Abstract
The induction of 2F5- and 4E10-like antibodies broadly neutralising HIV-1 and targeting the membrane external proximal region (MPER) of the transmembrane envelope protein gp41 would be a major advancement for the development of a preventive HIV-1 vaccine, but successful attempts remain rare. Recent studies demonstrated that broadly reactive antibodies develop relatively late during infection and after intensive affinity maturation. Therefore, a prolonged antigen delivery might be beneficial to induce them. Replicating foamy viruses which are characterised by apathogenic but persistent infection could represent suitable carrier viruses for this purpose. In order to develop such a system, we modified the accessory foamy virus Bet protein to contain the MPER of gp41, or the MPER linked to the stabilising fusion peptide proximal region of gp41 and analysed here the antigenic and immunogenic properties of such hybrid proteins. The antigens, expressed and purified to homogeneity, were recognised by the monoclonal antibodies 2F5 and 4E10 with nanomolar affinities and induced high levels of antibodies specific to gp41 after immunisation of rats. The antisera also bound to virus particles attached to infected cells, and peptide-based epitope mapping showed that they recognised the 2F5 epitope. Although no HIV-1 neutralising activity was observed, the presented data demonstrate that using the foamy virus Bet for HIV-1 epitope delivery is successfully applicable. Together with the attractive potential for sustained antigen expression after transfer to replicating virus, these results should therefore provide a first basis for the development of chimeric foamy viruses as novel HIV-1 vaccine vectors.
Similar content being viewed by others
Introduction
For HIV-1 vaccine development, induction of broadly neutralising antibodies (bnAb) is the most promising approach to prevent infection and to avoid integration of the provirus into the genome of the target cell where it may persist [1, 2]. The viral envelope proteins composed of the surface and transmembrane (TM) subunits are the main target of neutralising antibodies, and several potent bnAb directed against each subunit have been isolated from infected patients (reviewed in [2, 3]). Most of these antibodies show unique properties such as a high degree of somatic mutation, prolonged CDR3 loops and polyreactivity and occur in about 20 % of infected individuals only after about 4 years of infection [4–7]. The findings suggest that these antibodies may arise from rare B cell stimulation events or after intensive affinity maturation [1, 6, 8], which is only partly achieved by repeated antigen administration [9]. Given that various attempts to induce bnAb completely failed or resulted in low-titred neutralising antibody responses with limited breadth [2], alternative approaches should be explored. Replicating viral vectors and attenuated vaccines have been found to be the most effective way to prevent infection in the non-human primate model [10–13] and permit long-term antigen delivery by simultaneously stimulating strong cellular immune responses [10]. However, most of the replicating systems available to date raise safety concerns that limit/exclude their application in humans [14]. Foamy viruses (FV) are apathogenic viruses of the Spumaviriniae subfamily which are characterised by persistent infection, preferential integration sites, genetic stability and wide tissue tropism [15–18], favouring their application for gene therapy and vaccine purposes [19–22]. Similarly, the feline foamy virus (FFV) was used as convenient model for the application in cats [23–26]. In order to develop a vector system that could be tested in small animals and encouraged by previous immunisation studies with an antigen of the feline calicivirus [25], a strategy for HIV-1 epitope delivery using the accessory FFV Bet protein was evaluated here. Bet is particularly useful as carrier antigen as it is highly expressed in infected cells, resulting in a strong antibody response in infected animals [27–29], and it was reported to be secreted and taken up by adjacent cells [30, 31], permitting the presentation of proteasome-unprocessed conformational epitopes to immune cells. As HIV-1 antigen, the membrane proximal external region (MPER) of the TM protein gp41 of HIV was selected to be appended to the FFV Bet protein. This domain is targeted by three bnAb, 2F5, Z13 and 4E10, and is therefore of particular interest as vaccine target [32–35]. However, MPER-based vaccine development is complicated by the structural flexibility of the MPER required to promote membrane fusion and uncertainty about the conformation able to induce bnAb [36–41]. Furthermore, the necessity for a lipid environment for induction of MPER-directed bnAb is still under intensive investigation [42, 43]. In structural analyses, the 2F5-bound epitope ELDKWA forms a slightly distorted type 1 β-turn compared with an α-helical structure when the MPER is analysed alone [37, 39, 44]. Importantly, 2F5 also shows a higher affinity to antigens fixing its epitope as a β-turn compared to an α-helix, indicating that this conformation might resemble the initial B cell trigger [45–48]. In contrast, the bnAb 4E10 recognises the adjacent α-helical core sequence NWFN/DIT in the MPER, and its affinity is enhanced when the antigens support α-helical structures [49–51]. Recent investigations supported an interaction of the MPER with the fusion peptide proximal region (FPPR) of gp41 and also showed that peptides corresponding to the FPPR increased binding of 2F5 to its epitope [52–55]. This was attributed to a FPPR-induced orientation of MPER residues coinciding with an increased level of β-turn/β-sheet conformation [56, 57]. In order to use its constraining property, an additional antigen containing the FPPR and the MPER was included here. With the objective to evaluate bnAb binding to such novel Bet/HIV-1 gp41 hybrids before transfer to replicating FFV and to study the epitope specificity of antibodies obtained in response to vaccination, recombinant fusion proteins of the FFV Bet protein and the MPER and FPPR sequences of gp41 of HIV-1 were produced in this study and characterised and the antibody response in immunised rats was studied.
Materials and methods
Expression plasmids
To clone pQE-Bet and pQE-Bet/HIV-1 constructs, the complete FFV Bet sequence (UniProt ID O93036) with additional four amino acids (AGAA) was amplified from the plasmid pBC12-Bet with the primers FFV Bet-fwd (5′-atggcttcaaaatacccggaagaaggacca-3′) and FFV Bet-rev (5′-tcatctgactctgaagctggtgcagccggctag-3′) containing a NaeI site and introduced into the StuI and BamHI linearised prokaryotic expression vector pQE-30-Xa (Qiagen, Hilden, Germany). The constructs pQE-BetE1, pQE-BetE2 and pQE-BetE1–E2 were obtained by subcloning E1, E2 or E1–E2 sequences (Fig. 1a) with additional N-terminal linker amino acids (SGSAGAGAGGG) from two synthetic plasmids (GeneArt, Regensburg, Germany) which were introduced into the NaeI/HindIII digested pQE-Bet vector. All constructs were verified by sequencing.
