Abstract
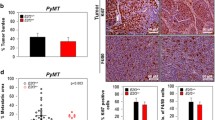
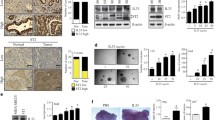
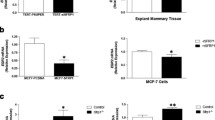
The prolactin-inducible protein (PIP) is considered a valuable biomarker that is associated with both benign and malignant pathological conditions of the mammary gland. The function of PIP in breast tumorigenesis remains unknown; however, evidence from our laboratory and others suggest that it regulates host immunity. Studies with PIP-deficient (PIP−/−) mice demonstrated significantly lower numbers of CD4+ T cells in their secondary lymphoid organs, impaired Th1 response, and impaired nitric oxide (NO) production. To further delineate the immunoregulatory role of PIP, we compared the expression of IFN-γR and TLR4, pro-inflammatory cytokine production, and intracellular signaling events by IFN-γ and lipopolysaccharide (LPS)-stimulated macrophages from wild-type (WT) and PIP−/− mice. We showed that although the expressions of IFN-γR and TLR4 were comparable, productions of pro-inflammatory cytokines were decreased in PIP−/− macrophages. This was associated with decreased phosphorylation of mitogen-activated protein kinase (MAPK) and signal transducer of activation of transcription (STAT) proteins in macrophages from PIP−/− mice. Interestingly, the expression of suppressors of cytokine signaling (SOCS) 1 and 3 proteins, known to suppress IFN-γ and LPS signaling, was higher in PIP−/− macrophages compared to those from WT mice. Collectively, our studies show that deficiency of PIP significantly affects intracellular signaling events leading to decreased pro-inflammatory cytokine production, and further confirms a role for PIP as an important immunoregulatory protein. This direct link between PIP and cell-mediated immunity, a key component of the immune system that is critical for cancer control, may have significant therapeutic implications.






Similar content being viewed by others
References
Emens LA. Breast cancer immunobiology driving immunotherapy: vaccines and immune checkpoint blockade. Expert Rev Anticancer Ther. 2012;12(12):1597–611. https://doi.org/10.1586/era.12.147.
Emens LA, Jaffee EM. Cancer vaccines: an old idea comes of age. Cancer Biol Ther. 2003;2(4 Suppl 1):S161–8.
Andre F, Dieci MV, Dubsky P, Sotiriou C, Curigliano G, Denkert C, et al. Molecular pathways: involvement of immune pathways in the therapeutic response and outcome in breast cancer. Clin Cancer Res. 2013;19(1):28–33. https://doi.org/10.1158/1078-0432.CCR-11-2701.
Cimino-Mathews A, Foote JB, Emens LA. Immune targeting in breast cancer. Oncology (Williston Park). 2015;29(5):375–85.
DeNardo DG, Brennan DJ, Rexhepaj E, Ruffell B, Shiao SL, Madden SF, et al. Leukocyte complexity predicts breast cancer survival and functionally regulates response to chemotherapy. Cancer Discov. 2011;1(1):54–67. https://doi.org/10.1158/2159-8274.CD-10-0028.
Thompson E, Taube JM, Elwood H, Sharma R, Meeker A, Warzecha HN, et al. The immune microenvironment of breast ductal carcinoma in situ. Mod Pathol. 2016;29(3):249–58. https://doi.org/10.1038/modpathol.2015.158.
Quigley DA, Kristensen V. Predicting prognosis and therapeutic response from interactions between lymphocytes and tumor cells. Mol Oncol. 2015;9(10):2054–62. https://doi.org/10.1016/j.molonc.2015.10.003.
Murphy LC, Lee-Wing M, Goldenberg GJ, Shiu RP. Expression of the gene encoding a prolactin-inducible protein by human breast cancers in vivo: correlation with steroid receptor status. Cancer Res. 1987;47(15):4160–4.
Myal Y, Iwasiow B, Cosby H, Yarmill A, Blanchard A, Tsuyuki D, et al. Analysis of tissue- and hormone-specific regulation of the human prolactin-inducible protein/gross cystic disease fluid protein-15 gene in transgenic mice. J Mol Endocrinol. 1998;21(2):217–23.
Myal Y, Robinson DB, Iwasiow B, Tsuyuki D, Wong P, Shiu RP. The prolactin-inducible protein (PIP/GCDFP-15) gene: cloning, structure and regulation. Mol Cell Endocrinol. 1991;80(1–3):165–75.
Clark JW, Snell L, Shiu RP, Orr FW, Maitre N, Vary CP, et al. The potential role for prolactin-inducible protein (PIP) as a marker of human breast cancer micrometastasis. Br J Cancer. 1999;81(6):1002–8. https://doi.org/10.1038/sj.bjc.6690799.
