Abstract
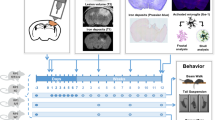
Mild traumatic brain injury (mTBI) can result in permanent impairment in memory and learning and may be a precursor to other neurological sequelae. Clinical treatments to ameliorate the effects of mTBI are lacking. Inhibition of microRNA-181a (miR-181a) is protective in several models of cerebral injury, but its role in mTBI has not been investigated. In the present study, miR-181a-5p antagomir was injected intracerebroventricularly 24 h prior to closed-skull cortical impact in young adult male mice. Paw withdrawal, open field, zero maze, Y maze, object location and novel object recognition tests were performed to assess neurocognitive dysfunction. Brains were assessed immunohistologically for the neuronal marker NeuN, the perineuronal net marker wisteria floribunda lectin (WFA), cFos, and the interneuron marker parvalbumin. Protein quantification was performed with immunoblots for synaptophysin and postsynaptic density 95 (PSD95). Fluorescent in situ hybridization was utilized to localize hippocampal miR-181a expression. MiR-181a antagomir treatment reduced neuronal miR-181a expression after mTBI, restored deficits in novel object recognition and increased hippocampal parvalbumin expression in the dentate gyrus. These changes were associated with decreased dentate gyrus hyperactivity indicated by a relative reduction in PSD95 and cFos expression. These results suggest that miR-181a inhibition may be a therapeutic approach to reduce hippocampal excitotoxicity and prevent cognitive dysfunction following mTBI.





Similar content being viewed by others
References
Amaral, D. G., Scharfman, H. E., & Lavenex, P. (2007). The dentate gyrus: Fundamental neuroanatomical organization (dentate gyrus for dummies). Progress in brain research, 163, 3–22. https://doi.org/10.1016/S0079-6123(07)63001-5.
Amaral, D. G., & Witter, M. P. (1989). The three-dimensional organization of the hippocampal formation: A review of anatomical data. Neuroscience, 31(3), 571–591. https://doi.org/10.1016/0306-4522(89)90424-7.
Aungst, S. L., Kabadi, S. V., Thompson, S. M., Stoica, B. A., & Faden, A. I. (2014). Repeated mild traumatic brain injury causes chronic neuroinflammation, changes in hippocampal synaptic plasticity, and associated cognitive deficits. Journal of Cerebral Blood Flow & Metabolism, 34(7), 1223–1232. https://doi.org/10.1038/jcbfm.2014.75.
Bhushan, R., Grünhagen, J., Becker, J., Robinson, P. N., Ott, C.-E., & Knaus, P. (2013). miR-181a promotes osteoblastic differentiation through repression of TGF-β signaling molecules. The International Journal of Biochemistry & Cell Biology, 45(3), 696–705. https://doi.org/10.1016/j.biocel.2012.12.008.
Boone, D. K., Weisz, H. A., Bi, M., Falduto, M. T., Torres, K. E. O., Willey, H. E., et al. (2017). Evidence linking microRNA suppression of essential prosurvival genes with hippocampal cell death after traumatic brain injury. Scientific Reports, 7(1), 6645. https://doi.org/10.1038/s41598-017-06341-6.
Broadbent, N. J., Gaskin, S., Squire, L. R., & Clark, R. E. (2010). Object recognition memory and the rodent hippocampus. Learning & Memory, 17(1), 5–11. https://doi.org/10.1101/lm.1650110.
Cabungcal, J.-H., Steullet, P., Morishita, H., Kraftsik, R., Cuenod, M., Hensch, T. K., & Do, K. Q. (2013). Perineuronal nets protect fast-spiking interneurons against oxidative stress. Proceedings of the National Academy of Sciences, 110(22), 9130–9135. https://doi.org/10.1073/pnas.1300454110.
Chandrasekar, V., & Dreyer, J.-L. (2009). microRNAs miR-124, let-7d and miR-181a regulate Cocaine-induced Plasticity. Molecular and Cellular Neuroscience, 42(4), 350–362. https://doi.org/10.1016/j.mcn.2009.08.009.
Chandrasekar, V., & Dreyer, J.-L. (2011). Regulation of MiR-124, Let-7d, and MiR-181a in the accumbens affects the expression, extinction, and reinstatement of cocaine-induced conditioned place preference. Neuropsychopharmacology, 36(6), 1149–1164. https://doi.org/10.1038/npp.2010.250.
