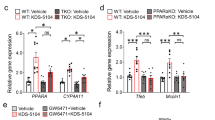
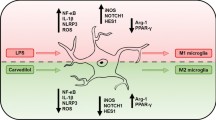
Abstract
Microglial activation is a pathological feature of many neurodegenerative diseases and the role of cellular lipids in these diseases is mostly unknown. It was known that the special ether lipid plasmalogens (Pls) were reduced in the brain and blood samples of Alzheimer’s disease (AD) patients. It has recently been reported that the oral ingestion of scallop-derived Pls (sPls) improved cognition among mild AD patients, which led us to investigate the role of sPls in the microglial activation. We used the lipopolysaccharides (LPS)-induced microglial activation model and found that sPls inhibit the LPS-mediated TLR4 endocytosis and the downstream caspases activation. By using the specific inhibitors, we also confirmed that the TLR4 endocytosis and the caspases activation strictly controlled the pro-inflammatory cytokine expression. In addition, the reduction of cellular Pls by sh-RNA-mediated knockdown of GNPAT (glyceronephosphate O-acyltransferase), a Pls synthesizing enzyme, enhanced the endocytosis of TLR4 and activation of caspase-3 which resulted in the enhanced pro-inflammatory cytokine expression. We also report for the first time that the TLR4 endocytosis was significantly higher in the cortex of aged mice and AD model mice brains, proposing a significant link between the age-related reduction of Pls and microglial activation. Interestingly, the sPls drinking in AD model mice significantly reduced the TLR4 endocytosis. Our cumulative data indicates that the cellular Pls attenuate the microglial activation by maintaining the endocytosis of TLR4, suggesting a possible mechanism of the cognition improvement effect of sPls among mild AD patients.











Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- ANOVA:
-
one-way analysis of variance
- BCA:
-
bicinchoninic acid
- BSA:
-
bovine serum albumin
- CDE:
-
clathrin-dependent endocytic
- CHO:
-
Chinese hamster ovary
- CIE:
-
clathrin-independent endocytic
- C/LR:
-
caveolin/lipid raft-mediated endocytic
- DAPI:
-
4′,6-diamidino-2-phenylindole
- DEVD:
-
Z-DEVD-fmk
- DMEM:
-
Dulbecco’s modified Eagle medium
- FBS:
-
fetal bovine serum
- GAPDH:
-
glyceraldehyde-3-phosphae dehydrogenase
- GNPAT:
-
glycerone phosphate O-acyltransferase
- HEK:
-
human embryonic kidney
- IL-1β:
-
interleukin-1β
- IL-1R:
-
interleukin-I receptor
- IRF:
-
interferon regulatory factor-3
- LPS:
-
lipopolysaccharide
- MβCD:
-
methyl-β-cyclodextrin
- NF-κB:
-
nuclear factor-κB
- PARP1:
-
polyadenosine diphosphate ribosepolymerase1
- PBS:
-
phosphate-buffered saline
- PCR:
-
polymerase chain reaction
- Pls:
-
plasmalogens
- Pls-Etn:
-
ethanolamine plasmalogens
- RCDP:
-
rhizomelic chondrodysplasia punctate
- RIPA:
-
radio-immunoprecipitation assay
- SDS-PAGE:
-
sodium dodecyl sulfate–polyacrylamide gel electrophoresis
- sh-RNA:
-
short hairpin-RNA
- TBS:
-
Tris-buffered saline
- TDU:
-
transduction unites
- TIRAP:
-
Toll/IL-1R domain-containing adaptor protein
- TLR4:
-
toll-like receptor 4
- TNF-α:
-
tumor necrosis factor-α
- TRAM:
-
Toll/interleukin-1 receptor domain-containing adaptor inducing type I interferons-related adaptor molecule
- TRIF:
-
Toll/interleukin-1 receptor domain-containing adaptor inducing type I interferons
- VAD:
-
Z-VAD-fmk
References
Tanzi RE, Bertram L (2005) Twenty years of the Alzheimer’s disease amyloid hypothesis: a genetic perspective. Cell 120(4):545–555. https://doi.org/10.1016/j.cell.2005.02.008
Haass C, Selkoe DJ (2007) Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer’s amyloid beta-peptide. Nat Rev Mol Cell Biol 8(2):101–112. https://doi.org/10.1038/nrm2101