Hassan MI, Bilgrami S, Kumar V, Singh N, Yadav S, Kaur P, et al. Crystal structure of the novel complex formed between zinc alpha2-glycoprotein (ZAG) and prolactin-inducible protein (PIP) from human seminal plasma. J Mol Biol. 2008;384(3):663–72. https://doi.org/10.1016/j.jmb.2008.09.072.
Bhargava R, Beriwal S, Dabbs DJ. Mammaglobin vs GCDFP-15: an immunohistologic validation survey for sensitivity and specificity. Am J Clin Pathol. 2007;127(1):103–13. https://doi.org/10.1309/TDP92PQLDE2HLEET.
Fiel MI, Cernaianu G, Burstein DE, Batheja N. Value of GCDFP-15 (BRST-2) as a specific immunocytochemical marker for breast carcinoma in cytologic specimens. Acta Cytol. 1996;40(4):637–41.
Yan Z, Gidley J, Horton D, Roberson J, Eltoum IE, Chhieng DC. Diagnostic utility of mammaglobin and GCDFP-15 in the identification of metastatic breast carcinoma in fluid specimens. Diagn Cytopathol. 2009;37(7):475–8. https://doi.org/10.1002/dc.21039.
Darb-Esfahani S, von Minckwitz G, Denkert C, Ataseven B, Hogel B, Mehta K, et al. Gross cystic disease fluid protein 15 (GCDFP-15) expression in breast cancer subtypes. BMC Cancer. 2014;14:546. https://doi.org/10.1186/1471-2407-14-546.
Luo MH, Huang YH, Ni YB, Tsang JY, Chan SK, Shao MM, et al. Expression of mammaglobin and gross cystic disease fluid protein-15 in breast carcinomas. Hum Pathol. 2013;44(7):1241–50. https://doi.org/10.1016/j.humpath.2012.10.009.
Baniwal SK, Little GH, Chimge NO, Frenkel B. Runx2 controls a feed-forward loop between androgen and prolactin-induced protein (PIP) in stimulating T47D cell proliferation. J Cell Physiol. 2012;227(5):2276–82. https://doi.org/10.1002/jcp.22966.
Lewis GH, Subhawong AP, Nassar H, Vang R, Illei PB, Park BH, et al. Relationship between molecular subtype of invasive breast carcinoma and expression of gross cystic disease fluid protein 15 and mammaglobin. Am J Clin Pathol. 2011;135(4):587–91. https://doi.org/10.1309/AJCPMFR6OA8ICHNH.
Autiero M, Cammarota G, Friedlein A, Zulauf M, Chiappetta G, Dragone V, et al. A 17-kDa CD4-binding glycoprotein present in human seminal plasma and in breast tumor cells. Eur J Immunol. 1995;25(5):1461–4.
Autiero M, Gaubin M, Mani JC, Castejon C, Martin M, El MS, et al. Surface plasmon resonance analysis of gp17, a natural CD4 ligand from human seminal plasma inhibiting human immunodeficiency virus type-1 gp120-mediated syncytium formation. Eur J Biochem. 1997;245(1):208–13.
Lee B, Bowden GH, Myal Y. Identification of mouse submaxillary gland protein in mouse saliva and its binding to mouse oral bacteria. Arch Oral Biol. 2002;47(4):327–32.
Schenkels LC, Walgreen-Weterings E, Oomen LC, Bolscher JG, Veerman EC, Nieuw Amerongen AV. In vivo binding of the salivary glycoprotein EP-GP (identical to GCDFP-15) to oral and non-oral bacteria detection and identification of EP-GP binding species. Biol Chem. 1997;378(2):83–8.
Blanchard A, Nistor A, Castaneda FE, Martin D, Hicks GG, Amara F, et al. Generation and initial characterization of the prolactin-inducible protein (PIP) null mouse: accompanying global changes in gene expression in the submandibular gland. Can J Physiol Pharmacol. 2009;87(10):859–72.
Myal Y, Iwasiow B, Cosby H, Yarmill A, R.P.C. S. Mouse mammary tumor virus (MMTV)-targeted gene expression of the human gross cystic disease fluid protein-15/prolactin inducible protein (GCDFP-15/PIP) in the mammary gland of transgenic mice. Transgenics. 1998;2:327–32.
Li J, Liu D, Mou Z, Ihedioha OC, Blanchard A, Jia P, et al. Deficiency of prolactin-inducible protein leads to impaired Th1 immune response and susceptibility to Leishmania major in mice. Eur J Immunol. 2015;45(4):1082–91. https://doi.org/10.1002/eji.201445078.