Chaplan, S. R., Bach, F. W., Pogrel, J. W., Chung, J. M., & Yaksh, T. L. (1994). Quantitative assessment of tactile allodynia in the rat paw. Journal of Neuroscience Methods, 53(1), 55–63. https://doi.org/10.1016/0165-0270(94)90144-9.
Czigner, A., Mihály, A., Farkas, O., Büki, A., Krisztin-Péva, B., Dobó, E., & Barzó, P. (2004). Dynamics and regional distribution of c-fos protein expression in rat brain after a closed head injury. International Journal of Molecular Medicine, 14(2), 247–252.
Datta Chaudhuri, A., Yelamanchili, S. V., & Fox, H. S. (2013). Combined fluorescent in situ hybridization for detection of microRNAs and immunofluorescent labeling for cell-type markers. Frontiers in Cellular Neuroscience. https://doi.org/10.3389/fncel.2013.00160.
de Planell-Saguer, M., Rodicio, M. C., & Mourelatos, Z. (2010). Rapid in situ codetection of noncoding RNAs and proteins in cells and formalin-fixed paraffin-embedded tissue sections without protease treatment. Nature Protocols, 5(6), 1061–1073. https://doi.org/10.1038/nprot.2010.62.
Faden, A. I., O’Leary, D. M., Fan, L., Bao, W., Mullins, P. G. M., & Movsesyan, V. A. (2001). Selective blockade of the mGluR1 receptor reduces traumatic neuronal injury in vitro and improves outcome after brain trauma. Experimental Neurology, 167(2), 435–444. https://doi.org/10.1006/exnr.2000.7577.
Fasulo, L., Brandi, R., Arisi, I., La Regina, F., Berretta, N., Capsoni, S., et al. (2017). ProNGF drives localized and cell selective parvalbumin interneuron and perineuronal net depletion in the dentate gyrus of transgenic mice. Frontiers in Molecular Neuroscience, 10, 20. https://doi.org/10.3389/fnmol.2017.00020.
Gardner, R. C., & Yaffe, K. (2015). Epidemiology of mild traumatic brain injury and neurodegenerative disease. Molecular and Cellular Neuroscience, 66, 75–80. https://doi.org/10.1016/j.mcn.2015.03.001.
Hayes, R. L., Lyeth, B. G., Jenkins, L. W., Zimmerman, R., McIntosh, T. K., Clifton, G. L., & Young, H. F. (2009). Possible protective effect of endogenous opioids in traumatic brain injury. Collections, 116(6), 252–261. https://doi.org/10.3171/jns.1990.72.2.0252@col.2012.116.issue-6.
Hegarty, S. V., Sullivan, A. M., & O’Keeffe, G. W. (2018). Inhibition of miR-181a promotes midbrain neuronal growth through a Smad1/5-dependent mechanism: Implications for Parkinson’s disease. Neuronal Signaling, 2(1), NS20170181. https://doi.org/10.1042/NS20170181.
Hicks, R. r., Smith, D. h., Lowenstein, D. h., Marie, R. S., & McINTOSH, T. k (1993). Mild experimental brain injury in the rat induces cognitive deficits associated with regional neuronal loss in the hippocampus. Journal of Neurotrauma, 10(4), 405–414. https://doi.org/10.1089/neu.1993.10.405.
Hsieh, T.-H., Lee, H. H. C., Hameed, M. Q., Pascual-Leone, A., Hensch, T. K., & Rotenberg, A. (2017). Trajectory of parvalbumin Cell impairment and loss of cortical inhibition in traumatic brain injury. Cerebral Cortex, 27(12), 5509–5524. https://doi.org/10.1093/cercor/bhw318.
Huang, Y., Liu, X., Liao, Y., Luo, C., Zou, D., Wei, X., et al. (2015). MiR-181a influences the cognitive function of epileptic rats induced by pentylenetetrazol. International Journal of Clinical and Experimental Pathology, 8(10), 12861–12868.
Hughes, R. N. (2004). The value of spontaneous alternation behavior (SAB) as a test of retention in pharmacological investigations of memory. Neuroscience & Biobehavioral Reviews, 28(5), 497–505. https://doi.org/10.1016/j.neubiorev.2004.06.006.
Hunsaker, M. R., Mooy, G. G., Swift, J. S., & Kesner, R. P. (2007). Dissociations of the medial and lateral perforant path projections into dorsal DG, CA3, and CA1 for spatial and nonspatial (visual object) information processing. Behavioral Neuroscience, 121(4), 742–750. https://doi.org/10.1037/0735-7044.121.4.742.
