Abstract
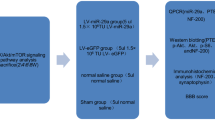
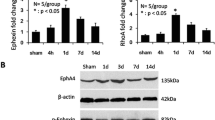
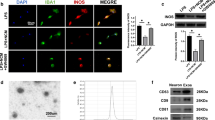
Based on the observation that microRNA (miRNA) 133b enhances regeneration after spinal cord injury in the adult zebrafish, we investigated whether this miRNA would be beneficial in a mammalian system in vitro and in vivo. We found that infection of cultured neurons with miR-133b promotes neurite outgrowth in vitro on an inhibitory substrate consisting of mixed chondroitin sulfate proteoglycans, when compared to infection with green fluorescent protein (GFP) for control. In vivo, viral infection of the injured adult mouse spinal cord at the time of injury at and in the vicinity of the lesion site enhanced expression of miR-133b. Measurements of locomotor recovery by Basso Mouse Scale (BMS) showed improvement of recovery starting at 4 weeks after injury and virus injection. This improvement was associated with downregulation of the expression levels of Ras homolog gene family member A (RhoA), chondroitin sulfate proteoglycans, and microglia/macrophage marker in the spinal cord as assayed 6 weeks after injury. Potential inhibitory molecules carrying consensus sequences for binding of miR-133b were identified in silico and verified in a reporter assay in vitro showing reductions in expression of RhoA, xylosyltransferase 1 (Xylt1), ephrin receptor A7 (Epha7), and purinergic receptor P2X ligand-gated ion channel 4 (P2RX4). These results encourage targeting miR-133 for therapy.









Similar content being viewed by others
References
Goldshmit Y, Kanner S, Zacs M, Frisca F, Pinto AR, Currie PD, Pinkas-Kramarski R (2015) Rapamycin increases neuronal survival, reduces inflammation and astrocyte proliferation after spinal cord injury. Mol Cell Neurosci 68:82–91. doi:10.1016/j.mcn.2015.04.006
Donnelly DJ, Popovich PG (2008) Inflammation and its role in neuroprotection, axonal regeneration and functional recovery after spinal cord injury. Exp Neurol 209(2):378–388. doi:10.1016/j.expneurol.2007.06.009
Schnell L, Fearn S, Schwab ME, Perry VH, Anthony DC (1999) Cytokine-induced acute inflammation in the brain and spinal cord. J Neuropathol Exp Neurol 58(3):245–254
Beattie CE, Melancon E, Eisen JS (2000) Mutations in the stumpy gene reveal intermediate targets for zebrafish motor axons. Development 127(12):2653–2662
David S, Greenhalgh AD, Kroner A (2015) Macrophage and microglial plasticity in the injured spinal cord. Neuroscience 307:311–318. doi:10.1016/j.neuroscience.2015.08.064
De Luca C, Savarese L, Colangelo AM, Bianco MR, Cirillo G, Alberghina L, Papa M (2016) Astrocytes and microglia-mediated immune response in maladaptive plasticity is differently modulated by NGF in the ventral horn of the spinal cord following peripheral nerve injury. Cell Mol Neurobiol 36(1):37–46. doi:10.1007/s10571-015-0218-2
Ren Y, Young W (2013) Managing inflammation after spinal cord injury through manipulation of macrophage function. Neural Plast 2013:945034. doi:10.1155/2013/945034
Trivedi A, Olivas AD, Noble-Haeusslein LJ (2006) Inflammation and spinal cord injury: infiltrating leukocytes as determinants of injury and repair processes. Clin Neurosci Res 6(5):283–292. doi:10.1016/j.cnr.2006.09.007