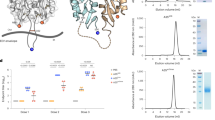
Schematic presentation of gp41 of HIV-1 (a) and of FFV Bet and the FFV Bet/HIV-1 fusion proteins used for immunisation (b). FP fusion peptide, FPPR fusion peptide proximal region, NHR/CHR N-terminal and C-terminal helical region, MPER membrane proximal external region, MSD membrane spanning domain, C-C cysteine loop, TM transmembrane envelope protein, His histidine tag, Factor Xa factor Xa protease cleavage site, Bet full-length FFV Bet sequence, Linker 4 (FFV Bet) or 15 (all other constructs) amino acid spacer. E1–HIV-1 FPPR-derived domain, E2–HIV-1 MPER-derived domain, E1–E2, HIV-1 E1 and E2 domains connected by a flexible loop sequence. The theoretical molecular mass of all antigens is indicated. Lines below the E2 sequence of gp41 of HIV-1 mark the location of the neutralising epitopes of 2F5 and 4E10. (c) Purity of the produced antigens. Two micrograms of recombinant proteins was loaded for SDS–PAGE and stained by Coomassie blue. The molecular mass of the marker is indicated. (d) Recognition of the produced antigens by the monoclonal antibodies 2F5 and 4E10 and a FPPR-specific antiserum. One hundred and fifty nanograms of recombinant proteins was transferred onto a PVDF membrane and tested with the antibodies 2F5 (0.4 μg/ml), 4E10 (1.2 μg/ml) and an E1-specific rat serum (1:800) to confirm the presence of the corresponding HIV-1 domains
Protein purification
After optimisation in 96-deep-well plates [58], proteins were expressed in Escherichia coli SCS-1 bacteria (Agilent Technologies, Austin, USA) using 2YT medium at 37 °C and 1 mM IPTG. For lysis, bacterial pellets were resuspended in PBS containing 0.1 mg/ml lysozyme, incubated on ice for 15 min and then sonicated for 3 × 20 s. Inclusion bodies were collected by centrifugation (10,000g, 20 min) and resuspended in buffer A (100 mM NaH2PO4, 10 mM Tris–HCl, 6 M GuHCl, 300 mM NaCl and 10 mM imidazole, pH 8.0). After two hours of centrifugation (25,000g, 20 min, 4 °C), the supernatant was loaded onto a HisTrap Ni–NTA column (GE Healthcare, Munich, Germany), washed with buffer B (100 mM NaH2PO4, 10 mM Tris–HCl, 8 M urea, 300 mM NaCl and 10 mM imidazole, pH 8.0) and proteins which bound were eluted using buffer C (100 mM NaH2PO4, 10 mM Tris–HCl, 8 M urea, 300 mM NaCl and 500 mM imidazole, pH 8.0). Eluted fractions (2 mg/ml) were incubated with 20 mM β-mercaptoethanol for at least 3 h and renatured by 1:20 dilution in refolding buffer (50 mM HEPES, 300 mM NaCl and 100 mM l-arginine, pH 9.0) for 24 h at room temperature followed by dialysis overnight against phosphate buffer (50 mM NaH2PO4, 300 mM NaCl, pH 8.0). Proteins were concentrated by another Ni–NTA chromatography step using imidazole-supplied phosphate buffers.
ELISA and serum titration
ELISA plates (Greiner Bio-One, Frickenhausen, Germany) were coated with 100 ng/well of Bet/HIV-1 antigens or 200 ng/well of purified gp41 (UniProt ID P04578, amino acids 532-681) expressed in bacteria or synthetic HIV peptides (E1: AAGSTMGAASMTLTVQARLLLSKKKK, E2: KKKKEQELLELDKWASLWNWFNITNWL, GeneCust, Dudelange, Luxembourg) and then blocked for 1 h at 37 °C with 5 % BSA in PBS/0.05 % Tween (PBS-T). For each serum a tenfold dilution in 2.5 % BSA with PBS-T was prepared in triplicate, and plates were then incubated with the samples for 1 h at 37 °C. After washing and addition of a peroxidase (HRP)-coupled anti-rat IgG-HRP antibody (Dako, Hamburg, Germany, 1:3000), plates were developed with OPD (o-phenylenediamine dichloride) and read at 492 nm. For the calculation of endpoint titres, the absorbance measured for immune sera at every dilution was plotted against the dilution step and the data were fitted by a logarithmic regression function. Endpoint titres were defined as the highest serum dilution exceeding the mean absorbance of six control wells of pre-immune serum plus three standard deviations, corresponding to a confidence level of 98 % [59]. Data were statistically evaluated using the VassarStats software (http://vassarstats.net) by applying Shapiro–Wilk and F-test analysis for testing normal distribution and equality of variances. For comparisons between groups the Kruskal–Wallis one-way analysis of variance and the Welch’s t test for direct comparison between two groups were used. Differences were considered significant, if p values below 0.05 were obtained.
Surface plasmon resonance (SPR)
SPR measurements were performed on a Biacore X100 device with anti-human IgG-Fc-specific antibodies immobilised on CM5 chips (all GE Healthcare, Munich, Germany). 2F5 or 4E10 in 50 mM NaH2PO4 and 300 mM NaCl, pH 8.0, was captured at 300–500 response units (RU) on flow cell 2, and three increasing concentrations of fusion proteins diluted in 50 mM NaH2PO4 and 300 mM NaCl, pH 8.0, were injected on both flow cells. After regeneration with 10 mM glycine, pH 2.0, and antibody recapturing, blank resonance was measured by three injections of dilution buffer. Sensograms were reference cell–subtracted and blank-normalised values from at least two repeated experiments. Curves were fitted using the 1:1 Langmuir algorithm, and quality of fitting was checked by assuring K D values in the range of measured protein concentrations, plausible R max values, small χ 2 values as well as residuals within recommended borders.
Immunisation
Wistar rats (Charles River, Wilmington, USA) in groups of four were immunised three times at intervals of 3 weeks with 250 μg of each antigen. One millilitre of protein emulsified in Freund’s adjuvant was injected either subcutaneously (s.c.) at four sites of the animal for the first injection or s.c. (4 × 200 μl) and intramuscularly (2 × 100 μl) for the second and third injections, respectively. Blood was taken by retrobulbar bleeding before immunisation (pre-immune sera, PI), 2 days before boost injections and 3 weeks after the last boost (serum 1, 2 and 3, respectively). All manipulations were in accordance with regular animal welfare guidelines and approved by the Landesamt für Gesundheit und Soziales, Berlin, Germany.
