Abstract
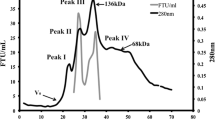
Phytase (myo-inositol hexaphosphate phosphohydrolase) belongs to phosphatases. It catalyzes the hydrolysis of phytate to less-phosphorylated inorganic phosphates and phytate. Phytase is used primarily for the feeding of simple hermit animals in order to increase the usability of amino acids, minerals, phosphorus and energy. In the present study, phytase isolation from the Lactobacillus coryniformis strain, isolated from Lor cheese sources, phytase purification and characterization were studied. The phytase was purified in simple three steps. The enzyme was obtained with 2.60% recovery and a specific activity of 202.25 (EU/mg protein). The molecular mass of the enzyme was determined to be 43.25 kDa with the sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) method. The optimum temperature and pH for the enzyme were found as 60 °C and 5.0 and respectively. To defined the substrate specificity of the phytase, the hydrolysis of several phosphorylated compounds by the purified enzyme was studied and sodium phytate showed high specificity. Furthermore, the effects of Ca2+, Ag+, Mg2+, Cu2+, Co2+, Pb2+, Zn2+ and Ni2+ metal ions on the enzyme were studied.




Similar content being viewed by others
References
Filannino, P., Di Cagno, R., & Gobbetti, M. (2018). Metabolic and functional paths of lactic acid bacteria in plant foods: Get out of the labyrinth. Current Opinion in Biotechnology, 49, 64–72.
Papadimitriou, K., Alegría, Á, Bron, P. A., et al. (2016). Stress physiology of lactic acid bacteria. Microbiology and Molecular Biology Reviews, 80(3), 837–890.
Mendes, R., Garbeva, P., & Raaijmakers, J. M. (2013). The rhizosphere microbiome: Significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. FEMS Microbiology Reviews, 37, 634–663.
De Angelis, M., Gallo, G., Corbo, M. R., McSweeney, P. L., Faccia, M., Giovine, M., et al. (2003). Phytase activity in sourdough lactic acid bacteria: Purification and characterization of a phytase from Lactobacillus sanfranciscensis CB1. International Journal of Food Microbiology, 87, 259–270.
Urbano, G., Lopez-Jurado, M., Aranda, P., Vidal-Valverde, C., Tenorio, E., & Porres, J. (2000). The role of phytic acid in legumes: Antinutrient or beneficial function? Journal of Physiology and Biochemistry, 56, 283–294.
Diouf-Lewis, A., Commereuc, S., & Verney, V. (2017). Toward greener polyolefins: Antioxidant effect of phytic acid from cereal waste. European Polymer Journal, 96, 190–199.
Gülçin, I., & Beydemir, Ş (2013). Phenolic compounds as antioxidants: Carbonic anhydrase isoenzymes inhibitors. Mini-Reviews in Medicinal Chemistry, 13(3), 408–430.
Gülçin, I. (2012). Antioxidant activity of food constituents: An overview. Archives of Toxicology, 86(3), 345–391.
Singh, B., & Satyanarayana, T. (2009). Characterization of a HAP—phytase from a thermophilic mould Sporotrichum thermophile. Bioresource Technology, 100, 2046–2051.
Bohn, L., Meyer, A. S., & Rasmussen, S. K. (2008). Phytate: Impact on environment and human nutrition. A challenge for molecular breeding. Journal of Zhejiang University Science B, 9(3), 165–191.
Kalsi, H. K., Singh, R., Dhaliwal, H. S., & Kumar, V. (2016). Phytases from Enterobacter and Serratia species with desirable characteristics for food and feed applications. Biotechnology, 6, 1–13.
Demir, Y., Şenol Kotan, M., Dikbas, N., & Beydemir, S. (2017). Phytase from Weissella halotolerans: Purification, partial characterisation and the effect of some metals. International Journal of Food Properties, 20(2), 2127–2137.
Bischoff, K. M., Rooney, A. P., Li, X. L., Liu, S., & Hughes, R. (2006). Purification and characterization of a family 5 Endoglucanase from a moderately thermophilic strain of Bacillus Licheniformis. Biotechnology Letters, 28, 1761–1765.
