Abstract
Renalase is considered as a novel candidate gene for type 2 diabetes. In this study, we aimed to investigate the relationship of serum renalase and two single nucleotide polymorphisms with gestational diabetes mellitus. One hundred and ninety-eight normotensive pregnant females (n = 99 gestational diabetes mellitus; n = 99 euglycemic pregnant controls) were classified according to the International Association of the Diabetes and Pregnancy Study criteria. Fasting and 2-h post glucose load blood levels and anthropometric assessment was performed. Serum renalase was measured using enzyme-linked immunosorbent assay, whereas DNA samples were genotyped for renalase single nucleotide polymorphisms rs2576178 and rs10887800 using Polymerase chain reaction-Restriction fragment length polymorphism method. In an age-matched case control study, no difference was observed in the serum levels of renalase (p > 0.05). The variant rs10887800 showed an association with gestational diabetes mellitus and remained significant after multiple adjustments (p < 0.05), whereas rs2576178 showed weak association (p = 0.030) that was lost after multiple adjustments (p = 0.09). We inferred a modest association of the rs10887800 polymorphism with gestational diabetes. Although gestational diabetes mellitus is self-reversible, yet presence of this minor G allele might predispose to metabolic syndrome phenotypes in near the future.

Similar content being viewed by others
References
R. Seshadri, American diabetes association gestational diabetes mellitus. Diabetes Care 25, S94–S96 (2002)
R. Iqbal, G. Rafique, S. Badruddin, R. Qureshi, R. Cue, K. Gray-Donald, Increased body fat percentage and physical inactivity are independent predictors of gestational diabetes mellitus in South Asian women. Eur. J. Clin. Nutr. 61(6), 736–742 (2007)
T.A. Buchanan, A.H. Xiang, K.A. Page, Gestational diabetes mellitus: risks and management during and after pregnancy. Nat. Rev. Endocrinol. 8(11), 639–649 (2012)
ADA, Standards of medical care in diabetes—2015 abridged for primary care providers. Clin. Diabetes 33(2), 97–111 (2015)
S. Johansson, E. Villamor, M. Altman, A.-K.E. Bonamy, F. Granath, S. Cnattingius, Maternal overweight and obesity in early pregnancy and risk of infant mortality: a population based cohort study in Sweden. Br. Med. J. 349, g6572 (2014)
L. Bellamy, J.-P. Casas, A.D. Hingorani, D. Williams, Type 2 diabetes mellitus after gestational diabetes: a systematic review and meta-analysis. The Lancet. 373(9677), 1773–1779 (2009)
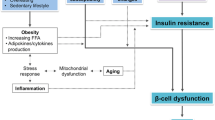
M. Qatanani, M.A. Lazar, Mechanisms of obesity-associated insulin resistance: many choices on the menu. Genes Dev. 21(12), 1443–1455 (2007)
S.S. Fatima, F. Alam, B. Chaudhry, T.A. Khan, Elevated levels of chemerin, leptin and interleukin–18 in gestational diabetes mellitus. J. Matern. Fetal Neonatal Med.: pp. 1–18 (2016) (just accepted)
K.F. Petersen, G.I. Shulman, Etiology of insulin resistance. Am. J. Med. 119(5), S10–S16 (2006)
J. Zheng, X. Xiao, Q. Zhang, L. Mao, M. Li, M. Yu, J. Xu, Y. Wang, Correlation of high-molecular-weight adiponectin and leptin concentrations with anthropometric parameters and insulin sensitivity in newborns. Int. J. Endocrinol. (2014). doi:10.1155/2014/435376
R. Wildman, P. Muntner, K. Reynolds, A. McGinn, S. Rajpathak, J. Wylie-Rosett, M. Sowers, The obese without cardiometabolic risk factor clustering and The Journal of Korean Diabetes the normal weight with cardiometabolic risk factor clustering: prevalence and correlates of 2 phenotypes among the US population (NHANES 1999-2004). Arch. Intern. Med. 168, 1617–1624 (2008)
C. Zhang, W. Bao, Y. Rong, H. Yang, K. Bowers, E. Yeung, M. Kiely, Genetic variants and the risk of gestational diabetes mellitus: a systematic review. Hum. Reprod. Update: dmt013 (2013).
