Abstract
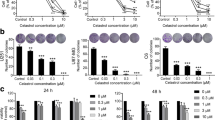
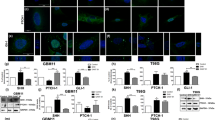
WAC is closely related to the occurrence and development of tumors. However, its role in human glioblastoma (GBM) and its potential regulatory mechanisms have not been investigated. This study demonstrated that WAC is downregulated in GBM, and its low expression predicts a poor prognosis. We investigated the effect of WAC on the proliferation of glioma cells through a CCK-8 assay, EdU incorporation, and cell formation. The effects of WAC on apoptosis and autophagy in glioma were determined by flow cytometry, TUNEL detection, immunofluorescence, q-PCR, WB, and scanning electron microscopy. We found that overexpression of WAC inhibited the proliferation of glioma cells, promoted apoptosis, and induced autophagy. Therefore, WAC is likely to play a role as a new regulatory molecule in glioma.





Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, et al. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007;114(2):97–109. https://doi.org/10.1007/s00401-007-0243-4.
Biernat W, Tohma Y, Yonekawa Y, Kleihues P, Ohgaki H. Alterations of cell cycle regulatory genes in primary (de novo) and secondary glioblastomas. Acta Neuropathol. 1997;94(4):303–9. https://doi.org/10.1007/s004010050711.
Ostrom QT, Bauchet L, Davis FG, Deltour I, Fisher JL, Langer CE, et al. The epidemiology of glioma in adults: a “state of the science” review. Neuro Oncol. 2014;16(7):896–913. https://doi.org/10.1093/neuonc/nou087.
Ostrom QT, Gittleman H, Stetson L, Virk SM, Barnholtz-Sloan JS. Epidemiology of gliomas. Cancer Treat Res. 2015;163:1–14. https://doi.org/10.1007/978-3-319-12048-5_1.
Ito M, Minamiya Y, Kawai H, Saito S, Saito H, Nakagawa T, et al. Tumor-derived TGFbeta-1 induces dendritic cell apoptosis in the sentinel lymph node. J Immunol (Baltimore, Md: 1950). 2006;176(9):5637–43. https://doi.org/10.4049/jimmunol.176.9.5637.
Feng Y, He D, Yao Z, Klionsky DJ. The machinery of macroautophagy. Cell Res. 2014;24(1):24–41. https://doi.org/10.1038/cr.2013.168.
Kimmelman AC, White E. Autophagy and tumor metabolism. Cell Metab. 2017;25(5):1037–43. https://doi.org/10.1016/j.cmet.2017.04.004.
Abdul Rahim SA, Dirkse A, Oudin A, Schuster A, Bohler J, Barthelemy V, et al. Regulation of hypoxia-induced autophagy in glioblastoma involves ATG9A. Br J Cancer. 2017;117(6):813–25. https://doi.org/10.1038/bjc.2017.263.
Choi AM, Ryter SW, Levine B. Autophagy in human health and disease. N Engl J Med. 2013;368(7):651–62. https://doi.org/10.1056/NEJMra1205406.
Xu GM, Arnaout MA. WAC, a novel WW domain-containing adapter with a coiled-coil region, is colocalized with splicing factor SC35. Genomics. 2002;79(1):87–94. https://doi.org/10.1006/geno.2001.6684.
Hein C, Springael JY, Volland C, Haguenauer-Tsapis R, André B. NPl1, an essential yeast gene involved in induced degradation of Gap1 and Fur4 permeases, encodes the Rsp5 ubiquitin-protein ligase. Mol Microbiol. 1995;18(1):77–87. https://doi.org/10.1111/j.1365-2958.1995.mmi_18010077.x.
Staub O, Dho S, Henry P, Correa J, Ishikawa T, McGlade J, et al. WW domains of Nedd4 bind to the proline-rich PY motifs in the epithelial Na+ channel deleted in Liddle’s syndrome. EMBO J. 1996;15(10):2371–80.
Maleszka R, Lupas A, Hanes SD, Miklos GL. The dodo gene family encodes a novel protein involved in signal transduction and protein folding. Gene. 1997;203(2):89–93. https://doi.org/10.1016/s0378-1119(97)00522-2.
Sudol M, Sliwa K, Russo T. Functions of WW domains in the nucleus. FEBS Lett. 2001;490(3):190–5. https://doi.org/10.1016/s0014-5793(01)02122-6.
Kao HY, Siliciano PG. Identification of Prp40, a novel essential yeast splicing factor associated with the U1 small nuclear ribonucleoprotein particle. Mol Cell Biol. 1996;16(3):960–7. https://doi.org/10.1128/mcb.16.3.960.
Zhang F, Yu X. WAC, a functional partner of RNF20/40, regulates histone H2B ubiquitination and gene transcription. Mol Cell. 2011;41(4):384–97. https://doi.org/10.1016/j.molcel.2011.01.024.
