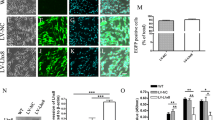
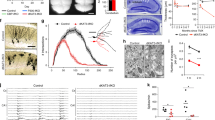
Abstract
The mouse neuroblastoma N18TG2 clone is unable to differentiate and is defective for the enzymes of the biosynthesis of neurotransmitters. The forced expression of choline acetyltransferase (ChAT) in these cells results in the synthesis and release of acetylcholine (Ach) and hence in the expression of neurospecific features and markers. To understand how the expression of ChAT triggered neuronal differentiation, we studied the differences in genome-wide transcription profiles between the N18TG2 parental cells and its ChAT-expressing 2/4 derived clone. The engagement of the 2/4 cells in the neuronal developmental program was confirmed by the increase of the expression level of several differentiation-related genes and by the reduction of the amount of transcripts of cell cycle genes. At the same time, we observed a massive reorganization of cytoskeletal proteins in terms of gene expression, with the accumulation of the nucleoskeletal lamina component Lamin A/C in differentiating cells. The increase of the Lmna transcripts induced by ChAT expression in 2/4 cells was mimicked treating the parental N18TG2 cells with the acetylcholine receptor agonist carbachol, thus demonstrating the direct role played by this receptor in neuron nuclei maturation. Conversely, a treatment of 2/4 cells with the muscarinic receptor antagonist atropine resulted in the reduction of the amount of Lmna RNA. Finally, the hypothesis that Lmna gene product might play a crucial role in the ChAT-dependent molecular differentiation cascade was strongly supported by Lmna knockdown in 2/4 cells leading to the downregulation of genes involved in differentiation and cytoskeleton formation and to the upregulation of genes known to regulate self-renewal and stemness.







Similar content being viewed by others
References
Nguyen L, Rigo JM, Rocher V, Belachew S, Malgrange B, Rogister B, Leprince P, Moonen G (2001) Neurotransmitters as early signals for central nervous system development. Cell Tissue Res 305(2):187–202
Lauder JM (1993) Neurotransmitters as growth regulatory signals: role of receptors and second messengers. Trends Neurosci 16(6):233–240
Mattson MP, Furukawa K (1998) Signaling events regulating the neurodevelopmental triad. Glutamate and secreted forms of beta-amyloid precursor protein as examples. Perspect Dev Neurobiol 5(4):337–352
Bovetti S, Gribaudo S, Puche AC, De Marchis S, Fasolo A (2011) From progenitors to integrated neurons: role of neurotransmitters in adult olfactory neurogenesis. J Chem Neuroanat 42(4):304–316. doi:10.1016/j.jchemneu.2011.05.006
Horiuchi Y, Kimura R, Kato N, Fujii T, Seki M, Endo T, Kato T, Kawashima K (2003) Evolutional study on acetylcholine expression. Life Sci 72(15):1745–1756
Wessler I, Kirkpatrick CJ (2008) Acetylcholine beyond neurons: the non-neuronal cholinergic system in humans. Br J Pharmacol 154(8):1558–1571. doi:10.1038/bjp.2008.185
Ruediger T, Bolz J (2007) Neurotransmitters and the development of neuronal circuits. In: Bagnard D (ed) Axon growth and guidance. Landes Bioscience and Springer Science
Nelson P, Christian C, Niremberg M (1976) Synapse formation between clonal neuroblastoma x glioma hybrid cells and striated muscle cells. Proc Natl Acad Sci U S A 73:123–127
Hamprecht B (1977) Structural, electrophysiological, biochemical, and pharmacological properties of neuroblastoma-glioma cell hybrids in cell culture. Int Rev Cytol 49:99–170
Bignami F, Bevilacqua P, Biagioni S, De Jaco A, Casamenti F, Felsani A, Augusti-Tocco G (1997) Cellular acetylcholine content and neuronal differentiation. J Neurochem 69(4):1374–1381
