Abstract
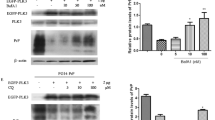
Autophagy is an important protein degradation pathway and a part of the innate immune system that is activated in the brain tissue during animal and human prion diseases. However, the possible mechanism by which prion infection triggers autophagy and the significance of activated autophagy on prion accumulation remain unknown. Here, we demonstrated that autophagic flux was enhanced in the persistent prion-infected cell line, SMB-S15. Knockdown of ATG5 and the presence of three autophagic inhibitors resulted in a significant increase of PrPSc. The mammalian target of rapamycin (MTOR) levels in SMB-S15 cells were also markedly decreased, in direct relation to PrPSc accumulation. F-box and WD repeat domain containing 7 (FBXW7) levels in SMB-S15 cells and in the brains of scrapie-agent 263K-infected hamsters were upregulated at the early stage of infection, leading to active ubiquitination and degradation of MTOR. Knockdown of FBXW7 in SMB-S15 cells remarkably inhibited autophagic flux and increased PrPSc accumulation. Thus, we conclude that prion infection induced the expression of FBXW7, which mediated MTOR ubiquitination and degradation, further altering phosphorylation status through cross talk between MTORC1 and AMPK and increasing autophagic flux. Autophagy may serve as innate immunity to degrade PrPSc and maintain prion homeostasis.











Similar content being viewed by others
References
Prusiner SB (1998) Prions. Proc Natl Acad Sci U S A 95(23):13363–13383
Wang F, Wang X, Yuan CG, Ma J (2010) Generating a prion with bacterially expressed recombinant prion protein. Science 327(5969):1132–1135. doi:10.1126/science.1183748
Nunziante M, Ackermann K, Dietrich K, Wolf H, Gadtke L, Gilch S, Vorberg I, Groschup M, Schatzl HM (2011) Proteasomal dysfunction and endoplasmic reticulum stress enhance trafficking of prion protein aggregates through the secretory pathway and increase accumulation of pathologic prion protein. J Biol Chem 286(39):33942–33953. doi:10.1074/jbc.M111.272617
Xu Y, Tian C, Wang SB, Xie WL, Guo Y, Zhang J, Shi Q, Chen C, Dong XP (2012) Activation of the macroautophagic system in scrapie-infected experimental animals and human genetic prion diseases. Autophagy 8(11):1604–1620. doi:10.4161/auto.21482
Rubinsztein DC (2006) The roles of intracellular protein-degradation pathways in neurodegeneration. Nature 443(7113):780–786. doi:10.1038/nature05291
Zhao Y, Li X, Cai MY, Ma K, Yang J, Zhou J, Fu W, Wei FZ, Wang L, Xie D, Zhu WG (2013) XBP-1u suppresses autophagy by promoting the degradation of FoxO1 in cancer cells. Cell Res 23(4):491–507. doi:10.1038/cr.2013.2
Zhang Y, Yan L, Zhou Z, Yang P, Tian E, Zhang K, Zhao Y, Li Z, Song B, Han J, Miao L, Zhang H (2009) SEPA-1 mediates the specific recognition and degradation of P granule components by autophagy in C. elegans. Cell 136(2):308–321. doi:10.1016/j.cell.2008.12.022
Pattingre S, Tassa A, Qu X, Garuti R, Liang XH, Mizushima N, Packer M, Schneider MD, Levine B (2005) Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell 122(6):927–939. doi:10.1016/j.cell.2005.07.002
Ravikumar B, Vacher C, Berger Z, Davies JE, Luo S, Oroz LG, Scaravilli F, Easton DF, Duden R, O’Kane CJ, Rubinsztein DC (2004) Inhibition of mTOR induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. Nat Genet 36(6):585–595. doi:10.1038/ng1362
Ravikumar B, Sarkar S, Davies JE, Futter M, Garcia-Arencibia M, Green-Thompson ZW, Jimenez-Sanchez M, Korolchuk VI, Lichtenberg M, Luo S, Massey DC, Menzies FM, Moreau K, Narayanan U, Renna M, Siddiqi FH, Underwood BR, Winslow AR, Rubinsztein DC (2010) Regulation of mammalian autophagy in physiology and pathophysiology. Physiol Rev 90(4):1383–1435. doi:10.1152/physrev.00030.2009
Boellaard JW, Kao M, Schlote W, Diringer H (1991) Neuronal autophagy in experimental scrapie. Acta Neuropathol 82(3):225–228
Sikorska B, Liberski PP, Giraud P, Kopp N, Brown P (2004) Autophagy is a part of ultrastructural synaptic pathology in Creutzfeldt-Jakob disease: a brain biopsy study. Int J Biochem Cell Biol 36(12):2563–2573. doi:10.1016/j.biocel.2004.04.014
Aguib Y, Heiseke A, Gilch S, Riemer C, Baier M, Schatzl HM, Ertmer A (2009) Autophagy induction by trehalose counteracts cellular prion infection. Autophagy 5(3):361–369
Heiseke A, Aguib Y, Riemer C, Baier M, Schatzl HM (2009) Lithium induces clearance of protease resistant prion protein in prion-infected cells by induction of autophagy. J Neurochem 109(1):25–34. doi:10.1111/j.1471-4159.2009.05906.x