G.V. Desir, A.J. Peixoto, Renalase in hypertension and kidney disease. Nephrol. Dial. Transplant. 29(1), 22–28 (2013)
G. Desir, Novel insights into the physiology of renalase and its role in hypertension and heart disease. Pediatr. Nephrol. 27(5), 719–725 (2012)
G.V. Desir, Regulation of blood pressure and cardiovascular function by renalase. Kidney Int. 76(4), 366–370 (2009)
S. Hennebry, N. Eikelis, F. Socratous, G. Desir, G. Lambert, M. Schlaich, Renalase, a novel soluble FAD-dependent protein, is synthesized in the brain and peripheral nerves.. Mol. Psychiatry 15(3), 234–236 (2010)
F.C. Luft, Renalase, a catecholamine-metabolizing hormone from the kidney. Cell Metab. 1(6), 358–360 (2005)
M. Ramanjaneya, B. Tan, V. Patel, J. Hu, H. Lehnert, E. Hillhouse, J. Chen, H. Randeva, Renalase a key regulator of brown adipose tissue activity. Endocrine Abstracts 34: P263 (2014)
J. Xu, G. Li, P. Wang, H. Velazquez, X. Yao, Y. Li, Y. Wu, A. Peixoto, S. Crowley, G.V. Desir, Renalase is a novel, soluble monoamine oxidase that regulates cardiac function and blood pressure. J. Clin. Invest. 115(5), 1275–1280 (2005)
M. Milani, F. Ciriello, S. Baroni, V. Pandini, G. Canevari, M. Bolognesi, A. Aliverti, FAD-binding site and NADP reactivity in human renalase: a new enzyme involved in blood pressure regulation. J. Mol. Biol. 411(2), 463–473 (2011)
X. Guo, L. Wang, H. Velazquez, R. Safirstein, G.V. Desir, Renalase: its role as a cytokine, and an update on its association with type 1 diabetes and ischemic stroke. Curr. Opin. Nephrol. Hypertens. 23(5), 513–518 (2014)
K.A. Elsetohy, M.A. Al-Ghussein, D. Sabry, A.M. Nada, A.A. Eldaly, A.H. Wahba, Are renalase rs2576178 and rs10887800 polymorphisms associated with pregnancy-induced hypertension? Mother and Child 2, 3 (2014)
M. Buraczynska, P. Zukowski, K. Buraczynska, S. Mozul, A. Ksiazek, Renalase gene polymorphisms in patients with type 2 diabetes, hypertension and stroke. NeuroMolecular. Med. 13(4), 321–327 (2011)
J.C. Barrett, D.G. Clayton, P. Concannon, B. Akolkar, J.D. Cooper, H.A. Erlich, C. Julier, G. Morahan, J. Nerup, C. Nierras, Genome-wide association study and meta-analysis find that over 40 loci affect risk of type 1 diabetes. Nat. Genet. 41(6), 703–707 (2009)
M.P.L. Reddy, H. Wang, S. Liu, B. Bode, J.C. Reed, R.D. Steed, S.W. Anderson, L. Steed, D. Hopkins, J.-X. She, Association between type 1 diabetes and GWAS SNPs in the southeast US Caucasian population. Genes Immun. 12(3), 208–212 (2011)
I.C. Panel, International Association of Diabetes and Pregnancy Study Groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 33(3), 676–682 (2010)
C. Snehalatha, V. Viswanathan, A. Ramachandran, Cutoff values for normal anthropometric variables in Asian Indian adults. Diabetes Care 26(5), 1380–1384 (2003)
E. Koc-Zorawska, J. Malyszko, E. Zbroch, J. Malyszko, M. Mysliwiec, Vascular adhesion protein-1 and renalase in regard to diabetes in hemodialysis patients. Arch. Med. Sci. 8(6), 1048–1052 (2012)
E.W. Gaber, H.A. El-Attar, The relationship between dopamine and renalase in type 2 diabetic patients with and without diabetic nephropathy. Br. J. Diabetes Vasc. Dis. 13(3), 130–137 (2013)
M. Zhou, T. Liang, Y. Wang, D. Jin, J. Wang, L. Jia, S. Zhang, Expression and tissue localization of renalase, a novel soluble FAD-dependent protein, in reproductive/steroidogenic systems. Mol. Biol. Rep. 40(6), 3987–3994 (2013)
B. Bagci, S. Karakus, G. Bagci, E. Sancakdar, Renalase gene polymorphism is associated with increased blood pressure in preeclampsia. Pregn. Hypertens. 6(2), 115–120 (2016)
M. Karachaliou, V. Georgiou, T. Roumeliotaki, G. Chalkiadaki, V. Daraki, S. Koinaki, E. Dermitzaki, K. Sarri, M. Vassilaki, M. Kogevinas, Association of trimester-specific gestational weight gain with fetal growth, offspring obesity, and cardiometabolic traits in early childhood. Am. J. Obstet. Gynecol. 212(4), 502. e1-502. e14 (2015)
J.L. Johnson, S.L. Farr, P.M. Dietz, A.J. Sharma, W.D. Barfield, C.L. Robbins, Trends in gestational weight gain: the pregnancy risk assessment monitoring system, 2000–2009. Am. J. Obstet. Gynecol. 212(6), 806. e1–806. e8 (2015)
C. Wallace, M. Rotival, J.D. Cooper, C.M. Rice, J.H. Yang, M. McNeill, D.J. Smyth, D. Niblett, F. Cambien, L. Tiret, Statistical colocalisation of monocyte gene expression and genetic risk variants for type 1 diabetes. Hum. Mol. Gen. 15(21(12)), 2815–2824 (2012)
J.M. Howson, J.D. Cooper, D.J. Smyth, N.M. Walker, H. Stevens, J.-X. She, G.S. Eisenbarth, M. Rewers, J.A. Todd, B. Akolkar, Evidence of gene-gene interaction and age-at-diagnosis effects in type 1 diabetes. Diabetes 61(11), 3012–3017 (2012)
X. Guo, L. Hollander, D. MacPherson, L. Wang, H. Velazquez, J. Chang, R. Safirstein, C. Cha, F. Gorelick, G.V. Desir, Inhibition of renalase expression and signaling has antitumor activity in pancreatic cancer. Sci. Rep. 14(6), 22996 (2016)
Funding source
Department of Biological and Biomedical Sciences, Research Module Fund.
Author contributions
SSF conceived and designed the study. HZM, SM, NR, MSA, AR, TS carried out the literature search, data collection, experimental analysis. SSF, FA, ZJ, and HZ were involved in manuscript writing. The final manuscript was approved by all authors for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Zehra Jamil and Faiza Alam have contributed equally to this work.
Hajira Zafar Malik, Sarosh Madhani, Noman Rehmani, Muhammad Saad Ahmad, Amna Rabbani, and Tayyab Shabbir have contributed equally to this work.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Fatima, S.S., Jamil, Z., Alam, F. et al. Polymorphism of the renalase gene in gestational diabetes mellitus. Endocrine 55, 124–129 (2017). https://doi.org/10.1007/s12020-016-1058-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-016-1058-7