Adiguzel, A., Ozkan, H., Baris, O., Inan, K., Gulluce, M., & Sahin, F. (2009). Identification and characterization of thermophilic bacteria Isolated from Hot Springs in Turkey. Journal of Microbiological Methods, 79, 321–328.
Altschul, S. F., Gish, W., Miller, W., Myers, E. W., & Lipman, D. J. (1990). Basic local alignment search tool. Journal of Molecular Biology, 215, 403–410.
Beydemir, S., & Demir, Y. (2016). Antiepileptic drugs: Impacts on human serum paraoxonase-1. Journal of Biochemical and Molecular Toxicology, 31(6), e21889.
Alım, Z., & Beydemir, S. (2016). Some anticancer agents act on human serum Paraoxonase-1 to reduce its activity. Chemical Biology and Drug Design, 88(2), 188–196.
Zou, L. K., Wang, H. N., Pan, X., Tian, G. B., Xie, Z. W., Wu, Q., et al. (2008). Expression, purification and characterization of a phyAm – phyCs fusion phytase. Journal of Zhejiang University Science B, 9, 536–545.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Analytical Biochemistry, 72, 248–254.
Laemmli, U. K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227, 680–685.
Demir, Y., & Beydemir, S. (2015). Purification, refolding, and characterization of recombinant human paraoxonase-1. Turkish Journal of Chemistry, 39, 764–776.
Sarıbuga, E., Nadaroglu, H., Dikbas, N., Senol, M., & Cetin, B. (2014). Purification, characterization of phytase enzyme from Lactobacillus Plantarum bacteria and determination of its kinetic properties. African Journal of Biotechnology, 13(23), 2373–2378.
Gülçin, I., Küfrevioğlu, O. I., & Oktay, M. (2005). Purification and characterization of polyphenol oxidase from nettle (Urtica dioica L.) and inhibitory effects of some chemicals on enzyme activity. Journal of Enzyme Inhibition and Medicinal Chemistry, 20(3), 297–302.
Oh, B. C., Choi, W. C., Park, S., Kim, Y. O., & Oh, T. K. (2004). Biochemical properties and substrate specificities of alkaline and histidine acid phytases. Applied Microbiology and Biotechnology, 63(4), 362–372.
Vohra, A., & Satyanarayana, T. (2003). Phytases: Microbial sources, production, purification, and potential biotechnological applications. Critical Reviews in Biotechnology, 23(1), 29–60.
Boukhris, I., Farhat-Khemakhem, A., Blibech, M., Bouchaala, K., & Chouayekh, H. (2015). Characterization of an extremely salt-tolerant and thermostable phytase from Bacillus amyloliquefaciens US573. International Journal of Biological Macromolecules, 80, 581–587.
Escobin-Mopera, L., Ohtani, M., Sekiguchi, S., Sone, T., Abe, A., Tanaka, M., et al. (2012). Purification and characterization of phytase from Klebsiella pneumoniae 9-3B. Journal of Bioscience and Bioengineering, 113(5), 562–567.
Parhamfar, M., Badoei-Dalfard, A., Khaleghi, M., & Hassanshahian, M. (2015). Purification and characterization of an acidic, thermophilic phytase from a newly isolated Geobacillus stearothermophilus strain DM12. Program in Biological Sciences, 5(1), 61–73.
Choi, Y. M., Suh, H. J., & Kim, J. M. (2001). Purification and properties of extracellular phytase from Bacillus sp. KHU-10. Journal of Protein Chemistry, 20(4), 287–292.
Konietzny, U., & Greiner, R. (2002). Molecular and catalytic properties of phytate-degrading enzymes (phytases). International Journal of Food Science & Technology, 37, 791–812.
Herter, T., Berezina, O. V., Zinin, N. V., Velikodvorskaya, G. A., Greiner, R., & Borriss, R. (2006). Glucose-1-phosphatase (AgpE) from Enterobacter cloacae displays enhanced phytase activity. Applied Microbiology and Biotechnology, 70, 60–64.
