kim HK, Kim KH, Abdi S (2018) Treatment of chemotherapy-induced peripheral neuropathy: systematic review and recommendations. Pain Physician 21(6):571–592
Kim W, Chung Y, Choi S, Min BI, Kim SK (2017) Duloxetine protects against oxaliplatin-induced neuropathic pain and spinal neuron hyperexcitability in rodents. Int J Mol Sci 18(12). https://doi.org/10.3390/ijms18122626
Baumann M, Janssen BJ, Hermans JJ, Peutz-Kootstra C, Witzke O, Smits JF, Struijker Boudier HA (2007) Transient AT1 receptor-inhibition in prehypertensive spontaneously hypertensive rats results in maintained cardiac protection until advanced age. J Hypertens 25(1):207–215. https://doi.org/10.1097/HJH.0b013e3280102bff
Ellis A, Bennett DL (2013) Neuroinflammation and the generation of neuropathic pain. Br J Anaesth 111 (1):26–37. doi:https://doi.org/10.1093/bja/aet128
Ebersberger A (2018) The analgesic potential of cytokine neutralization with biologicals. Eur J Pharmacol 835:19–30. https://doi.org/10.1016/j.ejphar.2018.07.040
Kim HK, Kwon JY, Yoo C, Abdi S (2015) The analgesic effect of rolipram, a phosphodiesterase 4 inhibitor, on chemotherapy-induced neuropathic pain in rats. Anesth Analg 121(3):822–828. https://doi.org/10.1213/ANE.0000000000000853
Houslay MD, Adams DR (2003) PDE4 cAMP phosphodiesterases: modular enzymes that orchestrate signalling cross-talk, desensitization and compartmentalization. Biochem J 370(1):1–18. https://doi.org/10.1042/BJ20021698
McIntyre KW, Shuster DJ, Gillooly KM, Dambach DM, Pattoli MA, Lu P, Zhou X-D, Qiu Y et al (2003) A highly selective inhibitor of IκB kinase, BMS-345541, blocks both joint inflammation and destruction in collagen-induced arthritis in mice. Arthritis Rheum 48(9):2652–2659. https://doi.org/10.1002/art.11131
Pollock G, Pennypacker KR, Mémet S, Israël A, Saporta S (2005) Activation of NF-κB in the mouse spinal cord following sciatic nerve transection. Exp Brain Res 165(4):470–477. https://doi.org/10.1007/s00221-005-2318-6
Nonaka M, Huang ZM (1990) Interleukin-1-mediated enhancement of mouse factor B gene expression via NF kappa B-like hepatoma nuclear factor. Mol Cell Biol 10(12):6283–6289
Barger SW, Horster D, Furukawa K, Goodman Y, Krieglstein J, Mattson MP (1995) Tumor necrosis factors alpha and beta protect neurons against amyloid beta-peptide toxicity: evidence for involvement of a kappa B-binding factor and attenuation of peroxide and Ca2+ accumulation. Proc Natl Acad Sci U S A 92(20):9328–9332
Deshmane SL, Kremlev S, Amini S, Sawaya BE (2009) Monocyte chemoattractant protein-1 (MCP-1): an overview. J Interf Cytokine Res 29(6):313–326. https://doi.org/10.1089/jir.2008.0027
Mollnau H, Wendt M, Szocs K, Lassegue B, Schulz E, Oelze M, Li H, Bodenschatz M et al (2002) Effects of angiotensin II infusion on the expression and function of NAD(P)H oxidase and components of nitric oxide/cGMP signaling. Circ Res 90(4):E58–E65
Marchesi C, Paradis P, Schiffrin EL (2008) Role of the renin-angiotensin system in vascular inflammation. Trends Pharmacol Sci 29(7):367–374. https://doi.org/10.1016/j.tips.2008.05.003
Gui WS, Wei X, Mai CL, Murugan M, Wu LJ, Xin WJ, Zhou LJ, Liu XG (2016) Interleukin-1beta overproduction is a common cause for neuropathic pain, memory deficit, and depression following peripheral nerve injury in rodents. Mol Pain 12:174480691664678. https://doi.org/10.1177/1744806916646784
Stemkowski PL, Garcia-Caballero A, Gadotti VM, M'Dahoma S, Chen L, Souza IA, Zamponi GW (2017) Identification of interleukin-1 beta as a key mediator in the upregulation of Cav3.2-USP5 interactions in the pain pathway. Mol Pain 13:1744806917724698. https://doi.org/10.1177/1744806917724698
Dahlöf B, Devereux RB, Kjeldsen SE, Julius S, Beevers G, de Faire U, Fyhrquist F, Ibsen H et al (2002) Cardiovascular morbidity and mortality in the Losartan Intervention For Endpoint reduction in hypertension study (LIFE): a randomised trial against atenolol. Lancet 359(9311):995–1003. https://doi.org/10.1016/S0140-6736(02)08089-3
Boersma C, Atthobari J, Gansevoort RT, de Jong-Van den Berg LT, de Jong PE, de Zeeuw D, Annemans LJ, Postma MJ (2006) Pharmacoeconomics of angiotensin II antagonists in type 2 diabetic patients with nephropathy: implications for decision making Pharmacoeconomics 24 (6):523–535
Irvine RJ, White JM, Head RJ (1995) The renin angiotensin system and nociception in spontaneously hypertensive rats. Life Sci 56(13):1073–1078
Sitsen JMA, Wd J (1984) Observations on pain perception and hypertension in spontaneously hypertensive rats. Clin Exp Hypertens A 6(7):1345–1356. https://doi.org/10.3109/10641968409039601
Zhu H, Tan L, Li Y, Li J, Qiu M, Li L, Zhang M, Liang M et al (2017) Increased apoptosis in the paraventricular nucleus mediated by AT1R/Ras/ERK1/2 signaling results in sympathetic hyperactivity and renovascular hypertension in rats after kidney injury. Front Physiol 8:41. https://doi.org/10.3389/fphys.2017.00041
Liang S, Kisseleva T, Brenner DA (2016) The role of NADPH oxidases (NOXs) in liver fibrosis and the activation of myofibroblasts. Front Physiol 7:17. https://doi.org/10.3389/fphys.2016.00017
Acknowledgements
This work was supported by grants to S.A. from the Peggy and Avinash Ahuja Foundation and the Helen Buchanan and Stanley Joseph Seeger Endowment at The University of Texas MD Anderson Cancer Center. The authors thank Joe Munch (Department of Scientific Publications, The University of Texas MD Anderson Cancer Center) for the editorial assistance.
Author information
Authors and Affiliations
Contributions
EK: conception, design, data acquisition, analysis, interpretation, and writing of the manuscript. S-HH: design, data acquisition, analysis, and interpretation and preparation of the manuscript. H-KK: conception and interpretation, and preparation of the manuscript. SA: conception, design, interpretation, and writing of the manuscript. HKK: conception, design, data acquisition, analysis, interpretation, and writing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
The animals used in the experiment were approved by the Institutional Animal Care and Use Committee (IACUC) of The University of Texas MD Anderson Cancer Center. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, E., Hwang, SH., Kim, HK. et al. Losartan, an Angiotensin II Type 1 Receptor Antagonist, Alleviates Mechanical Hyperalgesia in a Rat Model of Chemotherapy-Induced Neuropathic Pain by Inhibiting Inflammatory Cytokines in the Dorsal Root Ganglia. Mol Neurobiol 56, 7408–7419 (2019). https://doi.org/10.1007/s12035-019-1616-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-019-1616-0