Abstract
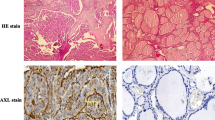
The purpose of this work is to study the activation of the hedgehog signalling pathway is associated with tumour progression in various types of cancer, hence the development of specific antagonists raises hope for new therapeutic strategies. Therefore, the expression of hedgehog pathway components in anaplastic thyroid cancer (ATC) and effects of the hedgehog inhibitor Cyclopamine on ATC cells were investigated in this study. Expression of the ligand Sonic Hedgehog (SHh), the transmembrane protein Smoothened (Smo), the receptor Patched (Ptc) and the target gene Gli-1 was evaluated in two ATC cell lines (Hth 74, C643) by RT-PCR and in tumour specimens by immunohistochemistry. The corresponding gene products were examined by western blotting analysis. After treatment with different concentrations of Cyclopamine the time-dependent course of cell viability in ATC cell lines was evaluated by MTT assay. SHh, Smo, Ptc and Gli were clearly expressed on mRNA and protein levels in both cell lines and in tumour samples (41 %SHh, 65 %Smo, 65 %Ptc and 65 %Gli). Treatment with Cyclopamine showed a time- and dose-dependent inhibition of cell numbers with IC50 values between 1 and 4 μM in both cell lines, comparable to other types of cancer. In conclusion, we believe that the hedgehog pathway is expressed in anaplastic thyroid carcinoma specimens and proliferation of ATC cell lines can be influenced by the Hh inhibitor Cyclopamine. Aberrant activation of this pathway might be involved in the aggressive biology of anaplastic cancer and further evaluation regarding a possible clinical impact of pathway inhibition is warranted.



Similar content being viewed by others
References
B. Aschebrook-Kilfoy, M.H. Ward, M.M. Sabra, S.S. Devesa, Thyroid cancer incidence patterns in the United States by histologic type, 1992–2006. Thyroid 21(2), 125–134 (2011). doi:10.1089/thy.2010.0021
F. Perri, G.D. Lorenzo, G.D. Scarpati, C. Buonerba, Anaplastic thyroid carcinoma: a comprehensive review of current and future therapeutic options. World J. Clin. Oncol. 2(3), 150–157 (2011). doi:10.5306/wjco.v2.i3.150
Y.H. Bian, S.H. Huang, L. Yang, X.L. Ma, J.W. Xie, H.W. Zhang, Sonic hedgehog-Gli1 pathway in colorectal adenocarcinomas. World J. Gastroenterol. 13(11), 1659–1665 (2007)
J. Dong, M.R. Gailani, S.L. Pomeroy, D. Reardon, A.E. Bale, Identification of PATCHED mutations in medulloblastomas by direct sequencing. Hum. Mutat. 16(1), 89–90 (2000). doi:10.1002/1098-1004(200007)16:1<89:AID-HUMU18>3.0.CO;2-7
L. Ghali, S.T. Wong, J. Green, N. Tidman, A.G. Quinn, Gli1 protein is expressed in basal cell carcinomas, outer root sheath keratinocytes and a subpopulation of mesenchymal cells in normal human skin. J. Invest. Dermatol. 113(4), 595–599 (1999). doi:10.1046/j.1523-1747.1999.00729.x
K.T. Jang, K.T. Lee, J.G. Lee, S.H. Choi, J.S. Heo, D.W. Choi, G. Ahn, Immunohistochemical expression of Sonic hedgehog in intraductal papillary mucinous tumor of the pancreas. Appl. Immunohistochem. Mol. Morphol. 15(3), 294–298 (2007). doi:10.1097/01.pai.0000213132.71041.da
M.S. Liu, P.Y. Yang, T.S. Yeh, Sonic hedgehog signalling pathway in pancreatic cystic neoplasms and ductal adenocarcinoma. Pancreas 34(3), 340–346 (2007). doi:10.1097/mpa.0b013e3180333ab5
Z. Yuan, J.A. Goetz, S. Singh, S.K. Ogden, W.J. Petty, C.C. Black, V.A. Memoli, E. Dmitrovsky, D.J. Robbins, Frequent requirement of hedgehog signalling in non-small cell lung carcinoma. Oncogene 26(7), 1046–1055 (2007). doi:10.1038/sj.onc.1209860
J.E.S. Hooper, M.P. Scott, The drosophila patched gene encodes a putative membrane protein required for segmental patterning. Cell 59(4), 751–765 (1989)
V. Marigo et al., Biochemical evidence that patched is the hedgehog receptor. Nature 384, 177–179 (1996)