You J, Hong SQ, Zhang MY, Zhao HL, Liu TZ, Zhou HL, Cai YQ, Xu ZM et al (2012) Passive immunization with tenascin-R (TN-R) polyclonal antibody promotes axonal regeneration and functional recovery after spinal cord injury in rats. Neurosci Lett 525(2):129–134. doi:10.1016/j.neulet.2012.08.002
Apostolova I, Irintchev A, Schachner M (2006) Tenascin-R restricts posttraumatic remodeling of motoneuron innervation and functional recovery after spinal cord injury in adult mice. J Neurosci 26(30):7849–7859. doi:10.1523/JNEUROSCI.1526-06.2006
Qiu J, Cai D, Dai H, McAtee M, Hoffman PN, Bregman BS, Filbin MT (2002) Spinal axon regeneration induced by elevation of cyclic AMP. Neuron 34(6):895–903
Schwab ME (2002) Increasing plasticity and functional recovery of the lesioned spinal cord. Prog Brain Res 137:351–359
Carulli D, Rhodes KE, Brown DJ, Bonnert TP, Pollack SJ, Oliver K, Strata P, Fawcett JW (2006) Composition of perineuronal nets in the adult rat cerebellum and the cellular origin of their components. J Comp Neurol 494(4):559–577. doi:10.1002/cne.20822
Atwal JK, Pinkston-Gosse J, Syken J, Stawicki S, Wu Y, Shatz C, Tessier-Lavigne M (2008) PirB is a functional receptor for myelin inhibitors of axonal regeneration. Science 322(5903):967–970. doi:10.1126/science.1161151
Dickendesher TL, Baldwin KT, Mironova YA, Koriyama Y, Raiker SJ, Askew KL, Wood A, Geoffroy CG et al (2012) NgR1 and NgR3 are receptors for chondroitin sulfate proteoglycans. Nat Neurosci 15(5):703–712. doi:10.1038/nn.3070
McKerracher L (2001) Spinal cord repair: strategies to promote axon regeneration. Neurobiol Dis 8(1):11–18. doi:10.1006/nbdi.2000.0359
Fujita Y, Yamashita T (2014) Axon growth inhibition by RhoA/ROCK in the central nervous system. Front Neurosci 8:338. doi:10.3389/fnins.2014.00338
Huo Y, Yin XL, Ji SX, Zou H, Lang M, Zheng Z, Cai XF, Liu W et al (2013) Inhibition of retinal ganglion cell axonal outgrowth through the Amino-Nogo-A signaling pathway. Neurochem Res 38(7):1365–1374. doi:10.1007/s11064-013-1032-1
Joshi AR, Bobylev I, Zhang G, Sheikh KA, Lehmann HC (2015) Inhibition of Rho-kinase differentially affects axon regeneration of peripheral motor and sensory nerves. Exp Neurol 263:28–38. doi:10.1016/j.expneurol.2014.09.012
Kubo T, Hata K, Yamaguchi A, Yamashita T (2007) Rho-ROCK inhibitors as emerging strategies to promote nerve regeneration. Curr Pharm Des 13(24):2493–2499
Keene JD (2001) Ribonucleoprotein infrastructure regulating the flow of genetic information between the genome and the proteome. Proc Natl Acad Sci U S A 98(13):7018–7024. doi:10.1073/pnas.111145598
Bartel DP, Chen CZ (2004) Micromanagers of gene expression: the potentially widespread influence of metazoan microRNAs. Nat Rev Genet 5(5):396–400. doi:10.1038/nrg1328
Lee RC, Feinbaum RL, Ambros V (1993) The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75(5):843–854
Johnston RJ, Hobert O (2003) A microRNA controlling left/right neuronal asymmetry in Caenorhabditis elegans. Nature 426(6968):845–849. doi:10.1038/nature02255
Belaich MN, Rodriguez VA, Bilen MF, Pilloff MG, Romanowski V, Sciocco-Cap A, Ghiringhelli PD (2006) Sequencing and characterisation of p74 gene in two isolates of Anticarsia gemmatalis MNPV. Virus Genes 32(1):59–70. doi:10.1007/s11262-005-5846-z
Gonzalez-Estevez C, Arseni V, Thambyrajah RS, Felix DA, Aboobaker AA (2009) Diverse miRNA spatial expression patterns suggest important roles in homeostasis and regeneration in planarians. Int J Dev Biol 53(4):493–505. doi:10.1387/ijdb.082825cg