Tahara K, Kim HD, Jin JJ, Maxwell JA, Li L, Fukuchi K (2006) Role of toll-like receptor signalling in Abeta uptake and clearance. Brain 129(Pt 11):3006–3019. https://doi.org/10.1093/brain/awl249
Apelt J, Schliebs R (2001) Beta-amyloid-induced glial expression of both pro- and anti-inflammatory cytokines in cerebral cortex of aged transgenic Tg2576 mice with Alzheimer plaque pathology. Brain Res 894(1):21–30
Salminen A, Ojala J, Suuronen T, Kaarniranta K, Kauppinen A (2008) Amyloid-beta oligomers set fire to inflammasomes and induce Alzheimer’s pathology. J Cell Mol Med 12(6A):2255–2262. https://doi.org/10.1111/j.1582-4934.2008.00496.x
Fahy E, Subramaniam S, Brown HA, Glass CK, Merrill AH Jr, Murphy RC, Raetz CR, Russell DW et al (2005) A comprehensive classification system for lipids. J Lipid Res 46(5):839–861. https://doi.org/10.1194/jlr.E400004-JLR200
Maeba R, Ueta N (2003) Ethanolamine plasmalogens prevent the oxidation of cholesterol by reducing the oxidizability of cholesterol in phospholipid bilayers. J Lipid Res 44(1):164–171
Guan Z, Wang Y, Cairns NJ, Lantos PL, Dallner G, Sindelar PJ (1999) Decrease and structural modifications of phosphatidylethanolamine plasmalogen in the brain with Alzheimer disease. J Neuropathol Exp Neurol 58(7):740–747
Han X, Holtzman DM, McKeel DW Jr (2001) Plasmalogen deficiency in early Alzheimer’s disease subjects and in animal models: molecular characterization using electrospray ionization mass spectrometry. J Neurochem 77(4):1168–1180
Hossain MS, Abe Y, Ali F, Youssef M, Honsho M, Fujiki Y, Katafuchi T (2017) Reduction of ether-type glycerophospholipids, plasmalogens, by NF-κB signal leading to microglial activation. J Neurosci 37(15):4074–4092. https://doi.org/10.1523/JNEUROSCI.3941-15.2017
Wood PL, Mankidy R, Ritchie S, Heath D, Wood JA, Flax J, Goodenowe DB (2010) Circulating plasmalogen levels and Alzheimer disease assessment scale-cognitive scores in Alzheimer patients. J Psychiatry Neurosci 35(1):59–62
Hossain MS, Ifuku M, Take S, Kawamura J, Miake K, Katafuchi T (2013) Plasmalogens rescue neuronal cell death through an activation of AKT and ERK survival signaling. PLoS One 8(12):e83508. https://doi.org/10.1371/journal.pone.0083508
Ifuku M, Katafuchi T, Mawatari S, Noda M, Miake K, Sugiyama M, Fujino T (2012) Anti-inflammatory/anti-amyloidogenic effects of plasmalogens in lipopolysaccharide-induced neuroinflammation in adult mice. J Neuroinflammation 9:197. https://doi.org/10.1186/1742-2094-9-197
Akira S, Takeda K (2004) Functions of toll-like receptors: lessons from KO mice. C R Biol 327(6):581–589
Kagan JC, Su T, Horng T, Chow A, Akira S, Medzhitov R (2008) TRAM couples endocytosis of Toll-like receptor 4 to the induction of interferon-β. Nat Immunol 9(4):361–368. https://doi.org/10.1038/ni1569
Wong SW, Kwon MJ, Choi AM, Kim HP, Nakahira K, Hwang DH (2009) Fatty acids modulate toll-like receptor 4 activation through regulation of receptor dimerization and recruitment into lipid rafts in a reactive oxygen species-dependent manner. J Biol Chem 284(40):27384–27392. https://doi.org/10.1074/jbc.M109.044065
Husebye H, Halaas O, Stenmark H, Tunheim G, Sandanger O, Bogen B, Brech A, Latz E et al (2006) Endocytic pathways regulate Toll-like receptor 4 signaling and link innate and adaptive immunity. EMBO J 25(4):683–692. https://doi.org/10.1038/sj.emboj.7600991
Shuto T, Kato K, Mori Y, Viriyakosol S, Oba M, Furuta T, Okiyoneda T, Arima H et al (2005) Membrane-anchored CD14 is required for LPS-induced TLR4 endocytosis in TLR4/MD-2/CD14 overexpressing CHO cells. Biochem Biophys Res Commun 338(3):1402–1409. https://doi.org/10.1016/j.bbrc.2005.10.102