Wyss, M., Brugger, R., Kronenberger, A., Remy, R., Fimbel, R., Oesterhelt, G., et al. (1999). Biochemical characterization of fungal phytases (myo-inositol hexakisphosphate phosphohydrolases): Catalytic properties. Applied and Environmental Microbiology, 65, 367–373.
Roy, M. P., Poddar, M., Singh, K. K., & Ghosh, S. (2012). Purification, characterization and properties of phytase from Shigella sp. CD2. Indian Journal of Biochemistry & Biophysics, 49(4), 266–271.
Fu, S., Sun, J., Qian, L., & Li, Z. (2008). Bacillus phytases: Present scenario and future perspectives. Applied Biochemistry and Biotechnology, 151, 1–8.
Boyce, A., & Walsh, G. (2007). Purification and characterisation of an acid phosphatase with phytase activity from Mucor hiemalis Wehmer. Journal of Biotechnology, 132(1), 82–87.
Fujita, J., Yamane, Y. I., Fukuda, H., Kizaki, Y., Wakabayashi, S., Shigeta, S., et al. (2003). Production and properties of phytase and acid phosphatase from a sake koji mold, Aspergillus oryzae. Journal of Bioscience and Bioengineering, 95(4), 348–353.
Greiner, R., & Konietzny, U. (2006). Phytase for food application. Food Technology and Biotechnology, 44(2), 125–140.
Quan, C. S., Tian, W. J., Fan, S. D., & Kikuchi, J. I. (2004). Purification and properties of a low- molecular-weight phytase from Cladosporium sp. FP-1. Journal of Bioscience and Bioengineering, 97(4), 260–266.
In, M. J., Seo, S. W., Kim, D. C., & Oh, N. S. (2009). Purification and biochemical properties of an extracellular acid phytase produced by the Saccharomyces cerevisiae CY strain. Process Biochemistry, 44(1), 122–126.
Haros, M., Bielecka, M., Honke, J., & Sanz, Y. (2008). Phytate-degrading activity in lactic acid bacteria. Polish Journal of Food and Nutrition Sciences, 58, 33–40.
Kücük, M., & Gulcin, I. (2016). Purification and characterization of the carbonic anhydrase enzyme from black sea trout (Salmo trutta Labrax Coruhensis) kidney and inhibition effects of some metal ions on enzyme activity. Environmental Toxicology and Pharmacology, 44, 134–139.
Kocyigit, U. M., Taslimi, P., & Gulçin, İ (2018). Characterization and inhibition effects of some metal ions on carbonic anhydrase enzyme from Kangal Akkaraman sheep. Journal of Biochemical and Molecular Toxicology. https://doi.org/10.1002/jbt.22172.
Bajaj, B. K., & Manhas, K. (2012). Production and characterization of xylanase from Bacillus Licheniformis P11(C) with potential for fruit juice and bakery industry. Biocatalysis and Agricultural Biotechnology, 1(4), 330–337.
Yu, P., & Chen, Y. (2013). Purification and characterization of a novel neutral and heat-tolerant phytase from a newly isolated strain Bacillus nealsonii ZJ0702. BMC Biotechnology, 13(78), 1–7.
Bajaj, B. K., & Wani, A. M. (2015). Purification and characterization of a novel phytase from Nocardia sp. MB 36. Biocatalysis and Biotransformation, 33(3), 141–149.
Hong, S. W., Chu, I. H., & Chung, K. S. (2011). Purification and biochemical characterization of thermostable phytase from newly isolated Bacillus subtilis CF92. Journal of the Korean Society for Applied Biological Chemistry, 54, 89–94.
Gulati, H. K., Chadha, B. S., & Saini, H. S. (2007). Production and characterization of thermostable alkaline phytase from Bacillus laevolacticus isolated from rhizosphere soil. Journal of Industrial Microbiology and Biotechnology, 34, 91–98.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Demir, Y., Dikbaş, N. & Beydemir, Ş. Purification and Biochemical Characterization of Phytase Enzyme from Lactobacillus coryniformis (MH121153). Mol Biotechnol 60, 783–790 (2018). https://doi.org/10.1007/s12033-018-0116-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-018-0116-1