Hutvagner G, Zamore PD (2002) A microRNA in a multiple-turnover RNAi enzyme complex. Science 297(5589):2056–2060. doi:10.1126/science.1073827
Shi M, Ishikawa M, Kamei N, Nakasa T, Adachi N, Deie M, Asahara T, Ochi M (2009) Acceleration of skeletal muscle regeneration in a rat skeletal muscle injury model by local injection of human peripheral blood-derived CD133-positive cells. Stem Cells 27(4):949–960. doi:10.1002/stem.4
Yu YM, Gibbs KM, Davila J, Campbell N, Sung S, Todorova TI, Otsuka S, Sabaawy HE et al (2011) MicroRNA miR-133b is essential for functional recovery after spinal cord injury in adult zebrafish. Eur J Neurosci 33(9):1587–1597. doi:10.1111/j.1460-9568.2011.07643.x
Schwab JM, Conrad S, Monnier PP, Julien S, Mueller BK, Schluesener HJ (2005) Spinal cord injury-induced lesional expression of the repulsive guidance molecule (RGM). Eur J Neurosci 21(6):1569–1576. doi:10.1111/j.1460-9568.2005.03962.x
Erschbamer MK, Hofstetter CP, Olson L (2005) RhoA, RhoB, RhoC, Rac1, Cdc42, and Tc10 mRNA levels in spinal cord, sensory ganglia, and corticospinal tract neurons and long-lasting specific changes following spinal cord injury. J Comp Neurol 484(2):224–233. doi:10.1002/cne.20471
Pixley FJ, Xiong Y, Yu RY, Sahai EA, Stanley ER, Ye BH (2005) BCL6 suppresses RhoA activity to alter macrophage morphology and motility. J Cell Sci 118(Pt 9):1873–1883. doi:10.1242/jcs.02314
Walchli T, Pernet V, Weinmann O, Shiu JY, Guzik-Kornacka A, Decrey G, Yuksel D, Schneider H et al (2013) Nogo-A is a negative regulator of CNS angiogenesis. Proc Natl Acad Sci U S A 110(21):E1943–1952. doi:10.1073/pnas.1216203110
McGee AW, Strittmatter SM (2003) The Nogo-66 receptor: focusing myelin inhibition of axon regeneration. Trends Neurosci 26(4):193–198. doi:10.1016/S0166-2236(03)00062-6
Ridley AJ, Leverrier Y (2001) Requirement for Rho GTPases and PI 3-kinases during apoptotic cell phagocytosis by macrophages. Curr Biol 11(3):195–9
Essler M, Hermann K, Amano M, Kaibuchi K, Heesemann J, Weber PC, Aepfelbacher M (1998) Pasteurella multocida toxin increases endothelial permeability via Rho kinase and myosin light chain phosphatase. J Immunol 161(10):5640–5646
Carbajal JM, Schaeffer RC Jr (1999) RhoA inactivation enhances endothelial barrier function. Am J Physiol 277(5 Pt 1):C955–964
Hordijk PL, Anthony E, Mul FP, Rientsma R, Oomen LC, Roos D (1999) Vascular-endothelial-cadherin modulates endothelial monolayer permeability. J Cell Sci 112(Pt 12):1915–1923
Ridley AJ (2001) Rho proteins, PI 3-kinases, and monocyte/macrophage motility. FEBS Lett 498(2–3):168–171
Persidsky Y, Heilman D, Haorah J, Zelivyanskaya M, Persidsky R, Weber GA, Shimokawa H, Kaibuchi K et al (2006) Rho-mediated regulation of tight junctions during monocyte migration across the blood–brain barrier in HIV-1 encephalitis (HIVE). Blood 107(12):4770–4780. doi:10.1182/blood-2005-11-4721
Dergham P, Ellezam B, Essagian C, Avedissian H, Lubell WD, McKerracher L (2002) Rho signaling pathway targeted to promote spinal cord repair. J Neurosci 22(15):6570–6577
Fournier AE, Takizawa BT, Strittmatter SM (2003) Rho kinase inhibition enhances axonal regeneration in the injured CNS. J Neurosci 23(4):1416–1423