Doh-Ura K, Iwaki T, Caughey B (2000) Lysosomotropic agents and cysteine protease inhibitors inhibit scrapie-associated prion protein accumulation. J Virol 74(10):4894–4897
Birkett CR, Hennion RM, Bembridge DA, Clarke MC, Chree A, Bruce ME, Bostock CJ (2001) Scrapie strains maintain biological phenotypes on propagation in a cell line in culture. EMBO J 20(13):3351–3358. doi:10.1093/emboj/20.13.3351
Kimura S, Noda T, Yoshimori T (2007) Dissection of the autophagosome maturation process by a novel reporter protein, tandem fluorescent-tagged LC3. Autophagy 3(5):452–460
Klionsky DJ, Abdalla FC, Abeliovich H, Abraham RT, Acevedo-Arozena A, Adeli K, Agholme L, Agnello M et al (2012) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 8(4):445–544
Kim J, Kundu M, Viollet B, Guan KL (2011) AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol 13(2):132–141. doi:10.1038/ncb2152
Egan DF, Shackelford DB, Mihaylova MM, Gelino S, Kohnz RA, Mair W, Vasquez DS, Joshi A, Gwinn DM, Taylor R, Asara JM, Fitzpatrick J, Dillin A, Viollet B, Kundu M, Hansen M, Shaw RJ (2011) Phosphorylation of ULK1 (hATG1) by AMP-activated protein kinase connects energy sensing to mitophagy. Science 331(6016):456–461. doi:10.1126/science.1196371
Long X, Lin Y, Ortiz-Vega S, Yonezawa K, Avruch J (2005) Rheb binds and regulates the mTOR kinase. Curr Biol 15(8):702–713. doi:10.1016/j.cub.2005.02.053
Mao JH, Kim IJ, Wu D, Climent J, Kang HC, DelRosario R, Balmain A (2008) FBXW7 targets mTOR for degradation and cooperates with PTEN in tumor suppression. Science 321(5895):1499–1502. doi:10.1126/science.1162981
Sir D, Ou JH (2010) Autophagy in viral replication and pathogenesis. Mol Cells 29(1):1–7. doi:10.1007/s10059-010-0014-2
Welcker M, Clurman BE (2008) FBW7 ubiquitin ligase: a tumour suppressor at the crossroads of cell division, growth and differentiation. Nat Rev Cancer 8(2):83–93. doi:10.1038/nrc2290
Laplante M, Sabatini DM (2012) mTOR signaling in growth control and disease. Cell 149(2):274–293. doi:10.1016/j.cell.2012.03.017
Li X, Alafuzoff I, Soininen H, Winblad B, Pei JJ (2005) Levels of mTOR and its downstream targets 4E-BP1, eEF2, and eEF2 kinase in relationships with tau in Alzheimer’s disease brain. FEBS J 272(16):4211–4220. doi:10.1111/j.1742-4658.2005.04833.x
Narita M, Young AR, Arakawa S, Samarajiwa SA, Nakashima T, Yoshida S, Hong S, Berry LS, Reichelt S, Ferreira M, Tavare S, Inoki K, Shimizu S (2011) Spatial coupling of mTOR and autophagy augments secretory phenotypes. Science 332(6032):966–970. doi:10.1126/science.1205407
Salvador N, Aguado C, Horst M, Knecht E (2000) Import of a cytosolic protein into lysosomes by chaperone-mediated autophagy depends on its folding state. J Biol Chem 275(35):27447–27456. doi:10.1074/jbc.M001394200
Caughey B, Baron GS (2006) Prions and their partners in crime. Nature 443(7113):803–810. doi:10.1038/nature05294
Sandberg MK, Al-Doujaily H, Sharps B, Clarke AR, Collinge J (2011) Prion propagation and toxicity in vivo occur in two distinct mechanistic phases. Nature 470(7335):540–542. doi:10.1038/nature09768
Bueler H, Raeber A, Sailer A, Fischer M, Aguzzi A, Weissmann C (1994) High prion and PrPSc levels but delayed onset of disease in scrapie-inoculated mice heterozygous for a disrupted PrP gene. Mol Med 1(1):19–30
Hill AF, Collinge J (2003) Subclinical prion infection. Trends Microbiol 11(12):578–584
Moreno JA, Radford H, Peretti D, Steinert JR, Verity N, Martin MG, Halliday M, Morgan J, Dinsdale D, Ortori CA, Barrett DA, Tsaytler P, Bertolotti A, Willis AE, Bushell M, Mallucci GR (2012) Sustained translational repression by eIF2alpha-P mediates prion neurodegeneration. Nature 485(7399):507–511. doi:10.1038/nature11058
Acknowledgments
This work was supported by Chinese National Natural Science Foundation Grant (81100980, 81101302 and 31100117), China Mega-Project for Infectious Disease (2011ZX10004-101, 2012ZX10004215), and SKLID Development Grant (2012SKLID102, 2011SKLID302, 2011SKLID204, 2011SKLID211).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(PPTX 206 kb)
Rights and permissions
About this article
Cite this article
Xu, Y., Tian, C., Sun, J. et al. FBXW7-Induced MTOR Degradation Forces Autophagy to Counteract Persistent Prion Infection. Mol Neurobiol 53, 706–719 (2016). https://doi.org/10.1007/s12035-014-9028-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-9028-7