Epitope mapping
Epitope mapping was performed with PepStar peptide microarrays (JPT, Berlin, Germany) spotted with 15-mer peptides (12-residue overlap) of the extended HIV-1 HXB2 FPPR domain (amino acids 512–559) and the extended MPER domain (amino acids 644–691) as well as rat IgG as internal control. Sera were incubated on the array using a 24-multi-well hybridisation chamber (ArrayIt, Sunnyvale, USA) following the protocol recommended by JPT. Bound antibodies were detected using a DyLight 649 conjugated AffiniPure goat anti-rat IgG antibody (Jackson Immuno Research Labs, West Grove, USA, 1:500) and read at 635 nm in a GenePix 4000 microarray scanner (Molecular Devices, Sunnyvale, USA). Epitopes were defined as central amino acids shared by overlapping peptides with signal intensities above a 5000 RLU threshold.
Flow cytometry and immunofluorescence
C8166 cells were infected at a MOI of 0.5 with cell-free supernatants obtained from pNL-4.3-transfected 293T cells and further cultivated for 48 or 72 h for indirect immunofluorescence or FACS analysis, respectively. Non-infected C8166 cells were assayed in parallel to assure the specificity of staining, and no binding was observed (not shown). Cells were pelleted and washed twice with PBS, fixed by incubation with 2 % paraformaldehyde in PBS for 15 min and then used either directly for FACS staining or attached to poly-lysine-coated glass slides, dried and rehydrated in PBS overnight. Samples were blocked with 10 % normal goat serum in PBS for 1 h and then antisera or a pre-immune rat serum as control (all 1:100 in 3 % goat serum, PBS) applied for 1 h. Cells were washed with PBS followed by incubation with a goat anti-rat IgG-FITC-conjugated secondary antibody (SantaCruz Biotechnology, Santa Cruz, USA, 1:400), washed again and then either analysed in a FACS Calibur flow cytometer (Becton–Dickinson, Heidelberg, Germany) or embedded in DAPI-containing mounting medium (Vectashield, Vector Laboratories, Burlingame, USA) for analysis on a Zeiss Axiovert 200 microscope using Cell-F imaging software (Olympus, Hamburg, Germany).
Neutralisation assay
TZM-bl cells (1 × 104/well) were seeded in a 96-well cell culture plate. The next day, antisera were diluted 1:25 with medium and 50 μl of the dilution was incubated with 50 μl of 200 infectious units of titrated HIV-1 pNL4.3 for 30 min at 37 °C and then added to the cells. As controls, non-infected, infected and cells incubated with virus and 2F5 were used. Infection was analysed after 48 h by staining the cells with X-Gal solution (5 mM K3Fe(CN)6, 5 mM K4Fe(CN)6, 2 mM MgCl2 and 1 mg/ml X-Gal in PBS) and ELISPOT counting as described previously [60]. Neutralisation was defined as 50 % reduction in stained cells when immune sera and pre-immune sera were compared.
Results
Antigen design, production and purification
To add HIV-1 gp41 sequences to the bet sequence of FFV, a Bet expression vector was constructed in which the FPPR-derived E1 sequence, the MPER-derived E2 sequence or both E1 and E2 sequences connected by a loop sequence were subsequently introduced to the C-terminus (Fig. 1a, b). Between Bet and the corresponding HIV-1 parts, a flexible 15-amino acid glycine–serine-rich linker was introduced with the intention that HIV-1 epitopes protrude from the fusion partner and are thus more accessible for antibodies and immune cells. The E1–E2 loop construct used here was derived from the previously described alpha-turn-alpha peptide [61], which promotes the interaction of the alpha helical parts as a coiled coil [62]. Replacing the original alpha helices by HIV-1 FPPR and MPER domains except for eight amino acids flanking both termini of the loop, the construct was designed to mimic a form of gp41, where MPER and FPPR are in close proximity [63]. Additionally, both domains were connected with an amino acid spacing that allows interaction of contacting residues as found in the crystal structure of gp41 [38]. This construct was optimised and validated by secondary structure prediction algorithms and α-helical wheel projections to fit the desired criteria (Online Resource 1). Then, all three fusion proteins and Bet alone were produced in E. coli, purified by affinity chromatography and refolded by rapid dilution with typical yields of 11–18 mg/L expression culture. Characterisation of the antigens by SDS–PAGE showed their purity which usually exceeded 90 % as judged by densitometric scanning of Coomassie-stained gels (Fig. 1c).
Recognition of the produced antigens by 2F5 and 4E10
To examine their reactivity with 2F5 and 4E10, antigens were first analysed in a Western blot assay. 2F5 and to a weaker extent also 4E10 reacted with all antigens containing the MPER epitopes but not with the unmodified Bet or the Bet-E1 protein (Fig. 1d). Conversely, an epitope-mapped rat antiserum specific to the HIV-1 gp41 FPPR (recognising the epitope 541-TLTVQARQL-570) reacted with all E1-containing proteins but not with the Bet-E2 antigen (Fig. 1d). Since denatured gp41 was generally better recognised by 2F5 and worse by 4E10 when compared to soluble gp41 [64], binding to non-denatured proteins was investigated in parallel (Fig. 2). In the case of 2F5, minor differences in binding to the different E2-hybrid antigens were observed, with a reduced binding to the Bet-E1–E2 protein (Fig. 2). In contrast, 4E10 bound strongly to the Bet-E1–E2 loop antigen, but poorly to the Bet-E2 or the combination of the Bet-E1 and Bet-E2 proteins and as expected not to Bet-E1 (Fig. 2). These results demonstrated that all hybrid antigens properly displayed the corresponding HIV-1 domain(s) and that the presence of the E1 domain was beneficial to the presentation of the 4E10 but not the 2F5 epitope.
Recognition of the produced antigens by the monoclonal antibodies 2F5 and 4E10 under non-denaturing conditions in an ELISA. One hundred nanograms of indicated Bet/HIV-1 antigens in phosphate buffer was coated and incubated with 2F5 (11.3 μg/μl) or 4E10 (11.9 μg/μl) serially diluted as indicated, followed by detection with an HRP anti-human IgG antibody and developed with OPD. Whereas only minor differences in 2F5 binding were detected, threefold increased binding of 4E10 to the FPPR-stabilised Bet-E1–E2 protein was found
SPR analysis of 2F5 and 4E10 binding to the produced antigens
To study the binding of 2F5 and 4E10 to the hybrid proteins in more detail, SPR experiments were performed (Table 1, Online Resource 2). 2F5 binding to antigens containing the MPER alone or in combination with the FPPR domain was detectable down to the nanomolar range (K D = 4.8, 5.9 and 19 nM for Bet-E2, Bet-E1 + Bet-E2 and Bet-E1–E2, respectively), whereas no interaction was observed for the Bet-E1 control construct (Table 1, Online Resource 2). As above, no increase in binding was measured when a mixture of Bet-E2 and Bet-E1 was tested. Also, the affinity of 2F5 to the loop antigen was lower compared to Bet-E2 due to a twofold slower association and dissociation rate (Table 1). In line with the weaker Western blot and ELISA reactivity of 4E10, much higher fusion protein concentrations were needed in the SPR studies. Due to the precipitation of recombinant Bet/HIV-1 proteins above 1 mg/ml (corresponding to 15-20 μM), no suitable concentration ranges to obtain reliable data for the Bet-E2 antigen and the combination of Bet-E2 and Bet-E1 could be selected. At the highest concentration tested (8 μM), no binding of Bet-E1 and a weak signal (15 RU) for Bet-E2 were observed, suggesting that the K D values of Bet-E2 were probably in the micromolar range (data not shown). However, when Bet-E1–E2 was analysed, kinetic constants could be determined, since nanomolar protein concentrations yielded stable data (Table 1). The dissociation constant of 223 nM resulted from slow target recognition (k on = 2.08 × 103/M s) and a moderate dissociation rate (k off = 4.66 × 10−4/s, Table 1). Thus, the SPR data substantiated the ELISA results and the beneficial effect of the FPPR residues for enhanced 4E10 antibody binding.