Biagioni S, Tata AM, De Jaco A, Augusti-Tocco G (2000) Acetylcholine synthesis and neuron differentiation. Int J Dev Biol 44(6):689–697
De Jaco A, Augusti-Tocco G, Biagioni S (2002) Muscarinic acetylcholine receptors induce neurite outgrowth and activate the synapsin I gene promoter in neuroblastoma clones. Neuroscience 113(2):331–338
Salani M, Anelli T, Tocco GA, Lucarini E, Mozzetta C, Poiana G, Tata AM, Biagioni S (2009) Acetylcholine-induced neuronal differentiation: muscarinic receptor activation regulates EGR-1 and REST expression in neuroblastoma cells. J Neurochem 108(3):821–834. doi:10.1111/j.1471-4159.2008.05829.x
Kapitein LC, Hoogenraad CC (2015) Building the neuronal microtubule cytoskeleton. Neuron 87(3):492–506. doi:10.1016/j.neuron.2015.05.046
Volk T (2013) Positioning nuclei within the cytoplasm of striated muscle fiber: cooperation between microtubules and KASH proteins. Nucleus 4(1):18–22. doi:10.4161/nucl.23086
Elhanany-Tamir H, Yu YV, Shnayder M, Jain A, Welte M, Volk T (2012) Organelle positioning in muscles requires cooperation between two KASH proteins and microtubules. J Cell Biol 198(5):833–846. doi:10.1083/jcb.201204102
Razafsky DS, Ward CL, Kolb T, Hodzic D (2013) Developmental regulation of linkers of the nucleoskeleton to the cytoskeleton during mouse postnatal retinogenesis. Nucleus 4(5):399–409. doi:10.4161/nucl.26244
Razafsky D, Blecher N, Markov A, Stewart-Hutchinson PJ, Hodzic D (2012) LINC complexes mediate the positioning of cone photoreceptor nuclei in mouse retina. PLoS One 7(10):e47180. doi:10.1371/journal.pone.0047180
Aebi U, Cohn J, Buhle L, Gerace L (1986) The nuclear lamina is a meshwork of intermediate-type filaments. Nature 323(6088):560–564. doi:10.1038/323560a0
Sullivan T, Escalante-Alcalde D, Bhatt H, Anver M, Bhat N, Nagashima K, Stewart CL, Burke B (1999) Loss of A-type lamin expression compromises nuclear envelope integrity leading to muscular dystrophy. J Cell Biol 147(5):913–920
Constantinescu D, Gray HL, Sammak PJ, Schatten GP, Csoka AB (2006) Lamin A/C expression is a marker of mouse and human embryonic stem cell differentiation. Stem Cells 24(1):177–185. doi:10.1634/stemcells.2004-0159
Maresca G, Natoli M, Nardella M, Arisi I, Trisciuoglio D, Desideri M, Brandi R, D’Aguanno S et al (2012) LMNA knock-down affects differentiation and progression of human neuroblastoma cells. PLoS One 7(9):e45513. doi:10.1371/journal.pone.0045513
Nardella M, Guglielmi L, Musa C, Iannetti I, Maresca G, Amendola D, Porru M, Carico E, Sessa G, Camerlingo R, Dominici C, Megiorni F, Milan M, Bearzi C, Rizzi R, Pirozzi G, Leonetti C, Bucci B, Mercanti D, Felsani A, D’Agnano I (2015) Down-regulation of the Lamin A/C in neuroblastoma triggers the expansion of tumor initiating cells. 2015
Vanburen V, Chen H (2012) Managing biological complexity across orthologs with a visual knowledgebase of documented biomolecular interactions. Sci Rep 2:1011. doi:10.1038/srep01011
Herlenius E, Lagercrantz H (2001) Neurotransmitters and neuromodulators during early human development. Early Hum Dev 65(1):21–37. doi:10.1016/S0378-3782(01)00189-X
Bolz TRaJ (2007) Neurotransmitters and the development of neuronal circuits. In: Bagnard D (ed) Axon growth and guidance. Landes Bioscience and Springer Science+ Business Media pp 104–115
Berg DA, Belnoue L, Song H, Simon A (2013) Neurotransmitter-mediated control of neurogenesis in the adult vertebrate brain. Development 140(12):2548–2561. doi:10.1242/dev.088005
Pathania M, Yan LD, Bordey A (2010) A symphony of signals conducts early and late stages of adult neurogenesis. Neuropharmacology 58(6):865–876. doi:10.1016/j.neuropharm.2010.01.010