P.T. Chuang, A.P. McMahon, Vertebrate hedgehog signalling modulated by induction of a hedgehog-binding protein. Nature 397(6720), 617–621 (1999). doi:10.1038/17611
L.V. Goodrich, L. Milenkovic, K.M. Higgins, M.P. Scott, Altered neural cell fates and medulloblastoma in mouse patched mutants. Science 277(5329), 1109–1113 (1997)
A. Ruiz i Altaba, C. Mas, B. Stecca, The Gli code: an information nexus regulating cell fate, stemness and cancer. Trends Cell Biol. 17(9), 438–447 (2007). doi:10.1016/j.tcb.2007.06.007
B. Stecca, I.A.A. Ruiz, Context-dependent regulation of the GLI code in cancer by hedgehog and non-hedgehog signals. J. Mol. Cell Biol. 2(2), 84–95 (2010). doi:10.1093/jmcb/mjp052
H. Kimura, D. Stephen, A. Joyner, T. Curran, Gli1 is important for medulloblastoma formation in Ptc1 ± mice. Oncogene 24(25), 4026–4036 (2005). doi:10.1038/sj.onc.1208567
N. Dahmane, J. Lee, P. Robins, P. Heller, A. Ruiz i Altaba, Activation of the transcription factor Gli1 and the Sonic hedgehog signalling pathway in skin tumours. Nature 389(6653), 876–881 (1997). doi:10.1038/39918
K.K. Nelson, P. Gattuso, X. Xu, R.A. Prinz, Expression of the sonic hedgehog pathway molecules in synchronous follicular adenoma and papillary carcinoma of the thyroid gland in predicting malignancy. Surgery 148(4), 654–660 (2010). doi:10.1016/j.surg.2010.07.030. discussion 660
X. Xu, H. Ding, G. Rao, S. Arora, C.P. Saclarides, J. Esparaz, P. Gattuso, C.C. Solorzano, R.A. Prinz, Activation of the Sonic hedgehog pathway in thyroid neoplasms and its potential role in tumor cell proliferation. Endocr. Relat. Cancer 19(2), 167–179 (2012). doi:10.1530/erc-11-0305
J. Taipale, J.K. Chen, M.K. Cooper, B. Wang, R.K. Mann, L. Milenkovic, M.P. Scott, P.A. Beachy, Effects of oncogenic mutations in smoothened and patched can be reversed by cyclopamine. Nature 406(6799), 1005–1009 (2000). doi:10.1038/35023008
D.M. Berman, S.S. Karhadkar, A.R. Hallahan, J.I. Pritchard, C.G. Eberhart, D.N. Watkins, J.K. Chen, M.K. Cooper, J. Taipale, J.M. Olson, P.A. Beachy, Medulloblastoma growth inhibition by hedgehog pathway blockade. Science 297(5586), 1559–1561 (2002). doi:10.1126/science.1073733
S.P. Thayer, M.P. di Magliano, P.W. Heiser, C.M. Nielsen, D.J. Roberts, G.Y. Lauwers, Y.P. Qi, S. Gysin, C. Fernandez-del Castillo, V. Yajnik, B. Antoniu, M. McMahon, A.L. Warshaw, M. Hebrok, Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 425(6960), 851–856 (2003). doi:10.1038/nature02009
M. Lauth, A. Bergstrom, T. Shimokawa, R. Toftgard, Inhibition of GLI-mediated transcription and tumor cell growth by small-molecule antagonists. Proc. Natl. Acad. Sci. USA 104(20), 8455–8460 (2007). doi:10.1073/pnas.0609699104
N.E. Heldin, B. Westermark, The molecular biology of the human anaplastic thyroid carcinoma cell. Thyroidology 3(3), 127–131 (1991)
A. Steg, H.M. Amm, Z. Novak, A.R. Frost, M.R. Johnson, Gli3 mediates cell survival and sensitivity to cyclopamine in pancreatic cancer. Cancer Biol. Ther. 10(9), 893–902 (2010). doi:10.4161/cbt.10.9.13252
S.M. Wiseman, O.L. Griffith, S. Deen, A. Rajput, H. Masoudi, B. Gilks, L. Goldstein, A. Gown, S.J. Jones, Identification of molecular markers altered during transformation of differentiated into anaplastic thyroid carcinoma. Arch. Surg. 142(8), 717–727 (2007). doi:10.1001/archsurg.142.8.717. discussion 727–719
A.R. Jonckheere, A distribution-free k-sample test against ordered alternatives. Biometrica 41, 133–145 (1954)
M.H. Hasler, L.A. Hothorn, Multiple contrast tests in the presence of heteroscedasticity. Biom. J. 50, 793–800 (2008)
N. Ting (ed.), Dose finding in drug developement (Springer, New York, 2006)
R. Bhattacharya, J. Kwon, B. Ali, E. Wang, S. Patra, V. Shridhar, P. Mukherjee, Role of hedgehog signalling in ovarian cancer. Clin. Cancer Res. 14(23), 7659–7666 (2008). doi:10.1158/1078-0432.ccr-08-1414