Immunogenicity of fusion proteins
After validation of their antigenicity, rats were immunised with the fusion proteins. All animals showed a strong immune response to the administered antigens with antibody titres of 104–106 already after the first immunisation (Fig. 3, Online Resource 3). The response peaked after the second injection and then remained at this level or slightly decreased, with no significant differences between the groups (Kruskal–Wallis, p = 0.262, p = 0.243 and p = 0.385 for first, second and third immunisation, respectively). To study the immune response against the introduced HIV-derived E1 and E2 domains and to analyse whether these antibodies are also able to bind to gp41 of HIV-1, ELISA was performed using synthetic E1 and E2 peptides as well as purified recombinant gp41 produced in bacteria. All sera recognised the HIV-1 peptides as well as gp41 with titres ranging from 102 to 105 (Fig. 4, Online Resource 4). Notably, the reaction against the E1 domain (mean titre 105) was significantly stronger than that against the E2 domain (mean titre 103) in groups immunised with Bet-E1 and Bet-E2 antigen (Welch’s t test p = 0.027, Fig. 4a, b). This was also true for sera from animals immunised with a mixture of both hybrid antigens (p = 0.0238) but did not reach significance in case of the hybrid E1–E2 loop antigen (p = 0.1565, Fig. 4c, d). Among all animals immunised with the Bet-E2 protein alone developed the strongest antibody response against the E2 peptide with mean titres up to 8 × 103 (p = 0.1274 and 0.0729 for comparisons to groups 366 and 375, respectively), followed by animals immunised with the mixture of Bet-E1 plus Bet-E2 (p = 0.0809 for group 375, Fig. 4b and b, Online Resource 4). The antibody response against recombinant gp41 correlated well with that against each individual domain (Fig. 4a–d) and was confirmed by Western blot analysis (Online Resource 5). Taken together, these results demonstrated that all antigens used were highly immunogenic and that HIV-1-specific antibodies were induced.
Titres of the immune sera after immunisation with Bet fusion proteins as determined by endpoint dilutions. (a) Sera from rat group 364 immunised with Bet-E1, (b) from group 365 immunised with Bet-E2, (c), (d) from group 366 immunised with both Bet/HIV-1 hybrid antigens (e) and from group 375 immunised with Bet-E1–E2. PI = pre-immune serum, 1, 2, 3 = serum of the first, second and third bleeding, respectively
Reactivity of sera from animals immunised with the hybrid proteins against gp41 and synthetic E1 and E2 peptides as measured in an ELISA. Sera were serially diluted in a range of 102–107. The endpoint titres of (a) sera from rat group 364, (b) sera from rat group 365 and (c), (d) sera from rat groups 366 and 375, all titrated against the E1 and/or E2 peptides as well as recombinant gp41, are given. The administered antigen is indicated in brackets
Epitope mapping of the antisera
By mapping sera using overlapping peptides corresponding to the FPPR and MPER, distinct epitopes were identified (Fig. 5). As shown by two representative mappings, all rats immunised with the Bet-E1 hybrid antigen developed a strong response against the E1 domain with the epitope TLTVQARQL. Administration of the Bet-E2 hybrid antigen to rats resulted in antibodies recognising the epitopes ELLELDKWA or ELLELDKWASLW overlapping with the 2F5 epitope (underlined). Sera from animals immunised with a mixture of the E1 and E2 hybrid protein showed a predominant response to the E1 epitope TLTVQARQL and a rather weak response to the E2 epitope NEQELLELDKWASLW. When sera from animals treated with the Bet-E1–E2 loop antigen were investigated, the main response was directed against the E1 epitope TLTVQARQL, but the E2 response was variable, with one serum reacting with NEQELLELDKWA, whereas no MPER reactivity was observed in case of a second animal (Fig. 5). These results were consistent with the prior ELISA titration experiments and confirmed the overall lower E2 reactivity observed before. In terms of consistency and strength in which 2F5 epitope-specific antibodies were induced, the Bet-E2 antigen appeared to be superior to all other antigens.
Epitope mapping of the sera from immunised animals. Microarrays spotted with overlapping 15-mer peptides of the prolonged HIV-1 FPPR and MPER domains were incubated with antisera and bound antibodies detected by a fluorescent secondary anti-rat antibody. Results are plotted as relative light units (RLU) against the individual FPPR and MPER peptides. The group number, the administered antigen and the recognised epitopes as defined as central amino acids shared by overlapping peptides exceeding a 5000 RLU intensity threshold (grey line) are indicated. As internal control, anti-rat IgG spotted on the slides were measured in parallel
Binding of induced antibodies to infected cells
Since prior analysis of antibody binding was mainly based on shorter fragments of the full-length Env, the binding of antisera to native protein as presented on the surface of infected cells should be also investigated. Therefore, C8166 cells were infected with HIV-1, and binding of antisera from animals with strong antibody titres along with a pre-immune rat serum as control was analysed by flow cytometry and indirect immunofluorescence (Fig. 6). Neglectable binding was observed with the control serum, but sera from immunised animals showed profound fluorescence ranging from moderate to strong dependent on the immunised antigen (Fig. 6a). Recognition of infected cells by the serum from group 364 (Bet E1) was rather weak (28.7 %); however, rat sera from groups 365, 366 and 375 reacted intensely with 65.9, 55.5 and 57.9 %, respectively. In indirect immunofluorescence analysis, the same sera gave signals with varying appearance. Antisera of rat 364 containing only FPPR-directed antibodies (Figs. 4, 5) caused rather weak and diffuse staining with no clear pattern. In contrast, all antisera that contained E2-directed antibodies (rats 365, 366, 375) gave spot-like signals distributed on the surface of the cells, most probably showing antibodies binding to the produced or infecting virus (Fig. 6b). This observation was in line with the finding that the gp41 MPER is most accessible in a fusion intermediate state after attachment of gp120 to its cellular receptors [65, 66]. Thus, at least antisera with MPER specificity seemed to be able to react with native Env as mandatory for virus neutralisation.