Benninghoff J, Rauh W, Brantl V, Schloesser RJ, Moessner R, Moller HJ, Rujescu D (2013) Cholinergic impact on neuroplasticity drives muscarinic M1 receptor mediated differentiation into neurons. World J Biol Psychiatry 14(3):241–246. doi:10.3109/15622975.2011.624121
Ma W, Maric D, Li BS, Hu Q, Andreadis JD, Grant GM, Liu QY, Shaffer KM et al (2000) Acetylcholine stimulates cortical precursor cell proliferation in vitro via muscarinic receptor activation and MAP kinase phosphorylation. Eur J Neurosci 12(4):1227–1240
Politis PK, Makri G, Thomaidou D, Geissen M, Rohrer H, Matsas R (2007) BM88/CEND1 coordinates cell cycle exit and differentiation of neuronal precursors. Proc Natl Acad Sci U S A 104(45):17861–17866. doi:10.1073/pnas.0610973104
Huang C, Qin D (2010) Role of Lef1 in sustaining self-renewal in mouse embryonic stem cells. J Genet Genomics 37(7):441–449. doi:10.1016/S1673-8527(09)60063-1
Lendahl U, Zimmerman LB, McKay RD (1990) CNS stem cells express a new class of intermediate filament protein. Cell 60(4):585–595
Hockfield S, McKay RD (1985) Identification of major cell classes in the developing mammalian nervous system. J Neurosci Off J Soc Neurosci 5(12):3310–3328
Weigmann A, Corbeil D, Hellwig A, Huttner WB (1997) Prominin, a novel microvilli-specific polytopic membrane protein of the apical surface of epithelial cells, is targeted to plasmalemmal protrusions of non-epithelial cells. Proc Natl Acad Sci U S A 94(23):12425–12430
Corbeil D, Karbanova J, Fargeas CA, Jaszai J (2013) Prominin-1 (CD133): molecular and cellular features across species. Adv Exp Med Biol 777:3–24. doi:10.1007/978-1-4614-5894-4_1
Hall DH, Treinin M (2011) How does morphology relate to function in sensory arbors? Trends Neurosci 34(9):443–451. doi:10.1016/j.tins.2011.07.004
Sasaki T, Matsuki N, Ikegaya Y (2012) Effects of axonal topology on the somatic modulation of synaptic outputs. J Neurosci Off J Soc Neurosci 32(8):2868–2876. doi:10.1523/JNEUROSCI.5365-11.2012
Stewart E, Shen K (2015) STORMing towards a clear picture of the cytoskeleton in neurons. eLife 4. doi:10.7554/eLife.06235
Bennett V, Lorenzo DN (2013) Spectrin- and ankyrin-based membrane domains and the evolution of vertebrates. Curr Top Membr 72:1–37. doi:10.1016/B978-0-12-417027-8.00001-5
Kapitein LC, Hoogenraad CC (2011) Which way to go? Cytoskeletal organization and polarized transport in neurons. Mol Cell Neurosci 46(1):9–20. doi:10.1016/j.mcn.2010.08.015
Machnicka B, Czogalla A, Hryniewicz-Jankowska A, Boguslawska DM, Grochowalska R, Heger E, Sikorski AF (2014) Spectrins: a structural platform for stabilization and activation of membrane channels, receptors and transporters. Biochim Biophys Acta 1838(2):620–634. doi:10.1016/j.bbamem.2013.05.002
Susuki K, Rasband MN (2008) Spectrin and ankyrin-based cytoskeletons at polarized domains in myelinated axons. Exp Biol Med (Maywood) 233(4):394–400. doi:10.3181/0709-MR-243
Causeret F, Jacobs T, Terao M, Heath O, Hoshino M, Nikolic M (2007) Neurabin-I is phosphorylated by Cdk5: implications for neuronal morphogenesis and cortical migration. Mol Biol Cell 18(11):4327–4342. doi:10.1091/mbc.E07-04-0372
Kawauchi T (2014) Cdk5 regulates multiple cellular events in neural development, function and disease. Dev Growth Differ 56(5):335–348. doi:10.1111/dgd.12138
Tanabe K, Yamazaki H, Inaguma Y, Asada A, Kimura T, Takahashi J, Taoka M, Ohshima T et al (2014) Phosphorylation of drebrin by cyclin-dependent kinase 5 and its role in neuronal migration. PLoS One 9(3):e92291. doi:10.1371/journal.pone.0092291