V. Dormoy, S. Danilin, V. Lindner, L. Thomas, S. Rothhut, C. Coquard, J.J. Helwig, D. Jacqmin, H. Lang, T. Massfelder, The sonic hedgehog signalling pathway is reactivated in human renal cell carcinoma and plays orchestral role in tumor growth. Mol. Cancer 8, 123 (2009). doi:10.1186/1476-4598-8-123
G.A. Raz, K.E. Allen, C. Kingsley, I. Cherni, S. Arora, A. Watanabe, C.D. Lorenzo, V.D.K. Edwards, S. Sridhar, G. Hostetter, G.J. Weiss, Hedgehog signalling pathway molecules and ALDH1A1 expression in early-stage non-small cell lung cancer. Lung Cancer 76(2), 191–196 (2011)
V. Fendrich, J. Waldmann, F. Esni, A. Ramaswamy, M. Mullendore, M. Buchholz, A. Maitra, G. Feldmann, Snail and Sonic hedgehog activation in neuroendocrine tumors of the ileum. Endocr. Relat. Cancer 14(3), 865–874 (2007). doi:10.1677/erc-07-0108
D.M. Berman, S.S. Karhadkar, A. Maitra, R. Montes De Oca, M.R. Gerstenblith, K. Briggs, A.R. Parker, Y. Shimada, J.R. Eshleman, D.N. Watkins, P.A. Beachy, Widespread requirement for Hedgehog ligand stimulation in growth of digestive tract tumours. Nature 425(6960), 846–851 (2003). doi:10.1038/nature01972
D.N. Watkins, D.M. Berman, S.G. Burkholder, B. Wang, P.A. Beachy, S.B. Baylin, Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature 422(6929), 313–317 (2003). doi:10.1038/nature01493
L. Mao, Y.P. Xia, Y.N. Zhou, R.L. Dai, X. Yang, S.J. Duan, X. Qiao, Y.W. Mei, B. Hu, H. Cui, A critical role of Sonic Hedgehog signalling in maintaining the tumorigenicity of neuroblastoma cells. Cancer Sci. 100(10), 1848–1855 (2009). doi:10.1111/j.1349-7006.2009.01262.x
T. Kawahara, N. Kawaguchi-Ihara, Y. Okuhashi, M. Itoh, N. Nara, S. Tohda, Cyclopamine and quercetin suppress the growth of leukemia and lymphoma cells. Anticancer Res. 29(11), 4629–4632 (2009)
P.M.R. LoRusso, C.M. Rudin, J.R. Reddy et al., Phase I trial of hedgehog pathway inhibitor vismodegib (GDC-0449) in patients with refractory, locally advanced or metastatic solid tumors. Clin. Cancer Res. 17(8), 2502–2511 (2011)
E.A. Barnes, K.J. Heidtman, D.J. Donoghue, Constitutive activation of the shh-ptc1 pathway by a patched1 mutation identified in BCC. Oncogene 24(5), 902–915 (2005). doi:10.1038/sj.onc.1208240
J. Reifenberger, M. Wolter, C.B. Knobbe, B. Kohler, A. Schonicke, C. Scharwachter, K. Kumar, B. Blaschke, T. Ruzicka, G. Reifenberger, Somatic mutations in the PTCH, SMOH, SUFUH and TP53 genes in sporadic basal cell carcinomas. Br. J. Dermatol. 152(1), 43–51 (2005). doi:10.1111/j.1365-2133.2005.06353.x
O. Munoz, F. Fend, R. de Beaumont, H. Husson, A. Astier, A.S. Freedman, TGFbeta-mediated activation of Smad1 in B-cell non-Hodgkin’s lymphoma and effect on cell proliferation. Leukemia 18(12), 2015–2025 (2004). doi:10.1038/sj.leu.2403485
E.G. Rizzatti, R.P. Falcao, R.A. Panepucci, R. Proto-Siqueira, W.T. Anselmo-Lima, O.K. Okamoto, M.A. Zago, Gene expression profiling of mantle cell lymphoma cells reveals aberrant expression of genes from the PI3 K-AKT, WNT and TGFbeta signalling pathways. Br. J. Haematol. 130(4), 516–526 (2005). doi:10.1111/j.1365-2141.2005.05630.x
S. Dennler, J. Andre, I. Alexaki, A. Li, T. Magnaldo, P. ten Dijke, X.J. Wang, F. Verrecchia, A. Mauviel, Induction of sonic hedgehog mediators by transforming growth factor-beta: Smad3-dependent activation of Gli2 and Gli1 expression in vitro and in vivo. Cancer Res. 67(14), 6981–6986 (2007). doi:10.1158/0008-5472.can-07-0491
R. Elisei, Anaplastic thyroid cancer therapy: dream or reality? Endocrine 42(3), 468–470 (2012)
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hinterseher, U., Wunderlich, A., Roth, S. et al. Expression of hedgehog signalling pathway in anaplastic thyroid cancer. Endocrine 45, 439–447 (2014). https://doi.org/10.1007/s12020-013-0015-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-013-0015-y