Hunsaker, M. R., Rosenberg, J. S., & Kesner, R. P. (2008). The role of the dentate gyrus, CA3a,b, and CA3c for detecting spatial and environmental novelty. Hippocampus, 18(10), 1064–1073. https://doi.org/10.1002/hipo.20464.
Hylin, M. J., Orsi, S. A., Moore, A. N., & Dash, P. K. (2013). Disruption of the perineuronal net in the hippocampus or medial prefrontal cortex impairs fear conditioning. Learning & Memory, 20(5), 267–273. https://doi.org/10.1101/lm.030197.112.
Jackson, J., Chugh, D., Nilsson, P., Wood, J., Carlström, K., Lindvall, O., & Ekdahl, C. T. (2012). Altered synaptic properties during integration of adult-born hippocampal neurons following a seizure insult. PLOS ONE, 7(4), e35557. https://doi.org/10.1371/journal.pone.0035557.
Janis, L. S., Hoane, M. R., Conde, D., Fulop, Z., & Stein, D. G. (1998). Acute ethanol administration reduces the cognitive deficits associated with traumatic brain injury in rats. Journal of Neurotrauma, 15(2), 105–115. https://doi.org/10.1089/neu.1998.15.105.
Jessberger, S., Clark, R. E., Broadbent, N. J., Clemenson, G. D., Consiglio, A., Lie, D. C., et al. (2009). Dentate gyrus-specific knockdown of adult neurogenesis impairs spatial and object recognition memory in adult rats. Learning & Memory, 16(2), 147–154. https://doi.org/10.1101/lm.1172609.
Johansen, F. F., Tønder, N., Zimmer, J., Baimbridge, K. G., & Diemer, N. H. (1990). Short-term changes of parvalbumin and calbindin immunoreactivity in the rat hippocampus following cerebral ischemia. Neuroscience Letters, 120(2), 171–174. https://doi.org/10.1016/0304-3940(90)90030-D.
Kazenwadel, J., Michael, M. Z., & Harvey, N. L. (2010). Prox1 expression is negatively regulated by miR-181 in endothelial cells. Blood, 116(13), 2395–2401. https://doi.org/10.1182/blood-2009-12-256297.
Keith, D. J., & El-Husseini, A. (2008). Excitation control: Balancing PSD-95 function at the synapse. Frontiers in Molecular Neuroscience. https://doi.org/10.3389/neuro.02.004.2008.
Li, X.-G., Somogyi, P., Ylinen, A., & Buzsáki, G. (1994). The hippocampal CA3 network: An in vivo intracellular labeling study. Journal of Comparative Neurology, 339(2), 181–208. https://doi.org/10.1002/cne.903390204.
Lowenstein, D. H., Thomas, M. J., Smith, D. H., & McIntosh, T. K. (1992). Selective vulnerability of dentate hilar neurons following traumatic brain injury: A potential mechanistic link between head trauma and disorders of the hippocampus. Journal of Neuroscience, 12(12), 4846–4853.
Luo, J., Nguyen, A., Villeda, S., Zhang, H., Ding, Z., Lindsey, D., et al. (2014). Long-term cognitive impairments and pathological alterations in a mouse model of repetitive mild traumatic brain injury. Frontiers in Neurology. https://doi.org/10.3389/fneur.2014.00012.
Mtchedlishvili, Z., Lepsveridze, E., Xu, H., Kharlamov, E. A., Lu, B., & Kelly, K. M. (2010). Increase of GABAA receptor-mediated tonic inhibition in dentate granule cells after traumatic brain injury. Neurobiology of Disease, 38(3), 464–475. https://doi.org/10.1016/j.nbd.2010.03.012.
Ouyang, Y.-B., Lu, Y., Yue, S., & Giffard, R. G. (2012). miR-181 targets multiple Bcl-2 family members and influences apoptosis and mitochondrial function in astrocytes. Mitochondrion, 12(2), 213–219. https://doi.org/10.1016/j.mito.2011.09.001.
Palmer, A. M., Marion, D. W., Botscheller, M. L., Swedlow, P. E., Styren, S. D., & DeKosky, S. T. (1993). Traumatic brain injury-induced excitotoxicity assessed in a controlled cortical impact model. Journal of Neurochemistry, 61(6), 2015–2024. https://doi.org/10.1111/j.1471-4159.1993.tb07437.x.
Pandey, D. K., Yadav, S. K., Mahesh, R., & Rajkumar, R. (2009). Depression-like and anxiety-like behavioural aftermaths of impact accelerated traumatic brain injury in rats: A model of comorbid depression and anxiety? Behavioural Brain Research, 205(2), 436–442. https://doi.org/10.1016/j.bbr.2009.07.027.