Pascual-Lucas M, Fernandez-Lizarbe S, Montesinos J, Guerri C (2014) LPS or ethanol triggers clathrin- and rafts/caveolae-dependent endocytosis of TLR4 in cortical astrocytes. J Neurochem 129(3):448–462. https://doi.org/10.1111/jnc.12639
Cai W, Du A, Feng K, Zhao X, Qian L, Ostrom RS, Xu C (2013) Adenylyl cyclase 6 activation negatively regulates TLR4 signaling through lipid raft-mediated endocytosis. J Immunol 191 (12):6093-6100. doi:https://doi.org/10.4049/jimmunol.1301912
Budihardjo I, Oliver H, Lutter M, Luo X, Wang X (1999) Biochemical pathways of caspase activation during apoptosis. Annu Rev Cell Dev Biol 15:269–290. https://doi.org/10.1146/annurev.cellbio.15.1.269
Wu X, Guo R, Chen P, Wang Q, Cunningham PN (2009) TNF induces caspase-dependent inflammation in renal endothelial cells through a Rho- and myosin light chain kinase-dependent mechanism. Am J Physiol Renal Physiol 297(2):F316–F326. https://doi.org/10.1152/ajprenal.00089.2009
Su JH, Zhao M, Anderson AJ, Srinivasan A, Cotman CW (2001) Activated caspase-3 expression in Alzheimer’s and aged control brain: correlation with Alzheimer pathology. Brain Res 898(2):350–357
D'Amelio M, Cavallucci V, Middei S, Marchetti C, Pacioni S, Ferri A, Diamantini A, De Zio D et al (2011) Caspase-3 triggers early synaptic dysfunction in a mouse model of Alzheimer’s disease. Nat Neurosci 14(1):69–76. https://doi.org/10.1038/nn.2709
Stone JR, Okonkwo DO, Singleton RH, Mutlu LK, Helm GA, Povlishock JT (2002) Caspase-3-mediated cleavage of amyloid precursor protein and formation of amyloid β peptide in traumatic axonal injury. J Neurotrauma 19(5):601–614. https://doi.org/10.1089/089771502753754073
Burguillos MA, Deierborg T, Kavanagh E, Persson A, Hajji N, Garcia-Quintanilla A, Cano J, Brundin P et al (2011) Caspase signalling controls microglia activation and neurotoxicity. Nature 472(7343):319–324. https://doi.org/10.1038/nature09788
Hossain MS, Abe Y, Ali F, Youssef M, Honsho M, Fujiki Y, Katafuchi T (2017) Reduction of ether-type glycerophospholipids, plasmalogens, by NF-kappaB signal leading to microglial activation. J Neurosci 37(15):4074–4092. https://doi.org/10.1523/JNEUROSCI.3941-15.2017
Fujino T, Yamada T, Asada T, Tsuboi Y, Wakana C, Mawatari S, Kono S (2017) Efficacy and blood plasmalogen changes by oral administration of plasmalogen in patients with mild Alzheimer’s disease and mild cognitive impairment: a multicenter, randomized, double-blind, placebo-controlled trial. EBioMedicine 17:199–205. https://doi.org/10.1016/j.ebiom.2017.02.012
Mawatari S, Yunoki K, Sugiyama M, Fujino T (2009) Simultaneous preparation of purified plasmalogens and sphingomyelin in human erythrocytes with phospholipase A1 from Aspergillus orizae. Biosci Biotechnol Biochem 73(12):2621–2625. https://doi.org/10.1271/bbb.90455
Braverman NE, Moser AB (2012) Functions of plasmalogen lipids in health and disease. Biochim Biophys Acta 1822(9):1442–1452. https://doi.org/10.1016/j.bbadis.2012.05.008
Oettinghaus B, Schulz JM, Restelli LM, Licci M, Savoia C, Schmidt A, Schmitt K, Grimm A et al (2016) Synaptic dysfunction, memory deficits and hippocampal atrophy due to ablation of mitochondrial fission in adult forebrain neurons. Cell Death Differ 23(1):18–28. https://doi.org/10.1038/cdd.2015.39
Ifuku M, Hossain SM, Noda M, Katafuchi T (2014) Induction of interleukin-1β by activated microglia is a prerequisite for immunologically induced fatigue. Eur J Neurosci 40(8):3253–3263. https://doi.org/10.1111/ejn.12668
Cohen GM (1997) Caspases: the executioners of apoptosis. Biochem J 326(Pt 1):1–16
Schmitz ML, Baeuerle PA (1991) The p65 subunit is responsible for the strong transcription activating potential of NF-κB. EMBO J 10(12):3805–3817