Abbas T, Dutta A. p21 in cancer: intricate networks and multiple activities. Nat Rev Cancer. 2009;9(6):400–14. https://doi.org/10.1038/nrc2657.
David-Morrison G, Xu Z, Rui YN, Charng WL, Jaiswal M, Yamamoto S, et al. WAC regulates mTOR activity by acting as an adaptor for the TTT and Pontin/Reptin complexes. Dev Cell. 2016;36(2):139–51. https://doi.org/10.1016/j.devcel.2015.12.019.
Joachim J, Jefferies HB, Razi M, Frith D, Snijders AP, Chakravarty P, et al. Activation of ULK kinase and autophagy by GABARAP trafficking from the centrosome is regulated by WAC and GM130. Mol Cell. 2015;60(6):899–913. https://doi.org/10.1016/j.molcel.2015.11.018.
Maiuri MC, Zalckvar E, Kimchi A, Kroemer G. Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat Rev Mol Cell Biol. 2007;8(9):741–52. https://doi.org/10.1038/nrm2239.
Jiang T, Mao Y, Ma W, Mao Q, You Y, Yang X, et al. CGCG clinical practice guidelines for the management of adult diffuse gliomas. Cancer Lett. 2016;375(2):263–73. https://doi.org/10.1016/j.canlet.2016.01.024.
Naldini L. Gene therapy returns to centre stage. Nature. 2015;526(7573):351–60. https://doi.org/10.1038/nature15818.
Klingler S, Guo B, Yao J, Yan H, Zhang L, Vaseva AV, et al. Development of resistance to EGFR-targeted therapy in malignant glioma can occur through EGFR-dependent and -independent mechanisms. Cancer Res. 2015;75(10):2109–19. https://doi.org/10.1158/0008-5472.Can-14-3122.
Ahn JH, Lee YW, Ahn SK, Lee M. Oncogenic BRAF inhibitor UAI-201 induces cell cycle arrest and autophagy in BRAF mutant glioma cells. Life Sci. 2014;104(1–2):38–46. https://doi.org/10.1016/j.lfs.2014.03.026.
Geraldo LHM, Garcia C, da Fonseca ACC, Dubois LGF, de Sampaio ESTCL, Matias D, et al. Glioblastoma therapy in the age of molecular medicine. Trends Cancer. 2019;5(1):46–65. https://doi.org/10.1016/j.trecan.2018.11.002.
Wang C, Zhao N, Zheng Q, Zhang D, Liu Y. BHLHE41 promotes U87 and U251 cell proliferation via ERK/cyclinD1 signaling pathway. Cancer Manage Res. 2019;11:7657–72. https://doi.org/10.2147/cmar.S214697.
Green DR, Kroemer G. The pathophysiology of mitochondrial cell death. Science (New York, NY). 2004;305(5684):626–9. https://doi.org/10.1126/science.1099320.
Gómez-Crisóstomo NP, López-Marure R, Zapata E, Zazueta C, Martínez-Abundis E. Bax induces cytochrome c release by multiple mechanisms in mitochondria from MCF7 cells. J Bioenerg Biomembr. 2013;45(5):441–8. https://doi.org/10.1007/s10863-013-9508-x.
Doherty J, Baehrecke EH. Life, death and autophagy. Nat Cell Biol. 2018;20(10):1110–7. https://doi.org/10.1038/s41556-018-0201-5.
Denton D, Kumar S. Autophagy-dependent cell death. Cell Death Differ. 2019;26(4):605–16. https://doi.org/10.1038/s41418-018-0252-y.
Acknowledgements
The authors thank the personnel in the department of neurosurgery and the central laboratory at the Renmin Hospital of Wuhan University.
Funding
No fundings.
Author information
Authors and Affiliations
Contributions
BL and DT designed the research. YW and SZ carried out all the experimental work. FY, QS, LZ, ZY wrote the paper; YL, RW collected and assembled data; PH and HJ analyzed the data. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The following information was supplied related to ethical approvals (i.e., approving body and any reference numbers): Institutional Ethics Committee of the Faculty of Medicine at Renmin Hospital of Wuhan University approval (2012LKSZ (010) H) to carry out the study within its facilities.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12032_2021_1580_MOESM1_ESM.tif
Supplementary Fig. S1 WAC mRNA expression in different WHO grade according to TCGA, Gravendell and Rembrandt datasets. ns, no significance. ***P < 0.001. (TIF 208 kb)
Rights and permissions
About this article
Cite this article
Wang, Y., Zhang, S., Sun, Q. et al. WAC, a novel GBM tumor suppressor, induces GBM cell apoptosis and promotes autophagy. Med Oncol 38, 132 (2021). https://doi.org/10.1007/s12032-021-01580-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-021-01580-0