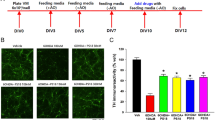
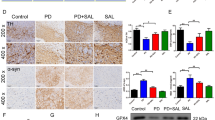
Abstract
Pituitary adenylate cyclase-activating polypeptide (PACAP) is an endogenous 38 amino acid containing neuropeptide with various cytoprotective functions including neuroprotection. Administration of PACAP has been shown to reduce damage induced by ischemia, trauma, or exogenous toxic substances. Moreover, mice deficient in PACAP are more vulnerable to damaging insults. In this study, we sought to determine whether PACAP may also be protective against salsolinol-induced toxicity in SH-SY5Y cells and, if so, elucidate its mechanism(s) of action. Salsolinol (SALS) is an endogenous dopamine metabolite with selective toxicity to nigral dopaminergic neurons, which are directly implicated in Parkinson’s disease (PD). SH-SY5Y cells, derived from human neuroblastoma cells, express high levels of dopaminergic activity and are used extensively as a model to study these neurons. Exposure of SH-SY5Y cells to 400 μM SALS for 24 h resulted in approximately 50 % cell death that was mediated by apoptosis as determined by cell flow cytometry and increases in caspase-3 levels. Cellular toxicity was also associated with reductions in brain-derived neurotrophic factor and phosphorylated cyclic AMP response element-binding protein. Pretreatment with PACAP dose-dependently attenuated SALS-induced toxicity and the associated apoptosis and the chemical changes. PACAP receptor antagonist PACAP6-38, in turn, dose-dependently blocked the effects of PACAP. Neither PACAP nor PACAP antagonist had any effect of its own on cellular viability. These results suggest the protective effects of PACAP in a cellular model of PD. Hence, PACAP or its agonists could be of therapeutic benefit in PD.





Similar content being viewed by others
References
Bitner S (2012) Cyclic AMP response element-binding protein (CREB) phosphorylation: a mechanistic marker in the development of memory enhancing Alzheimer's disease therapeutics. Biochem Pharmacol 83:705–714
Bollimuntha S, Ebadi M, Singh BB (2006) TRPC1 protects human SH-SY5Y cells against salsolinol-induced cytotoxicity by inhibiting apoptosis. Brain Res 1099:141–149
Botia B, Jolivel V, Burel D et al (2011) Neuroprotective effects of PACAP against ethanol-induced toxicity in the developing rat cerebellum. Neurotox Res 19:423–434
Copeland RL, Das JR, Kanaan YM (2007) Antiapoptotic effects of nicotine in its protection against salsolinol-induced cytotoxicity. Neurotox Res 12:61–69
Das JR, Tizabi Y (2009) Additive protective effects of donepezil and nicotine against salsolinol-induced cytotoxicity in SH-SY5Y cells. Neurotox Res 16:194–204
Dejda A, Sokolowska P, Nowak JZ (2005) Neuroprotective potential of three neuropeptides PACAP, VIP and PHI. Pharmacol Rep 57:307–320
Dogrukol-Ak D, Kumar VB, Ryerse JS, Farr SA, Verma S, Nonaka N (2009) Isolation of peptide transport system-6 from brain endothelial cells: therapeutic effects with antisense inhibition in Alzheimer and stroke models. J Cereb Blood Flow Metab 29:411–422
Fernandez HH (2012) Updates in the medical management of Parkinson disease. Cleve Clin J Med 79:28–35
Frechilla D, Garcia-Osta A, Palacios S, Cenarruzabebeitia E, Del Rio J (2001) BDNF mediates the neuroprotective effect of PACAP-38 on rat cortical neurons. Neuroreport 12:919–923
Guillot TS, Richardson JR, Wang MZ et al (2008) PACAP38 increases vesicular monoamine transporter 2 (VMAT2) expression and attenuates methamphetamine toxicity. Neuropeptides 42:423–434
Han BH, D’Costa A, Back SA et al (2000) BDNF blocks caspase-3 activation in neonatal hypoxia–ischemia. Neurobiol Dis 7:38–53
Healy DG, Abou-Sleiman PM, Wood NW (2004) PINK, PANK, or PARK? A clinicians’ guide to familial parkinsonism. Lancet Neurol 3:652–662
Jankovic J (2008) Parkinson’s disease: clinical features and diagnosis. J Neurol Neurosurg Psychiatry 79:368–376
