Abstract
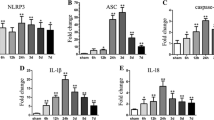
Interleukin-33 (IL-33) is a recently identified member of the IL-1 family that exerts biologic functions by binding to a heterodimer composed of IL-1 receptor-related protein ST2L and IL-1RAcP. However, the role of IL-33 and whether IL-33 accounts for inflammation, apoptotic, and autophagic neuropathology after intracerebral hemorrhage (ICH) are not clear. Here, we established a mouse ICH model in this study, to determine the role of IL-33 and explore the underlying mechanism. Male mice were subjected to an infusion of type IV collagenase/saline into the left striatum to induce ICH/sham model. IL-33, soluble ST2 (sST2), or saline were also administered by a single intracerebroventricular (i.c.v.) injection, respectively. The results showed that the expression level of IL-33 markedly decreased within 6 h and reached the valleys at 6 and 72 h after ICH vs. sham group. In parallel, ST2L (a transmembrane form receptor of IL-33) significantly increased within 6 h and reached the peaks at 6 h and 24 h after ICH vs. sham group. In addition, administration of IL-33 alleviated cerebral water contents, reduced the number of PI- and TUNEL-positive cells, and improved neurological function after ICH. Moreover, IL-33 treatment apparently suppressed the expression of pro-inflammation cytokines IL-1β and TNF-α, evidently increased Bcl-2 but decreased cleaved-caspase-3, and obviously decreased the levels of autophagy-associated proteins LC3-II and Beclin-1 but maintained P62 at high level after ICH. On the contrary, treatment with sST2, a decoy receptor of IL-33, exacerbated ICH-induced brain damage and neurological dysfunction by promoting apoptosis, and enhancing autophagic activity. In conclusion, IL-33 provides neuroprotection through suppressing inflammation, apoptotic, and autophagic activation in collagenase-induced ICH model.






Similar content being viewed by others
References
Xi G, Keep RF, Hoff JT (2006) Mechanisms of brain injury after intracerebral haemorrhage. Lancet Neurol 5(1):53–63. doi:10.1016/S1474-4422(05)70283-0
Krafft PR, Altay O, Rolland WB, Duris K, Lekic T, Tang J, Zhang JH (2012) Alpha7 nicotinic acetylcholine receptor agonism confers neuroprotection through GSK-3beta inhibition in a mouse model of intracerebral hemorrhage. Stroke 43(3):844–850. doi:10.1161/STROKEAHA.111.639989
Hwang BY, Appelboom G, Ayer A, Kellner CP, Kotchetkov IS, Gigante PR, Haque R, Kellner M (2011) Advances in neuroprotective strategies: potential therapies for intracerebral hemorrhage. Cerebrovasc Dis 31(3):211–222. doi:10.1159/000321870
Schmitz J, Owyang A, Oldham E, Song Y, Murphy E, McClanahan TK, Zurawski G, Moshrefi M, Qin J, Li X, Gorman DM, Bazan JF, Kastelein RA (2005) IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 23(5):479–490. doi:10.1016/j.immuni.2005.09.015
Iwahana H, Yanagisawa K, Ito-Kosaka A, Kuroiwa K, Tago K, Komatsu N, Katashima R, Itakura M, Tominaga S (1999) Different promoter usage and multiple transcription initiation sites of the interleukin-1 receptor-related human ST2 gene in UT-7 and TM12 cells. Eur J Biochem 264(2):397–406
Yasuoka S, Kawanokuchi J, Parajuli B, Jin S, Doi Y, Noda M, Sonobe Y, Takeuchi H, Mizuno T, Suzumura A (2011) Production and functions of IL-33 in the central nervous system. Brain Res 1385:8–17. doi:10.1016/j.brainres.2011.02.045
Hudson CA, Christophi GP, Gruber RC, Wilmore JR, Lawrence DA, Massa PT (2008) Induction of IL-33 expression and activity in central nervous system glia. J Leukoc Biol 84(3):631–643. doi:10.1189/jlb.1207830
Palmer G, Talabot-Ayer D, Lamacchia C, Toy D, Seemayer CA, Viatte S, Finckh A, Smith DE, Gabay C (2009) Inhibition of interleukin-33 signaling attenuates the severity of experimental arthritis. Arthritis Rheum 60(3):738–749. doi:10.1002/art.24305