Shah K, Lahiri DK (2014) Cdk5 activity in the brain—multiple paths of regulation. J Cell Sci 127(Pt 11):2391–2400. doi:10.1242/jcs.147553
Tischfield MA, Baris HN, Wu C, Rudolph G, Van Maldergem L, He W, Chan WM, Andrews C et al (2010) Human TUBB3 mutations perturb microtubule dynamics, kinesin interactions, and axon guidance. Cell 140(1):74–87. doi:10.1016/j.cell.2009.12.011
Qu C, Dwyer T, Shao Q, Yang T, Huang H, Liu G (2013) Direct binding of TUBB3 with DCC couples netrin-1 signaling to intracellular microtubule dynamics in axon outgrowth and guidance. J Cell Sci 126(Pt 14):3070–3081. doi:10.1242/jcs.122184
van Haren J, Draegestein K, Keijzer N, Abrahams JP, Grosveld F, Peeters PJ, Moechars D, Galjart N (2009) Mammalian navigators are microtubule plus-end tracking proteins that can reorganize the cytoskeleton to induce neurite-like extensions. Cell Motil Cytoskeleton 66(10):824–838. doi:10.1002/cm.20370
Wanschers B, van de Vorstenbosch R, Wijers M, Wieringa B, King SM, Fransen J (2008) Rab6 family proteins interact with the dynein light chain protein DYNLRB1. Cell Motil Cytoskeleton 65(3):183–196. doi:10.1002/cm.20254
Street M, Marsh SJ, Stabach PR, Morrow JS, Brown DA, Buckley NJ (2006) Stimulation of Galphaq-coupled M1 muscarinic receptor causes reversible spectrin redistribution mediated by PLC, PKC and ROCK. J Cell Sci 119(Pt 8):1528–1536. doi:10.1242/jcs.02872
Teber I, Kohling R, Speckmann EJ, Barnekow A, Kremerskothen J (2004) Muscarinic acetylcholine receptor stimulation induces expression of the activity-regulated cytoskeleton-associated gene (ARC). Brain Res Mol Brain Res 121(1–2):131–136. doi:10.1016/j.molbrainres.2003.11.017
Belyantseva IA, Perrin BJ, Sonnemann KJ, Zhu M, Stepanyan R, McGee J, Frolenkov GI, Walsh EJ et al (2009) Gamma-actin is required for cytoskeletal maintenance but not development. Proc Natl Acad Sci U S A 106(24):9703–9708. doi:10.1073/pnas.0900221106
Navratil AM, Dozier MG, Whitesell JD, Clay CM, Roberson MS (2014) Role of cortactin in dynamic actin remodeling events in gonadotrope cells. Endocrinology 155(2):548–557. doi:10.1210/en.2012-1924
Yokoyama T, Copeland NG, Jenkins NA, Montgomery CA, Elder FF, Overbeek PA (1993) Reversal of left-right asymmetry: a situs inversus mutation. Science 260(5108):679–682
Arstikaitis P, Gauthier-Campbell C, Carolina Gutierrez Herrera R, Huang K, Levinson JN, Murphy TH, Kilimann MW, Sala C et al (2008) Paralemmin-1, a modulator of filopodia induction is required for spine maturation. Mol Biol Cell 19(5):2026–2038. doi:10.1091/mbc.E07-08-0802
Kang HJ, Voleti B, Hajszan T, Rajkowska G, Stockmeier CA, Licznerski P, Lepack A, Majik MS et al (2012) Decreased expression of synapse-related genes and loss of synapses in major depressive disorder. Nat Med 18(9):1413–1417. doi:10.1038/nm.2886
Grigoriev I, Splinter D, Keijzer N, Wulf PS, Demmers J, Ohtsuka T, Modesti M, Maly IV et al (2007) Rab6 regulates transport and targeting of exocytotic carriers. Dev Cell 13(2):305–314. doi:10.1016/j.devcel.2007.06.010
Wiche G, Winter L (2011) Plectin isoforms as organizers of intermediate filament cytoarchitecture. Bioarchitecture 1(1):14–20. doi:10.4161/bioa.1.1.14630
Zhang Q, Ragnauth C, Greener MJ, Shanahan CM, Roberts RG (2002) The nesprins are giant actin-binding proteins, orthologous to Drosophila melanogaster muscle protein MSP-300. Genomics 80(5):473–481
Wilhelmsen K, Litjens SH, Kuikman I, Tshimbalanga N, Janssen H, van den Bout I, Raymond K, Sonnenberg A (2005) Nesprin-3, a novel outer nuclear membrane protein, associates with the cytoskeletal linker protein plectin. J Cell Biol 171(5):799–810. doi:10.1083/jcb.200506083