Kamei N, Oishi Y, Tanaka N, Ishida O, Fujiwara Y, Ochi M (2004) Neural progenitor cells promote corticospinal axon growth in organotypic co-cultures. Neuroreport 15(17):2579–2583
Care A, Catalucci D, Felicetti F, Bonci D, Addario A, Gallo P, Bang ML, Segnalini P et al (2007) MicroRNA-133 controls cardiac hypertrophy. Nat Med 13(5):613–618. doi:10.1038/nm1582
Chen J, Wu J, Apostolova I, Skup M, Irintchev A, Kugler S, Schachner M (2007) Adeno-associated virus-mediated L1 expression promotes functional recovery after spinal cord injury. Brain 130(Pt 4):954–969. doi:10.1093/brain/awm049
Drinkut A, Tereshchenko Y, Schulz JB, Bahr M, Kugler S (2012) Efficient gene therapy for Parkinson’s disease using astrocytes as hosts for localized neurotrophic factor delivery. Mol Ther 20(3):534–543. doi:10.1038/mt.2011.249
Steward O, Zheng B, Tessier-Lavigne M (2003) False resurrections: distinguishing regenerated from spared axons in the injured central nervous system. J Comp Neurol 459(1):1–8. doi:10.1002/cne.10593
Curtis R, Green D, Lindsay RM, Wilkin GP (1993) Up-regulation of GAP-43 and growth of axons in rat spinal cord after compression injury. J Neurocytol 22(1):51–64
Basso DM, Fisher LC, Anderson AJ, Jakeman LB, McTigue DM, Popovich PG (2006) Basso Mouse Scale for locomotion detects differences in recovery after spinal cord injury in five common mouse strains. J Neurotrauma 23(5):635–659. doi:10.1089/neu.2006.23.635
Yoo M, Lee GA, Park C, Cohen RI, Schachner M (2014) Analysis of human embryonic stem cells with regulatable expression of the cell adhesion molecule l1 in regeneration after spinal cord injury. J Neurotrauma 31(6):553–564. doi:10.1089/neu.2013.2886
Matsuda T, Cepko CL (2004) Electroporation and RNA interference in the rodent retina in vivo and in vitro. Proc Natl Acad Sci U S A 101(1):16–22. doi:10.1073/pnas.2235688100
Zhu B, Cai G, Hall EO, Freeman GJ (2007) In-fusion assembly: seamless engineering of multidomain fusion proteins, modular vectors, and mutations. Biotechniques 43(3):354–359
Salter MW, Beggs S (2014) Sublime microglia: expanding roles for the guardians of the CNS. Cell 158(1):15–24. doi:10.1016/j.cell.2014.06.008
Ji RR, Xu ZZ, Gao YJ (2014) Emerging targets in neuroinflammation-driven chronic pain. Nat Rev Drug Discov 13(7):533–548. doi:10.1038/nrd4334
Stoolmiller AC, Horwitz AL, Dorfman A (1972) Biosynthesis of the chondroitin sulfate proteoglycan. Purification and properties of xylosyltransferase. J Biol Chem 247(11):3525–3532
Morgenstern DA, Asher RA, Fawcett JW (2002) Chondroitin sulphate proteoglycans in the CNS injury response. Prog Brain Res 137:313–332
Bradbury EJ, Moon LD, Popat RJ, King VR, Bennett GS, Patel PN, Fawcett JW, McMahon SB (2002) Chondroitinase ABC promotes functional recovery after spinal cord injury. Nature 416(6881):636–640. doi:10.1038/416636a
Grimpe B, Silver J (2004) A novel DNA enzyme reduces glycosaminoglycan chains in the glial scar and allows microtransplanted dorsal root ganglia axons to regenerate beyond lesions in the spinal cord. J Neurosci 24(6):1393–1397. doi:10.1523/JNEUROSCI.4986-03.2004
Pietri S, Dimidschstein J, Tiberi L, Sotiropoulou PA, Bilheu A, Goffinet A, Achouri Y, Tissir F et al (2012) Transcriptional mechanisms of EphA7 gene expression in the developing cerebral cortex. Cereb Cortex 22(7):1678–1689. doi:10.1093/cercor/bhr245