Binding of induced antisera to HIV-1 pNL-4.3-infected C8166 cells. (a) Analysis of antibody binding by flow cytometry 72 h after infection. Cells were probed with pre-immune rat serum (PI) or antisera from a representative animal from the rat group immunised with the antigen indicated above. (b) Indirect immunofluorescence of C8166 cells 48 h after infection. Fixed cells were transferred to slides where they were incubated with the same sera as in (a). Rat IgGs were detected by an Alexa-Fluor 488 antibody and analysed at a magnification of 20× (upper panel) or 60× (lower panel). No reactivity was observed for the control (Ctrl), but antisera showed either diffuse staining when containing FPPR-targeting antibodies (rats 364) or distinct spots predominantly on the cell surface in antisera where MPER-directed antibodies were present (rats 365, 366 and 375)
Analysis of neutralising activity
Although some of the immunised animals developed antibodies binding to epitopes overlapping with the 2F5 epitope and bound to virus particles, all sera did not prevent HIV-1 infection in a TZM-bl indicator cell line-based neutralisation assay when used at the dilution of 1:50 (Online Resource 6). 2F5 was effectively inhibiting infection.
Discussion
In the present study a novel strategy for HIV-1 epitope delivery based on FFV Bet/HIV-gp41 fusion proteins containing the epitopes of the bnAb 2F5 and 4E10 was evaluated. Milligram amounts of these proteins as needed for immunisation and antigen characterisation were produced in E. coli, purified to homogeneity and renatured to obtain soluble molecules. The antigens specifically reacted with 2F5, 4E10 and a serum against the E1 domain and were immunogenic in rats generating antibodies targeting an epitope overlapping with the 2F5 epitope; however, they did not prevent HIV-1 infection in a TZM-bl-based neutralisation assay. The antigens comprised the HIV-1 MPER-derived E2 domain because the corresponding bnAb are characterised by strong somatic mutations which could benefit from prolonged antigen delivery by replicating vectors [67]. Additionally, some Bet hybrids contained the FPPR-derived E1 domain and the E2 domain linked by a loop to stabilise a MPER conformation that increases 2F5 binding [53, 54, 56, 57]. Introducing the MPER domain alone resulted in high-affinity recognition by 2F5, with values comparable to or better than previously reported for MPER peptides or MPER epitope scaffolds [47, 68, 69]. Unexpectedly, the affinity of 2F5 to its epitope decreased to some extent when the FPPR residues were present (Table 1). This suggests that the selected positioning of the FPPR residues might not allow proper orientation of the 2F5 epitope or formation of the affinity enhancing β-turn structure and might result from a divergence between the bioinformatic predictions used to design this antigen and the actual structure. However, by stepwise optimisation of the FPPR and MPER epitope presentation with an extended set of loop antigens, enhanced antigenicity and better binding to 2F5 was observed in later experiments (Mühle et al., manuscript in preparation). In contrast to 2F5, the FPPR residues remarkably improved 4E10 binding (Fig. 2; Table 1). Crystallisation experiments with 2F5-Fab have shown that the presence of FPPR residues not only stabilised the β-turn structure of the 2F5 epitope but also induced the formation of α-helices in the adjacent C-terminal residues [56]. Such an increase in α-helical content might explain the higher affinity of 4E10 to the loop construct used here, since enhanced binding of 4E10 to α-helical peptides has been reported [50]. However, the affinity of 4E10 to the loop antigen was still weak when compared to 2F5 (223 nM compared to 19 nM, Table 1) or previous antigens designed for induction of 4E10-like antibodies [49, 50] and potentially explains the 2F5 epitope-dominated response observed (Fig. 5). For improvement, the E2 domain may be extended by at least three additional amino acids (YIK) to increase the affinity of 4E10 [49, 50].
When used as protein immunogen, Bet was found to be an effective carrier molecule for the small HIV-1 epitopes, triggering a profound humoral response to the introduced inserts (Fig. 4, Online Resource 4). Importantly, antibodies targeting ELDKWA and binding to gp41 and virions were induced, particularly with the Bet-E2 antigen which also showed the highest affinity to 2F5 and the strongest antibody response against the MPER (Table 1; Fig. 4). Despite the high titres of binding antibodies, none of the immune sera were neutralising, indicating that high affinity and immunogenicity do not predict the ability of antigens to induce antibodies capable of preventing HIV-1 infection as was found also by others [47, 48, 68, 70]. It is difficult to judge whether the antigens used here did not provide a conformation able to induce neutralising antibodies or whether their amount in the antibody population was too low to be detected. To further increase MPER-specific responses, certain mutations that have been recently described to enhance its immunogenicity could be beneficial [71]. Also, in order to avoid the FPPR-dominated responses observed here when the E1 domain was present, introduction of a glycosylation site into the FPPR might be considered to immunologically silence the E1 reactivity and focus the immune system to the MPER region [72]. As the antisera were binding to virus particles (Fig. 6) but were not neutralising, one could speculate that the induced antibodies lack certain characteristics like (1) lipid binding, which is not promoted by the strategy used here, or (2) prolonged CDRH3 loops, which could arise as a result of somatic mutation after extended B cell stimulation. Whereas the first point will have to be tested with other strategies, for example, by placing the HIV-1 MPER epitope into the TM protein of FV, the second question could be addressed by extended antigen delivery. This might be achieved in part by extended boosting as shown for a recombinant gp120 antigen inducing a limited degree of affinity maturation [9] or more efficiently by using replicating FV vectors which permanently express the antigen. Thus, after showing the general feasibility of using Bet/HIV-1 fusion proteins for induction of HIV-1 MPER-specific antibodies, optimised hybrid proteins should now be expressed in replication-competent virus and the expression of the HIV epitopes monitored in infected eukaryotic cells. These replication-competent viruses may first be used in cats in order to analyse whether prolonged and systemic delivery of such proteins induce not only antibodies binding to epitopes overlapping with the 2F5 epitope, but also antibodies neutralising HIV-1. If successful, this would justify transfer to primate FV and testing in non-human primates.
References
Burton DR, Weiss RA. AIDS/HIV. A boost for HIV vaccine design. Science. 2010;329(5993):770–3. doi:10.1126/science.1194693.