Pike LJ, Han X, Chung KN, Gross RW (2002) Lipid rafts are enriched in arachidonic acid and plasmenylethanolamine and their composition is independent of caveolin-1 expression: a quantitative electrospray ionization/mass spectrometric analysis. Biochemistry 41(6):2075–2088
Rodemer C, Thai TP, Brugger B, Kaercher T, Werner H, Nave KA, Wieland F, Gorgas K et al (2003) Inactivation of ether lipid biosynthesis causes male infertility, defects in eye development and optic nerve hypoplasia in mice. Hum Mol Genet 12(15):1881–1895
Thai TP, Rodemer C, Jauch A, Hunziker A, Moser A, Gorgas K, Just WW (2001) Impaired membrane traffic in defective ether lipid biosynthesis. Hum Mol Genet 10(2):127–136
Wanders RJ, Waterham HR (2005) Peroxisomal disorders I: biochemistry and genetics of peroxisome biogenesis disorders. Clin Genet 67(2):107–133. https://doi.org/10.1111/j.1399-0004.2004.00329.x
Kagan JC, Medzhitov R (2006) Phosphoinositide-mediated adaptor recruitment controls Toll-like receptor signaling. Cell 125(5):943–955. https://doi.org/10.1016/j.cell.2006.03.047
Tanimura N, Saitoh S, Matsumoto F, Akashi-Takamura S, Miyake K (2008) Roles for LPS-dependent interaction and relocation of TLR4 and TRAM in TRIF-signaling. Biochem Biophys Res Commun 368(1):94–99. https://doi.org/10.1016/j.bbrc.2008.01.061
Doherty GJ, McMahon HT (2009) Mechanisms of endocytosis. Annu Rev Biochem 78:857–902. https://doi.org/10.1146/annurev.biochem.78.081307.110540
Grant BD, Donaldson JG (2009) Pathways and mechanisms of endocytic recycling. Nat Rev Mol Cell Biol 10(9):597–608. https://doi.org/10.1038/nrm2755
Nichols BJ (2002) A distinct class of endosome mediates clathrin-independent endocytosis to the Golgi complex. Nat Cell Biol 4(5):374–378. https://doi.org/10.1038/ncb787
Acknowledgements
We appreciate the technical assistance of the Research Support Center, Graduate School of Medical Sciences, Kyushu University. We thank Dr. A. Ibrahim for insightful discussion and suggestions to continue this study. We appreciate the technical assistance from Ayako Tajima to perform experiments.
Funding
This work was supported by JSPS KAKENHI grant number 26460320 to Toshihiko Katafuchi, JSPS Wakate B (16K19007) to MSH and Egyptian ministry of higher education scholarship for young scientist fellowship to F. Ali.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Figure S1.
Pls pretreatments attenuate the LPS-mediated cleavages of caspase-8 and caspase-3 proteins in microglial cells. Western blotting data showed that LPS (1 μg/ml) treatments for 6 hours induced cleavage of caspase-8 and caspase-3 (as shown in Fig. 1) in BV2 microglial cells, which were attenuated by pretreatments with 5 μg/ml of sPls for 12 hours. Here, we show the representative data of two independent experiments. (PPTX 153 kb)
Figure S2.
MβCD, an inhibitor of lipid raft function, abolishes the attenuating effect of Pls on LPS-induced IL-1β expression. BV2 cells were pretreated with Pls (5 μg/ml) for 12 hours then MβCD (5 μM) was applied 1 hour before LPS (1 μg/ml) treatment. Real time PCR analysis performed 6 hours after LPS treatment demonstrated that the LPS-induced increase in IL-1β mRNA expression was attenuated by the pretreatment with Pls. However, the attenuation was completely blocked by application of MβCD. MβCD treatment itself did not affect the LPS-induced IL-1β expression (**P<0.01 vs. control group, ##P<0.01 vs. LPS group, n=5) (PPTX 57 kb)
Rights and permissions
About this article
Cite this article
Ali, F., Hossain, M.S., Sejimo, S. et al. Plasmalogens Inhibit Endocytosis of Toll-like Receptor 4 to Attenuate the Inflammatory Signal in Microglial Cells. Mol Neurobiol 56, 3404–3419 (2019). https://doi.org/10.1007/s12035-018-1307-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1307-2