Volarevic V, Mitrovic M, Milovanovic M, Zelen I, Nikolic I, Mitrovic S, Pejnovic N, Arsenijevic N, Lukic ML (2012) Protective role of IL-33/ST2 axis in Con A-induced hepatitis. J Hepatol 56(1):26–33. doi:10.1016/j.jhep.2011.03.022
Xu D, Jiang HR, Kewin P, Li Y, Mu R, Fraser AR, Pitman N, Kurowska-Stolarska M, McKenzie AN, McInnes IB, Liew FY (2008) IL-33 exacerbates antigen-induced arthritis by activating mast cells. Proc Natl Acad Sci U S A 105(31):10913–10918. doi:10.1073/pnas.0801898105
Clark W, Gunion-Rinker L, Lessov N, Hazel K (1998) Citicoline treatment for experimental intracerebral hemorrhage in mice. Stroke 29(10):2136–2140
Garcia JH, Wagner S, Liu KF, Hu XJ (1995) Neurological deficit and extent of neuronal necrosis attributable to middle cerebral artery occlusion in rats. Statistical validation. Stroke 26(4):627–634, discussion 635
Hua Y, Schallert T, Keep RF, Wu J, Hoff JT, Xi G (2002) Behavioral tests after intracerebral hemorrhage in the rat. Stroke 33(10):2478–2484
Kitaoka T, Hua Y, Xi G, Hoff JT, Keep RF (2002) Delayed argatroban treatment reduces edema in a rat model of intracerebral hemorrhage. Stroke 33(12):3012–3018
Zhu X, Tao L, Tejima-Mandeville E, Qiu J, Park J, Garber K, Ericsson M, Lo EH, Whalen MJ (2012) Plasmalemma permeability and necrotic cell death phenotypes after intracerebral hemorrhage in mice. Stroke 43(2):524–531. doi:10.1161/STROKEAHA.111.635672
Arshad MI, Rauch M, L’Helgoualc’h A, Julia V, Leite-de-Moraes MC, Lucas-Clerc C, Piquet-Pellorce C, Samson M (2011) NKT cells are required to induce high IL-33 expression in hepatocytes during ConA-induced acute hepatitis. Eur J Immunol 41(8):2341–2348. doi:10.1002/eji.201041332
Yang GY, Betz AL, Hoff JT (1994) The effects of blood or plasma clot on brain edema in the rat with intracerebral hemorrhage. Acta Neurochir Suppl (Wien) 60:555–557
Whalen MJ, Dalkara T, You Z, Qiu J, Bermpohl D, Mehta N, Suter B, Bhide PG, Lo EH, Ericsson M, Moskowitz MA (2008) Acute plasmalemma permeability and protracted clearance of injured cells after controlled cortical impact in mice. J Cereb Blood Flow Metab 28(3):490–505. doi:10.1038/sj.jcbfm.9600544
Zhang M, Shan H, Chang P, Wang T, Dong W, Chen X, Tao L (2014) Hydrogen sulfide offers neuroprotection on traumatic brain injury in parallel with reduced apoptosis and autophagy in mice. PLoS One 9(1), e87241. doi:10.1371/journal.pone.0087241
Megyeri P, Abraham CS, Temesvari P, Kovacs J, Vas T, Speer CP (1992) Recombinant human tumor necrosis factor alpha constricts pial arterioles and increases blood-brain barrier permeability in newborn piglets. Neurosci Lett 148(1-2):137–140
Holmin S, Mathiesen T (2000) Intracerebral administration of interleukin-1beta and induction of inflammation, apoptosis, and vasogenic edema. J Neurosurg 92(1):108–120. doi:10.3171/jns.2000.92.1.0108
Carriere V, Roussel L, Ortega N, Lacorre DA, Americh L, Aguilar L, Bouche G, Girard JP (2007) IL-33, the IL-1-like cytokine ligand for ST2 receptor, is a chromatin-associated nuclear factor in vivo. Proc Natl Acad Sci U S A 104(1):282–287. doi:10.1073/pnas.0606854104
Peng H, Sun R, Zhang Q, Zhao J, Wei J, Zeng X, Zheng H, Wu Z (2013) Interleukin 33 mediates type 2 immunity and inflammation in the central nervous system of mice infected with Angiostrongylus cantonensis. J Infect Dis 207(5):860–869. doi:10.1093/infdis/jis682
Milovanovic M, Volarevic V, Radosavljevic G, Jovanovic I, Pejnovic N, Arsenijevic N, Lukic ML (2012) IL-33/ST2 axis in inflammation and immunopathology. Immunol Res 52(1-2):89–99. doi:10.1007/s12026-012-8283-9
Dinarello CA (1996) Biologic basis for interleukin-1 in disease. Blood 87(6):2095–2147
Dinarello CA (2000) Interleukin-18, a proinflammatory cytokine. Eur Cytokine Netw 11(3):483–486
Cayrol C, Girard JP (2009) The IL-1-like cytokine IL-33 is inactivated after maturation by caspase-1. Proc Natl Acad Sci U S A 106(22):9021–9026. doi:10.1073/pnas.0812690106