Mascola JR, Montefiori DC. The role of antibodies in HIV vaccines. Annu Rev Immunol. 2010;28:413–44. doi:10.1146/annurev-immunol-030409-101256.
Burton DR, Stanfield RL, Wilson IA. Antibody vs. HIV in a clash of evolutionary titans. Proc Natl Acad Sci USA. 2005;102(42):14943–8. doi:10.1073/pnas.0505126102.
Doria-Rose NA, Klein RM, Manion MM, O’Dell S, Phogat A, Chakrabarti B, et al. Frequency and phenotype of human immunodeficiency virus envelope-specific B cells from patients with broadly cross-neutralizing antibodies. J Virol. 2009;83(1):188–99. doi:10.1128/JVI.01583-08.
Gray ES, Madiga MC, Hermanus T, Moore PL, Wibmer CK, Tumba NL, et al. The neutralization breadth of HIV-1 develops incrementally over four years and is associated with CD4 + T cell decline and high viral load during acute infection. J Virol. 2011;85(10):4828–40. doi:10.1128/JVI.00198-11.
Haynes BF, Kelsoe G, Harrison SC, Kepler TB. B-cell-lineage immunogen design in vaccine development with HIV-1 as a case study. Nat Biotechnol. 2012;30(5):423–33. doi:10.1038/nbt.2197.
McMichael AJ, Borrow P, Tomaras GD, Goonetilleke N, Haynes BF. The immune response during acute HIV-1 infection: clues for vaccine development. Nat Rev Immunol. 2010;10(1):11–23. doi:10.1038/nri2674.
Burton DR, Desrosiers RC, Doms RW, Koff WC, Kwong PD, Moore JP, et al. HIV vaccine design and the neutralizing antibody problem. Nat Immunol. 2004;5(3):233–6. doi:10.1038/ni0304-233.
Moody MA, Yates NL, Amos JD, Drinker MS, Eudailey JA, Gurley TC, et al. HIV-1 gp120 Vaccine Induces Affinity Maturation in both New and Persistent Antibody Clonal Lineages. J Virol. 2012;86(14):7496–507. doi:10.1128/JVI.00426-12.
Hansen SG, Ford JC, Lewis MS, Ventura AB, Hughes CM, Coyne-Johnson L, et al. Profound early control of highly pathogenic SIV by an effector memory T-cell vaccine. Nature. 2011;473(7348):523–7. doi:10.1038/nature10003.
Hansen SG, Vieville C, Whizin N, Coyne-Johnson L, Siess DC, Drummond DD, et al. Effector memory T cell responses are associated with protection of rhesus monkeys from mucosal simian immunodeficiency virus challenge. Nat Med. 2009;15(3):293–9. doi:10.1038/nm.1935.
Johnson RP, Desrosiers RC. Protective immunity induced by live attenuated simian immunodeficiency virus. Curr Opin Immunol. 1998;10(4):436–43.
Koff WC, Johnson PR, Watkins DI, Burton DR, Lifson JD, Hasenkrug KJ, et al. HIV vaccine design: insights from live attenuated SIV vaccines. Nat Immunol. 2006;7(1):19–23. doi:10.1038/ni1296.
Picker LJ, Hansen SG, Lifson JD. New paradigms for HIV/AIDS vaccine development. Annu Rev Med. 2012;63:95–111. doi:10.1146/annurev-med-042010-085643.
Lindemann D, Rethwilm A. Foamy virus biology and its application for vector development. Viruses. 2011;3(5):561–85. doi:10.3390/v3050561.
Linial ML. Foamy viruses are unconventional retroviruses. J Virol. 1999;73(3):1747–55.
Rethwilm A. The replication strategy of foamy viruses. Curr Top Microbiol Immunol. 2003;277:1–26.
Trobridge GD, Miller DG, Jacobs MA, Allen JM, Kiem HP, Kaul R, et al. Foamy virus vector integration sites in normal human cells. Proc Natl Acad Sci USA. 2006;103(5):1498–503. doi:10.1073/pnas.0510046103.
Bauer TR Jr, Olson EM, Huo Y, Tuschong LM, Allen JM, Li Y, et al. Treatment of canine leukocyte adhesion deficiency by foamy virus vectors expressing CD18 from a PGK promoter. Gene Ther. 2011;18(6):553–9. doi:10.1038/gt.2010.169.
Erlwein O, McClure MO. Progress and prospects: foamy virus vectors enter a new age. Gene Ther. 2010;17(12):1423–9. doi:10.1038/gt.2010.95.
Taylor JA, Vojtech L, Bahner I, Kohn DB, Laer DV, Russell DW, et al. Foamy virus vectors expressing anti-HIV transgenes efficiently block HIV-1 replication. Mol Ther. 2008;16(1):46–51. doi:10.1038/sj.mt.6300335.
Trobridge GD. Foamy virus vectors for gene transfer. Expert Opin Biol Ther. 2009;9(11):1427–36. doi:10.1517/14712590903246388.
Bastone P, Romen F, Liu W, Wirtz R, Koch U, Josephson N, et al. Construction and characterization of efficient, stable and safe replication-deficient foamy virus vectors. Gene Ther. 2007;14(7):613–20. doi:10.1038/sj.gt.3302890.
Schwantes A, Ortlepp I, Löchelt M. Construction and functional characterization of feline foamy virus-based retroviral vectors. Virology. 2002;301(1):53–63.
Schwantes A, Truyen U, Weikel J, Weiss C, Löchelt M. Application of chimeric feline foamy virus-based retroviral vectors for the induction of antiviral immunity in cats. J Virol. 2003;77(14):7830–42.
Zemba M, Alke A, Bodem J, Winkler IG, Flower RL, Pfrepper K, et al. Construction of infectious feline foamy virus genomes: cat antisera do not cross-neutralize feline foamy virus chimera with serotype-specific Env sequences. Virology. 2000;266(1):150–6. doi:10.1006/viro.1999.0037.
Alke A, Schwantes A, Zemba M, Flügel RM, Löchelt M. Characterization of the humoral immune response and virus replication in cats experimentally infected with feline foamy virus. Virology. 2000;275(1):170–6. doi:10.1006/viro.2000.0537.
Bleiholder A, Mühle M, Hechler T, Bevins S, vandeWoude S, Denner J, et al. Pattern of seroreactivity against feline foamy virus proteins in domestic cats from Germany. Vet Immunol Immunopathol. 2011;143(3–4):292–300. doi:10.1016/j.vetimm.2011.06.007.