Rogers JH, Ciossek T, Ullrich A, West E, Hoare M, Muir EM (1999) Distribution of the receptor EphA7 and its ligands in development of the mouse nervous system. Brain Res Mol Brain Res 74(1–2):225–230
Willson CA, Irizarry-Ramirez M, Gaskins HE, Cruz-Orengo L, Figueroa JD, Whittemore SR, Miranda JD (2002) Upregulation of EphA receptor expression in the injured adult rat spinal cord. Cell Transplant 11(3):229–239
Figueroa JD, Benton RL, Velazquez I, Torrado AI, Ortiz CM, Hernandez CM, Diaz JJ, Magnuson D et al (2006) Inhibition of EphA7 up-regulation after spinal cord injury reduces apoptosis and promotes locomotor recovery. J Neurosci Res 84(7):1438–1451. doi:10.1002/jnr.21048
Le KT, Villeneuve P, Ramjaun AR, McPherson PS, Beaudet A, Seguela P (1998) Sensory presynaptic and widespread somatodendritic immunolocalization of central ionotropic P2X ATP receptors. Neuroscience 83(1):177–190
Schwab JM, Guo L, Schluesener HJ (2005) Spinal cord injury induces early and persistent lesional P2X4 receptor expression. J Neuroimmunol 163(1–2):185–189. doi:10.1016/j.jneuroim.2005.02.016
Li F, Wang L, Li JW, Gong M, He L, Feng R, Dai Z, Li SQ (2011) Hypoxia induced amoeboid microglial cell activation in postnatal rat brain is mediated by ATP receptor P2X4. BMC Neurosci 12:111. doi:10.1186/1471-2202-12-111
Xin H, Li Y, Liu Z, Wang X, Shang X, Cui Y, Zhang ZG, Chopp M (2013) MiR-133b promotes neural plasticity and functional recovery after treatment of stroke with multipotent mesenchymal stromal cells in rats via transfer of exosome-enriched extracellular particles. Stem Cells 31(12):2737–2746. doi:10.1002/stem.1409
Jee MK, Jung JS, Im YB, Jung SJ, Kang SK (2012) Silencing of miR20a is crucial for Ngn1-mediated neuroprotection in injured spinal cord. Hum Gene Ther 23(5):508–520. doi:10.1089/hum.2011.121
Jee MK, Jung JS, Choi JI, Jang JA, Kang KS, Im YB, Kang SK (2012) MicroRNA 486 is a potentially novel target for the treatment of spinal cord injury. Brain 135(Pt 4):1237–1252. doi:10.1093/brain/aws047
Bhalala OG, Pan L, Sahni V, McGuire TL, Gruner K, Tourtellotte WG, Kessler JA (2012) microRNA-21 regulates astrocytic response following spinal cord injury. J Neurosci 32(50):17935–17947. doi:10.1523/JNEUROSCI.3860-12.2012
Acknowledgments
We thank Ohyoon Kwon for contributing to the BMS scoring and Natasha Khatri for help with formatting. We are grateful to the New Jersey Commission on Spinal Cord Research for funding this project (CSCR12IRG014).
Author information
Authors and Affiliations
Corresponding author
Additional information
Thomas Theis and Myung Yoo contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Transfection efficiencies of the pCMV constructs are not altered by different culture conditions. (a) Representative images of N2A cells co-transfected with pCMV-GFP-133b and empty pmirGlo plasmids (−), pmirGlo-Xylt1 (Xylt1), pmirGlo-Epha7 (Epha7), pmirGlo-P2RX4 (P2RX4), or pmirGlo-RhoA (RhoA) constructs. Scale bar, 10 μm. (b) The bar diagram shows the percentage of GFP-positive cells among all cells. (TIF 41,658 kb)
Rights and permissions
About this article
Cite this article
Theis, T., Yoo, M., Park, C.S. et al. Lentiviral Delivery of miR-133b Improves Functional Recovery After Spinal Cord Injury in Mice. Mol Neurobiol 54, 4659–4671 (2017). https://doi.org/10.1007/s12035-016-0007-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-0007-z