Pernis A, Gupta S, Gollob KJ, Garfein E, Coffman RL, Schindler C, et al. Lack of interferon gamma receptor beta chain and the prevention of interferon gamma signaling in TH1 cells. Science. 1995;269(5221):245–7.
Pernis A, Gupta S, Yopp J, Garfein E, Kashleva H, Schindler C, et al. Gamma chain-associated cytokine receptors signal through distinct transducing factors. J Biol Chem. 1995;270(24):14517–22.
Vodovotz Y, Bogdan C, Paik J, Xie QW, Nathan C. Mechanisms of suppression of macrophage nitric oxide release by transforming growth factor beta. J Exp Med. 1993;178(2):605–13.
Yang H, Young DW, Gusovsky F, Chow JC. Cellular events mediated by lipopolysaccharide-stimulated toll-like receptor 4. MD-2 is required for activation of mitogen-activated protein kinases and Elk-1. J Biol Chem. 2000;275(27):20861–6. https://doi.org/10.1074/jbc.M002896200.
Liu D, Zhang T, Marshall AJ, Okkenhaug K, Vanhaesebroeck B, Uzonna JE. The p110delta isoform of phosphatidylinositol 3-kinase controls susceptibility to Leishmania major by regulating expansion and tissue homing of regulatory T cells. J Immunol. 2009;183(3):1921–33. https://doi.org/10.4049/jimmunol.0901099.
Wurster AL, Tanaka T, Grusby MJ. The biology of Stat4 and Stat6. Oncogene. 2000;19(21):2577–84. https://doi.org/10.1038/sj.onc.1203485.
Karin M, Ben-Neriah Y. Phosphorylation meets ubiquitination: the control of NF-[kappa]B activity. Annu Rev Immunol. 2000;18:621–63. https://doi.org/10.1146/annurev.immunol.18.1.621.
Naka T, Narazaki M, Hirata M, Matsumoto T, Minamoto S, Aono A, et al. Structure and function of a new STAT-induced STAT inhibitor. Nature. 1997;387(6636):924–9. https://doi.org/10.1038/43219.
Yasukawa H, Misawa H, Sakamoto H, Masuhara M, Sasaki A, Wakioka T, et al. The JAK-binding protein JAB inhibits Janus tyrosine kinase activity through binding in the activation loop. EMBO J. 1999;18(5):1309–20. https://doi.org/10.1093/emboj/18.5.1309.
Rao KM. Molecular mechanisms regulating iNOS expression in various cell types. J Toxicol Environ Health B Crit Rev. 2000;3(1):27–58. https://doi.org/10.1080/109374000281131.
Babon JJ, Kershaw NJ, Murphy JM, Varghese LN, Laktyushin A, Young SN, Lucet IS, Norton RS, Nicola NA. Suppression of cytokine signaling by SOCS3: characterization of the mode of inhibition and the basis of its specificity. Immunity, 2012;36(2):239–250.
Di Piazza M, Nowell CS, Koch U, Durham AD, Radtke F. Loss of cutaneous TSLP-dependent immune responses skews the balance of inflammation from tumor protective to tumor promoting. Cancer Cell. 2012;22(4):479–93. https://doi.org/10.1016/j.ccr.2012.08.016.
Lo Kuan E, Ziegler SF. Thymic stromal lymphopoietin and cancer. J Immunol. 2014;193(9):4283–8. https://doi.org/10.4049/jimmunol.1400864.
Pedroza-Gonzalez A, Xu K, Wu TC, Aspord C, Tindle S, Marches F, et al. Thymic stromal lymphopoietin fosters human breast tumor growth by promoting type 2 inflammation. J Exp Med. 2011;208(3):479–90. https://doi.org/10.1084/jem.20102131.
Demehri S, Cunningham TJ, Manivasagam S, Ngo KH, Moradi Tuchayi S, Reddy R, et al. Thymic stromal lymphopoietin blocks early stages of breast carcinogenesis. J Clin Invest. 2016;126(4):1458–70. https://doi.org/10.1172/JCI83724.
Funding
This study was supported by grants from the Natural Sciences and Engineering Research Council of Canada (NSERC), Manitoba Health Research Council (MHRC), and Canadian Institutes of Health Research (CIHR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The University of Manitoba Animal Care and Use Committee approved this project. All the mice used were maintained at the Central Animal Care Services under specific pathogen-free conditions and used according to guidelines stipulated by the Canadian Council for Animal Care.
Rights and permissions
About this article
Cite this article
Ihedioha, O., Blanchard, A.A., Balhara, J. et al. The human breast cancer-associated protein, the prolactin-inducible protein (PIP), regulates intracellular signaling events and cytokine production by macrophages. Immunol Res 66, 245–254 (2018). https://doi.org/10.1007/s12026-018-8987-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-018-8987-6