Jantas D, Pytel M, Mozrzymas JW et al (2008) The attenuating effect of memantine on staurosporine-, salsolinol- and doxorubicin-induced apoptosis in human neuroblastoma SH-SY5Y cells. Eurochem Int 52:864–877
Kim DH, Zhao X (2005) BDNF protects neurons following injury by modulation of caspase activity. Neurocrit Care 3:71–76
Lang AE (2009) When and how should treatment be started in Parkinson disease. Neurology 72:S39–S43
Lazarovici P, Cohen G, Arien-Zakay H et al (2012) Multimodal neuroprotection induced by PACAP38 in oxygen–glucose deprivation and middle cerebral artery occlusion stroke models. J Mol Neurosci 48:526–540
Lewis-Tuffin LJ, Quinn PG, Chikaraishi DM (2004) Tyrosine hydroxylase transcription depends primarily on cAMP response element activity, regardless of the type of inducing stimulus. Mol Cell Neurosci 25:536–547
Li N, Liu GT (2010) The novel squamosamide derivative FLZ enhances BDNF/TrkB/CREB signaling and inhibits neuronal apoptosis in APP/PS1 mice. Acta Pharmacol Sin 31:265–272
Maruyama W, Yi H, Takahashi T et al (2004) Neuroprotective function of R-(–)-1-(benzofuran-2-yl)-2-propylaminopentane, [R-(–)-BPAP], against apoptosis induced by N-methyl(R)salsolinol, an endogenous dopaminergic neurotoxin, in human dopaminergic neuroblastoma SH-SY5Y cells. Life Sci 75:107–117
Masuo Y, Ohtaki T, Masuda Y, Tsuda M, Fujino M (1992) Binding sites for pituitary adenylate cyclase activating polypeptide (PACAP): comparison with vasoactive intestinal polypeptide (VIP) binding site localization in rat brain sections. Brain Res 575:113–123
Masuo Y, Suzuki N, Matsumoto H et al (1993) Regional distribution of pituitary adenylate cyclase activating polypeptide (PACAP) in the rat central nervous system as determined by sandwich-enzyme immunoassay. Brain Res 602:657–663
Morris HR (2005) Genetics of Parkinson’s disease. Ann Med 37:86–96
Nakamachi T, Tsuchida M, Kagami N et al (2012) IL-6 and PACAP receptor expression and localization after global brain ischemia in mice. J Mol Neurosci 48:518–525
Naoi M, Maruyama W, Nagy GM (2004) Dopamine-derived salsolinol derivatives as endogenous monoamine oxidase inhibitors: occurrence, metabolism and function in human brains. Neurotoxicology 25:193–204
Offen D, Sherki Y, Melamed E, Fridkin M, Brenneman DE, Gozes I (2000) Vasoactive intestinal peptide (VIP) prevents neurotoxicity in neuronal cultures: relevance to neuroprotection in Parkinson’s disease. Brain Res 854:257–262
Priestley JV, Michael-Titus AT, Tetziaff W (2012) Limiting spinal cord injury by pharmacological intervention. Handb Clin Neurol 109:463–484
Puerta E, Hervias I, Barros-Minones L et al (2010) Sildenafil protects against 3-nitropropionic acid neurotoxicity through the modulation of calpain, CREB, and BDNF. Neurobiol Dis 38:237–245
Purves D, Augustine GJ, Fitzpatrick D, Hall WC, LaMantia A, White LE (2008) Neuroscience, 4th edn. Sinauer Associates, Sunderland, pp 170–176
Racz B, Tamas A, Kiss P et al (2006) Involvement of ERK and CREB signaling pathways in the protective effect of PACAP in monosodium glutamate-induced retinal lesion. Ann N Y Acad Sci 1070:507–511
Ramlochansingh C, Taylor RE, Tizabi Y (2011) Toxic effects of low alcohol and nicotine combinations in SH-SY5Y cells are apoptotically mediated. Neurotox Res 20:263–269
Rat D, Schmitt U, Tippmann F et al (2011) Neuropeptide pituitary adenylate cyclase-activating polypeptide (PACAP) slows down Alzheimer's disease-like pathology in amyloid precursor protein-transgenic mice. FASEB J 25:3208–3218
Reglödi D, Somogyvari-Vigh A, Vigh S, Kozicz T, Arimura A (2000) Delayed systemic administration of PACAP38 is neuroprotective in transient middle cerebral artery occlusion in the rat. Stroke 31:1411–1417
Reglödi D, Tamas A, Lubics A, Szalontay L, Lengvari I (2004) Morphological and functional effects of PACAP in 6-hydroxydopamine-induced lesion of the substantia nigra in rats. Regul Pept 123:85–94
Reglödi D, Tamas A, Lengvari I, Toth G, Szalontay L, Lubics A (2006) Comparative study on the effects of PACAP in young, aging, and castrated males in a rat model of Parkinson’s disease. Ann NY Acad Sci 1070:518–524