Haque F, Mazzeo D, Patel JT, Smallwood DT, Ellis JA, Shanahan CM, Shackleton S (2010) Mammalian SUN protein interaction networks at the inner nuclear membrane and their role in laminopathy disease processes. J Biol Chem 285(5):3487–3498. doi:10.1074/jbc.M109.071910
Dahl KN, Kalinowski A (2011) Nucleoskeleton mechanics at a glance. J Cell Sci 124(Pt 5):675–678. doi:10.1242/jcs.069096
Zhong Z, Chang SA, Kalinowski A, Wilson KL, Dahl KN (2010) Stabilization of the spectrin-like domains of nesprin-1alpha by the evolutionarily conserved “adaptive” domain. Cell Mol Bioeng 3(2):139–150. doi:10.1007/s12195-010-0121-3
Smith ER, Zhang XY, Capo-Chichi CD, Chen X, Xu XX (2011) Increased expression of Syne1/nesprin-1 facilitates nuclear envelope structure changes in embryonic stem cell differentiation. Dev Dyn Off Publ Am Assoc Anatomists 240(10):2245–2255. doi:10.1002/dvdy.22717
Schlager MA, Kapitein LC, Grigoriev I, Burzynski GM, Wulf PS, Keijzer N, de Graaff E, Fukuda M et al (2010) Pericentrosomal targeting of Rab6 secretory vesicles by Bicaudal-D-related protein 1 (BICDR-1) regulates neuritogenesis. EMBO J 29(10):1637–1651. doi:10.1038/emboj.2010.51
Huang DW, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4(1):44–57. doi:10.1038/nprot.2008.211
Saeed AI, Bhagabati NK, Braisted JC, Liang W, Sharov V, Howe EA, Li J, Thiagarajan M et al (2006) TM4 microarray software suite. Methods Enzymol 411:134–193. doi:10.1016/S0076-6879(06)11009-5
Acknowledgments
This study was partially supported by a Joint Grant CNR-EBRI “Molecular and cellular mechanisms of brain plasticity” and by PNR-CNR Aging Program 2012–2014. Authors affiliated to EBRI were supported by European FP7 PAINCAGE grant n.603191, coordinated by Antonino Cattaneo. The authors thank Dr. Delio Mercanti for his continuous and invaluable research support at CNR.
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Supplementary Fig. S1
a. Divergence of expression pattern in N18TG2 and 2/4 cells after RA treatment. Correlation plot between Log2 intensity fluorescence values of 2/4 and N18TG2 cells in RA-treated conditions. The values reported compare transcripts modulation during RA stimulus for each cell line. b. Correlation of expression pattern in 2/4 cells. Correlation plot between Log2 intensity fluorescence values of 2/4 cells in untreated (CTR) and RA-treated conditions. The values reported compare transcripts modulation after RA treatment in 2/4 cells. (PDF 1385 kb)
Supplementary Table S1
Total gene expression data after filtering-out the transcripts with replicate values showing a coefficient of variation larger than 2.0 and those with replicate-averaged fluorescence intensity values below the background. (XLSX 9980 kb)
Supplementary Table S2
Functional gene categories found by analyzing with the DAVID tool the differentially expressed genes between 2/4 and N18TG2 cell lines in basal culture conditions. Upregulated and downregulated GO categories, along with the gene list belonging to each category, are reported in different worksheets. (XLSX 170 kb)
Supplementary Table S3
Functional gene categories found by analyzing with the DAVID tool the common and differentially expressed genes between 2/4 and N18TG2 cell lines after RA treatment. Common and differentially upregulated and downregulated GO categories, along with the gene list belonging to each category, are reported in different worksheets. (XLSX 225 kb)
Rights and permissions
About this article
Cite this article
Guglielmi, L., Nardella, M., Musa, C. et al. Lamin A/C Is Required for ChAT-Dependent Neuroblastoma Differentiation. Mol Neurobiol 54, 3729–3744 (2017). https://doi.org/10.1007/s12035-016-9902-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-9902-6