Luthi AU, Cullen SP, McNeela EA, Duriez PJ, Afonina IS, Sheridan C, Brumatti G, Taylor RC, Kersse K, Vandenabeele P, Lavelle EC, Martin SJ (2009) Suppression of interleukin-33 bioactivity through proteolysis by apoptotic caspases. Immunity 31(1):84–98. doi:10.1016/j.immuni.2009.05.007
Ali S, Nguyen DQ, Falk W, Martin MU (2010) Caspase 3 inactivates biologically active full length interleukin-33 as a classical cytokine but does not prohibit nuclear translocation. Biochem Biophys Res Commun 391(3):1512–1516. doi:10.1016/j.bbrc.2009.12.107
Kumar S, Tzimas MN, Griswold DE, Young PR (1997) Expression of ST2, an interleukin-1 receptor homologue, is induced by proinflammatory stimuli. Biochem Biophys Res Commun 235(3):474–478. doi:10.1006/bbrc.1997.6810
Wicher G, Husic E, Nilsson G, Forsberg-Nilsson K (2013) Developmental expression of IL-33 in the mouse brain. Neurosci Lett 555:171–176. doi:10.1016/j.neulet.2013.09.046
Jiang HR, Milovanovic M, Allan D, Niedbala W, Besnard AG, Fukada SY, Alves-Filho JC, Togbe D, Goodyear CS, Linington C, Xu D, Lukic ML, Liew FY (2012) IL-33 attenuates EAE by suppressing IL-17 and IFN-gamma production and inducing alternatively activated macrophages. Eur J Immunol 42(7):1804–1814. doi:10.1002/eji.201141947
Huang LT, Li H, Sun Q, Liu M, Li WD, Li S, Yu Z, Wei WT, Hang CH (2015) IL-33 expression in the cerebral cortex following experimental subarachnoid hemorrhage in rats. Cell Mol Neurobiol 35(4):493–501. doi:10.1007/s10571-014-0143-9
Wu H, Zhao R, Qi J, Cong Y, Wang D, Liu T, Gu Y, Ban X, Huang Q (2008) The expression and the role of protease nexin-1 on brain edema after intracerebral hemorrhage. J Neurol Sci 270(1-2):172–183. doi:10.1016/j.jns.2008.03.010
Gordon CR, Merchant RS, Marmarou A, Rice CD, Marsh JT, Young HF (1990) Effect of murine recombinant interleukin-1 on brain oedema in the rat. Acta Neurochir Suppl (Wien) 51:268–270
Barone FC, Arvin B, White RF, Miller A, Webb CL, Willette RN, Lysko PG, Feuerstein GZ (1997) Tumor necrosis factor-alpha. A mediator of focal ischemic brain injury. Stroke 28(6):1233–1244
Guadagno J, Swan P, Shaikh R, Cregan SP (2015) Microglia-derived IL-1beta triggers p53-mediated cell cycle arrest and apoptosis in neural precursor cells. Cell Death Dis 6, e1779. doi:10.1038/cddis.2015.151
Tolosa L, Caraballo-Miralles V, Olmos G, Llado J (2011) TNF-alpha potentiates glutamate-induced spinal cord motoneuron death via NF-kappaB. Mol Cell Neurosci 46(1):176–186. doi:10.1016/j.mcn.2010.09.001
Matsushita K, Meng W, Wang X, Asahi M, Asahi K, Moskowitz MA, Lo EH (2000) Evidence for apoptosis after intercerebral hemorrhage in rat striatum. J Cereb Blood Flow Metab 20(2):396–404. doi:10.1097/00004647-200002000-00022
He Y, Liu W, Koch LG, Britton SL, Keep RF, Xi G, Hua Y (2012) Susceptibility to intracerebral hemorrhage-induced brain injury segregates with low aerobic capacity in rats. Neurobiol Dis 49C:22–28. doi:10.1016/j.nbd.2012.08.014
Pastore D, Della-Morte D, Coppola A, Capuani B, Lombardo MF, Pacifici F, Ferrelli F, Arriga R, Mammi C, Federici M, Bellia A, Di Daniele N, Tesauro M, Donadel G, Noto D, Sbraccia P, Sconocchia G, Lauro D (2015) SGK-1 protects kidney cells against apoptosis induced by ceramide and TNF-alpha. Cell Death Dis 6, e1890. doi:10.1038/cddis.2015.232
Moriwaki T, Takagi Y, Sadamasa N, Aoki T, Nozaki K, Hashimoto N (2006) Impaired progression of cerebral aneurysms in interleukin-1beta-deficient mice. Stroke 37(3):900–905. doi:10.1161/01.STR.0000204028.39783.d9
Geddes DM, Cargill RS 2nd, LaPlaca MC (2003) Mechanical stretch to neurons results in a strain rate and magnitude-dependent increase in plasma membrane permeability. J Neurotrauma 20(10):1039–1049. doi:10.1089/089771503770195885
Soueid J, Nokkari A, Makoukji J (2015) Techniques and methods of animal brain surgery: perfusion, brain removal, and histological techniques. doi: NBK299226 [bookaccession]