Reglödi D, Kiss P, Lubics A, Tamas A (2011) Review on the protective effect of PACAP in models of neurodegenerative diseases in vitro and in vivo. Curr Pharm Des 17:962–972
Reglödi D, Kiss P, Szabadfi K et al (2012) PACAP is an endogenous protective factor-insights from PACAP-deficient mice. J Mol Neurosci 48:482–492
Samii A, Nutt JG, Ransom BR (2004) Parkinson’s disease. Lancet 363:1783–1793
Somogyvari-Vigh A, Reglödi D (2004) Pituitary adenylate cyclase activating polypeptide: a potential neuroprotective peptide. Curr Pharm Des 10:2861–2889
Sorimachi H, Mamitsuka H, Ono Y (2012) Understanding the substrate specificity of conventional calpains. Biol Chem 393:853–871
Storch A, Ott S, Hwang YI et al (2002) Selective dopaminergic neurotoxicity of isoquinoline derivatives related to Parkinson’s disease: studies using heterologous expression systems of the dopamine transporter. Biochem Pharmacol 63:909–920
Suk K, Park JH, Lee WH (2004) Neuropeptide PACAP inhibits hypoxic activation of brain microglia: a protective mechanism against microglial neurotoxicity in ischemia. Brain Res 1026:151–156
Szabadfi K, Atlasz T, Kiss P et al (2012) Mice deficient in pituitary adenylate cyclase activating polypeptide (PACAP) are more susceptible to retinal ischemic injury in vivo. Neurotox Res 21:41–48
Tamas A, Reglödi D, Farkas O et al (2012a) Effect of PACAP in central and peripheral nerve injuries. Int J Mol Sci 13:8430–8448
Tamas A, Szabadfi K, Nemeth A et al (2012b) Comparative examination of inner ear in wild type and pituitary adenylate cyclase activating polypeptide (PACAP)-deficient mice. Neurotox Res 21:435–444
Tsuchikawa D, Nakamachi T, Tsuchida M et al (2012) Neuroprotective effect of endogenous pituitary adenylate cyclase-activating polypeptide on spinal cord injury. J Mol Neurosci 48:508–517
Tuncel N, Korkmaz OT, Tekin N, Sener E, Akyuz F, Inal M (2012) Antioxidant and anti-apoptotic activity of vasoactive intestinal peptide (VIP) against 6-hydroxy dopamine toxicity in the rat corpus striatum. J Mol Neurosci 46:51–57
Vaudry D, Gonzalez BJ, Basille M et al (2000) The neuroprotective effect of pituitary adenylate cyclase-activating polypeptide on cerebellar granule cells in mediated through inhibition of the CED3-related cysteine protease caspase-3/CPP32. Proc Natl Acad Sci USA 97:13390–13395
Vaudry D, Falluel-Morel A, Bougault S et al (2009) Pituitary adenylate cyclase-activating polypeptide and its receptors: 20 years after the discovery. Pharmacol Rev 61:283–357
von Bohlen, Halbach O, Schober A, Krieglstein K (2004) Genes, proteins, and neurotoxins involved in Parkinson’s disease. Prog Neurobiol 73:151–177
Wang G, Qi C, Fan GH, Zhou HY, Chen SD (2005) PACAP protects neuronal differentiated PC12 cells against the neurotoxicity induced by a mitochondrial complex I inhibitor, rotenone. FEBS Lett 579:4005–4011
Wang G, Pan J, Tan YY et al (2008) Neuroprotective effects of PACAP27 in mice model of Parkinson’s disease involved in the modulation of K (ATP) subunits and D2 receptors in the striatum. Neuropeptides 42:267–276
Yaka R, He DY, Phamluong K, Ron D (2003) Pituitary adenylate cyclase-activating polypeptide (PACAP(1-38)) enhances N-methyl-d-aspartate receptor function and brain-derived neurotrophic factor expression via RACK1. J Biol Chem 278:9630–9963
Acknowledgments
This study was supported by NIH/NIGMS 2 SO6 GM08016-39 (YT) and MTA Momentum Program, TAMOP (4.2.1.B-10/2/KONV-2010-002, 4.2.2.A-11/1/KONV-2012-0024), Arimura Foundation, and OTKA K104984 (AT, DR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brown, D., Tamas, A., Reglödi, D. et al. PACAP Protects Against Salsolinol-Induced Toxicity in Dopaminergic SH-SY5Y Cells: Implication for Parkinson’s Disease. J Mol Neurosci 50, 600–607 (2013). https://doi.org/10.1007/s12031-013-0015-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-013-0015-7