Romen F, Pawlita M, Sehr P, Bachmann S, Schroder J, Lutz H, et al. Antibodies against Gag are diagnostic markers for feline foamy virus infections while Env and Bet reactivity is undetectable in a substantial fraction of infected cats. Virology. 2006;345(2):502–8. doi:10.1016/j.virol.2005.10.022.
Giron ML, de The H, Saib A. An evolutionarily conserved splice generates a secreted env-Bet fusion protein during human foamy virus infection. J Virol. 1998;72(6):4906–10.
Lecellier CH, Vermeulen W, Bachelerie F, Giron ML, Saib A. Intra- and intercellular trafficking of the foamy virus auxiliary bet protein. J Virol. 2002;76(7):3388–94.
Montero M, van Houten NE, Wang X, Scott JK. The membrane-proximal external region of the human immunodeficiency virus type 1 envelope: dominant site of antibody neutralization and target for vaccine design. Microbiol Mol Biol Rev. 2008;72(1):54–84, table of contents. doi:10.1128/MMBR.00020-07.
Muster T, Steindl F, Purtscher M, Trkola A, Klima A, Himmler G, et al. A conserved neutralizing epitope on gp41 of human immunodeficiency virus type 1. J Virol. 1993;67(11):6642–7.
Salzwedel K, West JT, Hunter E. A conserved tryptophan-rich motif in the membrane-proximal region of the human immunodeficiency virus type 1 gp41 ectodomain is important for Env-mediated fusion and virus infectivity. J Virol. 1999;73(3):2469–80.
Zwick MB, Labrijn AF, Wang M, Spenlehauer C, Saphire EO, Binley JM, et al. Broadly neutralizing antibodies targeted to the membrane-proximal external region of human immunodeficiency virus type 1 glycoprotein gp41. J Virol. 2001;75(22):10892–905. doi:10.1128/JVI.75.22.10892-10905.2001.
Biron Z, Khare S, Samson AO, Hayek Y, Naider F, Anglister J. A monomeric 3(10)-helix is formed in water by a 13-residue peptide representing the neutralizing determinant of HIV-1 on gp41. Biochemistry. 2002;41(42):12687–96.
Bryson S, Julien JP, Isenman DE, Kunert R, Katinger H, Pai EF. Crystal structure of the complex between the F(ab)’ fragment of the cross-neutralizing anti-HIV-1 antibody 2F5 and the F(ab) fragment of its anti-idiotypic antibody 3H6. J Mol Biol. 2008;382(4):910–9. doi:10.1016/j.jmb.2008.07.057.
Buzon V, Natrajan G, Schibli D, Campelo F, Kozlov MM, Weissenhorn W. Crystal structure of HIV-1 gp41 including both fusion peptide and membrane proximal external regions. PLoS Pathog. 2010;6(5):e1000880. doi:10.1371/journal.ppat.1000880.
Ofek G, Tang M, Sambor A, Katinger H, Mascola JR, Wyatt R, et al. Structure and mechanistic analysis of the anti-human immunodeficiency virus type 1 antibody 2F5 in complex with its gp41 epitope. J Virol. 2004;78(19):10724–37. doi:10.1128/JVI.78.19.10724-10737.2004.
Schibli DJ, Montelaro RC, Vogel HJ. The membrane-proximal tryptophan-rich region of the HIV glycoprotein, gp41, forms a well-defined helix in dodecylphosphocholine micelles. Biochemistry. 2001;40(32):9570–8.
Sun ZY, Oh KJ, Kim M, Yu J, Brusic V, Song L, et al. HIV-1 broadly neutralizing antibody extracts its epitope from a kinked gp41 ectodomain region on the viral membrane. Immunity. 2008;28(1):52–63. doi:10.1016/j.immuni.2007.11.018.
Alam SM, Morelli M, Dennison SM, Liao HX, Zhang R, Xia SM, et al. Role of HIV membrane in neutralization by two broadly neutralizing antibodies. Proc Natl Acad Sci USA. 2009;106(48):20234–9. doi:10.1073/pnas.0908713106.
Scherer EM, Leaman DP, Zwick MB, McMichael AJ, Burton DR. Aromatic residues at the edge of the antibody combining site facilitate viral glycoprotein recognition through membrane interactions. Proc Natl Acad Sci USA. 2010;107(4):1529–34. doi:10.1073/pnas.0909680107.
Julien JP, Bryson S, Nieva JL, Pai EF. Structural details of HIV-1 recognition by the broadly neutralizing monoclonal antibody 2F5: epitope conformation, antigen-recognition loop mobility, and anion-binding site. J Mol Biol. 2008;384(2):377–92. doi:10.1016/j.jmb.2008.09.024.
Coeffier E, Clement JM, Cussac V, Khodaei-Boorane N, Jehanno M, Rojas M, et al. Antigenicity and immunogenicity of the HIV-1 gp41 epitope ELDKWA inserted into permissive sites of the MalE protein. Vaccine. 2000;19(7–8):684–93.
Ho J, MacDonald KS, Barber BH. Construction of recombinant targeting immunogens incorporating an HIV-1 neutralizing epitope into sites of differing conformational constraint. Vaccine. 2002;20(7–8):1169–80.
Ho J, Uger RA, Zwick MB, Luscher MA, Barber BH, MacDonald KS. Conformational constraints imposed on a pan-neutralizing HIV-1 antibody epitope result in increased antigenicity but not neutralizing response. Vaccine. 2005;23(13):1559–73. doi:10.1016/j.vaccine.2004.09.037.
McGaughey GB, Citron M, Danzeisen RC, Freidinger RM, Garsky VM, Hurni WM, et al. HIV-1 vaccine development: constrained peptide immunogens show improved binding to the anti-HIV-1 gp41 MAb. Biochemistry. 2003;42(11):3214–23. doi:10.1021/bi026952u.
Brunel FM, Zwick MB, Cardoso RM, Nelson JD, Wilson IA, Burton DR, et al. Structure-function analysis of the epitope for 4E10, a broadly neutralizing human immunodeficiency virus type 1 antibody. J Virol. 2006;80(4):1680–7. doi:10.1128/JVI.80.4.1680-1687.2006.
Cardoso RM, Brunel FM, Ferguson S, Zwick M, Burton DR, Dawson PE, et al. Structural basis of enhanced binding of extended and helically constrained peptide epitopes of the broadly neutralizing HIV-1 antibody 4E10. J Mol Biol. 2007;365(5):1533–44. doi:10.1016/j.jmb.2006.10.088.
Cardoso RM, Zwick MB, Stanfield RL, Kunert R, Binley JM, Katinger H, et al. Broadly neutralizing anti-HIV antibody 4E10 recognizes a helical conformation of a highly conserved fusion-associated motif in gp41. Immunity. 2005;22(2):163–73. doi:10.1016/j.immuni.2004.12.011.