Wang JX, Kaieda S, Ameri S, Fishgal N, Dwyer D, Dellinger A, Kepley CL, Gurish MF, Nigrovic PA (2014) IL-33/ST2 axis promotes mast cell survival via BCLXL. Proc Natl Acad Sci U S A 111(28):10281–10286. doi:10.1073/pnas.1404182111
Sakai N, Van Sweringen HL, Quillin RC, Schuster R, Blanchard J, Burns JM, Tevar AD, Edwards MJ, Lentsch AB (2012) Interleukin-33 is hepatoprotective during liver ischemia/reperfusion in mice. Hepatology 56(4):1468–1478. doi:10.1002/hep.25768
He Y, Wan S, Hua Y, Keep RF, Xi G (2008) Autophagy after experimental intracerebral hemorrhage. J Cereb Blood Flow Metab 28(5):897–905. doi:10.1038/sj.jcbfm.9600578
Wang CW, Klionsky DJ (2003) The molecular mechanism of autophagy. Mol Med 9(3-4):65–76
Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T, Kominami E, Ohsumi Y, Yoshimori T (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 19(21):5720–5728. doi:10.1093/emboj/19.21.5720
Itakura E, Kishi C, Inoue K, Mizushima N (2008) Beclin 1 forms two distinct phosphatidylinositol 3-kinase complexes with mammalian Atg14 and UVRAG. Mol Biol Cell 19(12):5360–5372. doi:10.1091/mbc.E08-01-0080
Ichimura Y, Kominami E, Tanaka K, Komatsu M (2008) Selective turnover of p62/A170/SQSTM1 by autophagy. Autophagy 4(8):1063–1066
Thorburn A (2008) Apoptosis and autophagy: regulatory connections between two supposedly different processes. Apoptosis 13(1):1–9. doi:10.1007/s10495-007-0154-9
Satoo K, Noda NN, Kumeta H, Fujioka Y, Mizushima N, Ohsumi Y, Inagaki F (2009) The structure of Atg4B-LC3 complex reveals the mechanism of LC3 processing and delipidation during autophagy. EMBO J 28(9):1341–1350. doi:10.1038/emboj.2009.80
Pattingre S, Levine B (2006) Bcl-2 inhibition of autophagy: a new route to cancer? Cancer Res 66(6):2885–2888. doi:10.1158/0008-5472.CAN-05-4412
Pattingre S, Tassa A, Qu X, Garuti R, Liang XH, Mizushima N, Packer M, Schneider MD, Levine B (2005) Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell 122(6):927–939. doi:10.1016/j.cell.2005.07.002
Sadasivan S, Dunn WA Jr, Hayes RL, Wang KK (2008) Changes in autophagy proteins in a rat model of controlled cortical impact induced brain injury. Biochem Biophys Res Commun 373(4):478–481. doi:10.1016/j.bbrc.2008.05.031
Luo CL, Li BX, Li QQ, Chen XP, Sun YX, Bao HJ, Dai DK, Shen YW, Xu HF, Ni H, Wan L, Qin ZH, Tao LY, Zhao ZQ (2011) Autophagy is involved in traumatic brain injury-induced cell death and contributes to functional outcome deficits in mice. Neuroscience 184:54–63. doi:10.1016/j.neuroscience.2011.03.021
Jin Z, Li Y, Pitti R, Lawrence D, Pham VC, Lill JR, Ashkenazi A (2009) Cullin3-based polyubiquitination and p62-dependent aggregation of caspase-8 mediate extrinsic apoptosis signaling. Cell 137(4):721–735. doi:10.1016/j.cell.2009.03.015
Levine B, Mizushima N, Virgin HW (2011) Autophagy in immunity and inflammation. Nature 469(7330):323–335. doi:10.1038/nature09782
Harris J, Hartman M, Roche C, Zeng SG, O’Shea A, Sharp FA, Lambe EM, Creagh EM, Golenbock DT, Tschopp J, Kornfeld H, Fitzgerald KA, Lavelle EC (2011) Autophagy controls IL-1beta secretion by targeting pro-IL-1beta for degradation. J Biol Chem 286(11):9587–9597. doi:10.1074/jbc.M110.202911
Acknowledgments
This study was supported by grants 81271379, 81373251, and 81530062 from the National Natural Science Foundation of China, National High Technology Research and Development Program of China (863 Program, No.2015AA020503), and a Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Gao, Y., Ma, L., Luo, Cl. et al. IL-33 Exerts Neuroprotective Effect in Mice Intracerebral Hemorrhage Model Through Suppressing Inflammation/Apoptotic/Autophagic Pathway. Mol Neurobiol 54, 3879–3892 (2017). https://doi.org/10.1007/s12035-016-9947-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-9947-6