Abstract
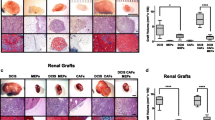
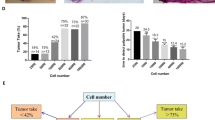
During the past decades, many efforts have been made in mimicking the clinical progress of human cancer in mouse models. Previously, we developed a human breast tissue-derived (HB) mouse model. Theoretically, it may mimic the interactions between “species-specific” mammary microenvironment of human origin and human breast cancer cells. However, detailed evidences are absent. The present study (in vivo, cellular, and molecular experiments) was designed to explore the regulatory role of human mammary microenvironment in the progress of human breast cancer cells. Subcutaneous (SUB), mammary fat pad (MFP), and HB mouse models were developed for in vivo comparisons. Then, the orthotopic tumor masses from three different mouse models were collected for primary culture. Finally, the biology of primary cultured human breast cancer cells was compared by cellular and molecular experiments. Results of in vivo mouse models indicated that human breast cancer cells grew better in human mammary microenvironment. Cellular and molecular experiments confirmed that primary cultured human breast cancer cells from HB mouse model showed a better proliferative and anti-apoptotic biology than those from SUB to MFP mouse models. Meanwhile, primary cultured human breast cancer cells from HB mouse model also obtained the migratory and invasive biology for “species-specific” tissue metastasis to human tissues. Comprehensive analyses suggest that “species-specific” mammary microenvironment of human origin better regulates the biology of human breast cancer cells in our humanized mouse model of breast cancer, which is more consistent with the clinical progress of human breast cancer.





Similar content being viewed by others
References
Liotta LA, Kohn EC. The microenvironment of the tumour-host interface. Nature. 2001;411:375–9.
Mueller MM, Fusenig NE. Friends or foes—bipolar effects of the tumour stroma in cancer. Nat Rev Cancer. 2004;4:839–49.
Tlsty TD, Coussens LM. Tumor stroma and regulation of cancer development. Annu Rev Pathol. 2006;1:119–50.
Bissell MJ, Hines WC. Why don’t we get more cancer? A proposed role of the microenvironment in restraining cancer progression. Nat Med. 2011;17:320–9.
Place AE, Jin HS, Polyak K. The microenvironment in breast cancer progression: biology and implications for treatment. Breast Cancer Res. 2011;13:227.
Swartz MA, Iida N, Roberts EW, Sangaletti S, Wong MH, Yull FE, Coussens LM, DeClerck YA. Tumor microenvironment complexity: emerging roles in cancer therapy. Cancer Res. 2012;72:2473–80.
Allred DC, Medina D. The relevance of mouse models to understanding the development and progression of human breast cancer. J Mammary Gland Biol Neoplasia. 2008;13:279–88.
Borowsky AD. Choosing a mouse model: experimental biology in context—the utility and limitations of mouse models of breast cancer. Cold Spring Harb Perspect Biol. 2011;3:a009670.
Roberts TG Jr, Goulart BH, Squitieri L, Stallings SC, Halpern EF, Chabner BA, Gazelle GS, Finkelstein SN, Clark JW. Trends in the risks and benefits to patients with cancer participating in phase 1 clinical trials. JAMA. 2004;292:2130–40.
Macchiarini F, Manz MG, Palucka AK, Shultz LD. Humanized mice: are we there yet. J Exp Med. 2005;202:1307–11.
Sierra A. Animal models of breast cancer for the study of pathogenesis and therapeutic insights. Clin Transl Oncol. 2009;11:721–7.
Chen H, Chen TH, Tseng TF, Lu JT, Kuo CC, Fu SC, Lee WJ, Tsai YF, Huang YY, Chuang EY, Hwang YJ, Sun CK. High-sensitivity in vivo THz transmission imaging of early human breast cancer in a subcutaneous xenograft mouse model. Opt Express. 2011;19:21552–62.
Potter SM, Dwyer RM, Hartmann MC, Khan S, Boyle MP, Curran CE, Kerin MJ. Influence of stromal–epithelial interactions on breast cancer in vitro and in vivo. Breast Cancer Res Treat. 2012;131:401–11.
Kodera Y, Katanasaka Y, Kitamura Y, Tsuda H, Nishio K, Tamura T, Koizumi F. Sunitinib inhibits lymphatic endothelial cell functions and lymph node metastasis in a breast cancer model through inhibition of vascular endothelial growth factor receptor 3. Breast Cancer Res. 2011;13:R66.