Bellamy-McIntyre AK, Lay CS, Baar S, Maerz AL, Talbo GH, Drummer HE, et al. Functional links between the fusion peptide-proximal polar segment and membrane-proximal region of human immunodeficiency virus gp41 in distinct phases of membrane fusion. J Biol Chem. 2007;282(32):23104–16. doi:10.1074/jbc.M703485200.
Fiebig U, Schmolke M, Eschricht M, Kurth R, Denner J. Mode of interaction between the HIV-1-neutralizing monoclonal antibody 2F5 and its epitope. AIDS. 2009;23(8):887–95. doi:10.1097/QAD.0b013e3283292153.
Lorizate M, Gomara MJ, de la Torre BG, Andreu D, Nieva JL. Membrane-transferring sequences of the HIV-1 Gp41 ectodomain assemble into an immunogenic complex. J Mol Biol. 2006;360(1):45–55. doi:10.1016/j.jmb.2006.04.056.
Noah E, Biron Z, Naider F, Arshava B, Anglister J. The membrane proximal external region of the HIV-1 envelope glycoprotein gp41 contributes to the stabilization of the six-helix bundle formed with a matching N’ peptide. Biochemistry. 2008;47(26):6782–92. doi:10.1021/bi7023139.
de la Arada I, Julien JP, de la Torre BG, Huarte N, Andreu D, Pai EF, et al. Structural constraints imposed by the conserved fusion peptide on the HIV-1 gp41 epitope recognized by the broadly neutralizing antibody 2F5. J Phys Chem B. 2009;113(41):13626–37. doi:10.1021/jp905965h.
Lorizate M, de la Arada I, Huarte N, Sanchez-Martinez S, de la Torre BG, Andreu D, et al. Structural analysis and assembly of the HIV-1 Gp41 amino-terminal fusion peptide and the pretransmembrane amphipathic-at-interface sequence. Biochemistry. 2006;45(48):14337–46. doi:10.1021/bi0612521.
Mühle M, Löchelt M, Denner J. Optimisation of expression and purification of the feline and primate foamy virus transmembrane envelope proteins using a 96 deep well screen. Protein Expr Purif. 2012;81(1):96–105. doi:10.1016/j.pep.2011.09.006.
Frey A, Di Canzio J, Zurakowski D. A statistically defined endpoint titer determination method for immunoassays. J Immunol Methods. 1998;221(1–2):35–41.
Mühle M, Bleiholder A, Kolb S, Hübner J, Löchelt M, Denner J. Immunological properties of the transmembrane envelope protein of the feline foamy virus and its use for serological screening. Virology. 2011;412(2):333–40. doi:10.1016/j.virol.2011.01.023.
Fezoui Y, Weaver DL, Osterhout JJ. De novo design and structural characterization of an alpha-helical hairpin peptide: a model system for the study of protein folding intermediates. Proc Natl Acad Sci USA. 1994;91(9):3675–9.
Fezoui Y, Connolly PJ, Osterhout JJ. Solution structure of alpha t alpha, a helical hairpin peptide of de novo design. Protein Sci. 1997;6(9):1869–77. doi:10.1002/pro.5560060907.
Gallo SA, Finnegan CM, Viard M, Raviv Y, Dimitrov A, Rawat SS, et al. The HIV Env-mediated fusion reaction. Biochim Biophys Acta. 2003;1614(1):36–50.
Zwick MB, Jensen R, Church S, Wang M, Stiegler G, Kunert R, et al. Anti-human immunodeficiency virus type 1 (HIV-1) antibodies 2F5 and 4E10 require surprisingly few crucial residues in the membrane-proximal external region of glycoprotein gp41 to neutralize HIV-1. J Virol. 2005;79(2):1252–61. doi:10.1128/JVI.79.2.1252-1261.2005.
Frey G, Chen J, Rits-Volloch S, Freeman MM, Zolla-Pazner S, Chen B. Distinct conformational states of HIV-1 gp41 are recognized by neutralizing and non-neutralizing antibodies. Nat Struct Mol Biol. 2010;17(12):1486–91. doi:10.1038/nsmb.1950.
Frey G, Peng H, Rits-Volloch S, Morelli M, Cheng Y, Chen B. A fusion-intermediate state of HIV-1 gp41 targeted by broadly neutralizing antibodies. Proc Natl Acad Sci USA. 2008;105(10):3739–44. doi:10.1073/pnas.0800255105.
Zhu Z, Qin HR, Chen W, Zhao Q, Shen X, Schutte R, et al. Cross-reactive HIV-1-neutralizing human monoclonal antibodies identified from a patient with 2F5-like antibodies. J Virol. 2011;85(21):11401–8. doi:10.1128/JVI.05312-11.
Guenaga J, Dosenovic P, Ofek G, Baker D, Schief WR, Kwong PD, et al. Heterologous epitope-scaffold prime: boosting immuno-focuses B cell responses to the HIV-1 gp41 2F5 neutralization determinant. PLoS ONE. 2011;6(1):e16074. doi:10.1371/journal.pone.0016074.
Ofek G, Guenaga FJ, Schief WR, Skinner J, Baker D, Wyatt R, et al. Elicitation of structure-specific antibodies by epitope scaffolds. Proc Natl Acad Sci USA. 2010;107(42):17880–7. doi:10.1073/pnas.1004728107.
McGaughey GB, Barbato G, Bianchi E, Freidinger RM, Garsky VM, Hurni WM, et al. Progress towards the development of a HIV-1 gp41-directed vaccine. Curr HIV Res. 2004;2(2):193–204.
Wang J, Tong P, Lu L, Zhou L, Xu L, Jiang S, et al. HIV-1 gp41 core with exposed membrane-proximal external region inducing broad HIV-1 neutralizing antibodies. PLoS ONE. 2011;6(3):e18233. doi:10.1371/journal.pone.0018233.
Garrity RR, Rimmelzwaan G, Minassian A, Tsai WP, Lin G, de Jong JJ, et al. Refocusing neutralizing antibody response by targeted dampening of an immunodominant epitope. J Immunol. 1997;159(1):279–89.
Acknowledgments
This work was supported by the Volkswagenstiftung. We thank C.M. Schmidt, M. Keller and D. Wirths for excellent technical support and A. Bleiholder for critical reading of the manuscript. All authors have approved the final article and declare that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mühle, M., Hoffmann, K., Löchelt, M. et al. Immunisation with foamy virus Bet fusion proteins as novel strategy for HIV-1 epitope delivery. Immunol Res 56, 61–72 (2013). https://doi.org/10.1007/s12026-013-8387-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-013-8387-x