Monsky WL, Mouta CC, Tsuzuki Y, Gohongi T, Fukumura D, Jain RK. Role of host microenvironment in angiogenesis and microvascular functions in human breast cancer xenografts: mammary fat pad versus cranial tumors. Clin Cancer Res. 2002;8:1008–13.
Iorns E, Clarke J, Ward T, Dean S, Lippman M. Simultaneous analysis of tumor and stromal gene expression profiles from xenograft models. Breast Cancer Res Treat. 2012;131:321–4.
Dang TT, Prechtl AM, Pearson GW. Breast cancer subtype-specific interactions with the microenvironment dictate mechanisms of invasion. Cancer Res. 2011;71:6857–66.
Salameh TS, Le TT, Nichols MB, Bauer E, Cheng J, Camarillo IG. An ex vivo co-culture model system to evaluate stromal–epithelial interactions in breast cancer. Int J Cancer. 2012;132:288–96.
Wang J, Xia TS, Liu XA, Ding Q, Du Q, Yin H, Wang S. A novel orthotopic and metastatic mouse model of breast cancer in human mammary microenvironment. Breast Cancer Res Treat. 2010;120:337–44.
Zheng MJ, Wang J, Chen YW, Xu L, Xue DD, Fu W, Zhang YF, Du Q, Zhao Y, Ling LJ, Ding Q, Liu XA, Zha XM, Zheng W, Xia TS, Wang S. A novel mouse model of gastric cancer with human gastric microenvironment. Cancer Lett. 2012;325:108–15.
Xia TS, Wang GZ, Ding Q, Liu XA, Zhou WB, Zhang YF, Zha XM, Du Q, Ni XJ, Wang J, Miao SY, Wang S. Bone metastasis in a novel breast cancer mouse model containing human breast and human bone. Breast Cancer Res Treat. 2012;132:471–86.
Baserga R. Growth regulation of the PCNA gene. J Cell Sci. 1991;98:433–6.
Hockenbery DM. The bcl-2 oncogene and apoptosis. Semin Immunol. 1992;4:413–20.
Semb H, Christofori G. The tumor-suppressor function of E-cadherin. Am J Hum Genet. 1998;63:1588–93.
Satelli A, Li S. Vimentin in cancer and its potential as a molecular target for cancer therapy. Cell Mol Life Sci. 2011;68:3033–46.
Sy MS, Mori H, Liu D. CD44 as a marker in human cancers. Curr Opin Oncol. 1997;9:108–12.
Yin H, Glass J. The phenotypic radiation resistance of CD44+/CD24− (or low) breast cancer cells is mediated through the enhanced activation of ATM signaling. PLoS One. 2011;6:e24080.
Akkiprik M, Nicorici D, Cogdell D, Jia YJ, Hategan A, Tabus I, Yli-Harja O, Dihua Y, Sahin A, Zhang W. Dissection of signaling pathways in fourteen breast cancer cell lines using reverse-phase protein lysate microarray. Technol Cancer Res Treat. 2006;5:543–51.
Yamanaka N, Wong CJ, Gertsenstein M, Casper RF, Nagy A, Rogers IM. Bone marrow transplantation results in human donor blood cells acquiring and displaying mouse recipient class I MHC and CD45 antigens on their surface. PLoS One. 2009;4:e8489.
Fleming JM, Miller TC, Kidacki M, Ginsburg E, Stuelten CH, Stewart DA, Troester MA, Vonderhaar BK. Paracrine interactions between primary human macrophages and human fibroblasts enhance murine mammary gland humanization in vivo. Breast Cancer Res. 2012;14:R97.
Acknowledgments
This research was supported in part by National Natural Science Foundation of China (81172502, 81302305), Program for Development of Innovative Research Team in the First Affiliated Hospital of NJMU (IRT-008), Natural Science Foundation of Jiangsu Province (BK20131027), Youth Talent Project in Jiangsu Province Hospital (FRC201308), a Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), and Jiangsu Province Innovation Project for Graduate Student of China (CXZZ11_0723, to MJZ).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ming-Jie Zheng and Jue Wang have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zheng, MJ., Wang, J., Xu, L. et al. Human mammary microenvironment better regulates the biology of human breast cancer in humanized mouse model. Med Oncol 32, 8 (2015). https://doi.org/10.1007/s12032-014-0427-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-014-0427-4