Phillips, L. l., & Belardo, E. t. (1992). Expression of c-fos in the hippocampus following mild and moderate fluid percussion brain injury. Journal of Neurotrauma, 9(4), 323–333. https://doi.org/10.1089/neu.1992.9.323.
Raghupathi, R., & McIntosh, T. K. (1996). Regionally and temporally distinct patterns of induction of c-fos, c-jun and junB mRNAs following experimental brain injury in the rat. Molecular Brain Research, 37(1), 134–144. https://doi.org/10.1016/0169-328X(95)00289-5.
Redell, J. B., Liu, Y., & Dash, P. K. (2009). Traumatic brain injury alters expression of hippocampal microRNAs: Potential regulators of multiple pathophysiological processes. Journal of Neuroscience Research, 87(6), 1435–1448. https://doi.org/10.1002/jnr.21945.
Reeves, T. M., Lyeth, B. G., Phillips, L. L., Hamm, R. J., & Povlishock, J. T. (1997). The effects of traumatic brain injury on inhibition in the hippocampus and dentate gyrus. Brain Research, 757(1), 119–132. https://doi.org/10.1016/S0006-8993(97)00170-4.
Ren, L., Zhu, R., & Li, X. (2016). Silencing miR-181a produces neuroprotection against hippocampus neuron cell apoptosis post-status epilepticus in a rat model and in children with temporal lobe epilepsy. Genetics and molecular research: GMR. https://doi.org/10.4238/gmr.15017798.
Rippo, M. R., Olivieri, F., Monsurrò, V., Prattichizzo, F., Albertini, M. C., & Procopio, A. D. (2014). MitomiRs in human inflamm-aging: A hypothesis involving miR-181a, miR-34a and miR-146a. Experimental Gerontology, 56, 154–163. https://doi.org/10.1016/j.exger.2014.03.002.
Sahbaie, P., Sun, Y., Liang, D.-Y., Shi, X.-Y., & Clark, J. D. (2014). Curcumin treatment attenuates pain and enhances functional recovery after incision. Anesthesia & Analgesia, 118(6), 1336–1344. https://doi.org/10.1213/ANE.0000000000000189.
Sahbaie, P., Tajerian, M., Yang, P., Irvine, K. A., Huang, T.-T., Luo, J., et al. (2018). Nociceptive and cognitive changes in a murine model of polytrauma. The Journal of Pain. https://doi.org/10.1016/j.jpain.2018.06.004.
Stary, C. M., Xu, L., Li, L., Sun, X., Ouyang, Y.-B., Xiong, X., et al. (2017). Inhibition of miR-181a protects female mice from transient focal cerebral ischemia by targeting astrocyte estrogen receptor-α. Molecular and Cellular Neurosciences, 82, 118–125.
Tajerian, M., Leu, D., Zou, Y., Sahbaie, P., Li, W., Khan, H., et al. (2014). Brain neuroplastic changes accompany anxiety and memory deficits in a model of complex regional pain syndrome. Anesthesiology: The Journal of the American Society of Anesthesiologists, 121(4), 852–865. https://doi.org/10.1097/ALN.0000000000000403.
Tajerian, M., Sahbaie, P., Sun, Y., Leu, D., Yang, H. Y., Li, W., et al. (2015). Sex differences in a murine model of complex regional pain syndrome. Neurobiology of Learning and Memory, 123, 100–109. https://doi.org/10.1016/j.nlm.2015.06.004.
Tang, Y.-P., Noda, Y., Hasegawa, T., & Nabeshima, T. (1997). A Concussive-like brain injury model in mice (II): Selective neuronal loss in the cortex and hippocampus. Journal of Neurotrauma, 14(11), 863–873. https://doi.org/10.1089/neu.1997.14.863.
Taylor, A. N., Tio, D. L., & Sutton, R. L. (2013). Restoration of Neuroendocrine stress response by glucocorticoid receptor or GABAA receptor antagonists after experimental traumatic brain injury. Journal of Neurotrauma, 30(14), 1250–1256. https://doi.org/10.1089/neu.2012.2847.
Thompson, E. H., Lensjø, K. K., Wigestrand, M. B., Malthe-Sørenssen, A., Hafting, T., & Fyhn, M. (2018). Removal of perineuronal nets disrupts recall of a remote fear memory. Proceedings of the National Academy of Sciences, 115(3), 607–612. https://doi.org/10.1073/pnas.1713530115.
Türeci, E., Dashti, R., Tanriverdi, T., Sanus, G. Z., Öz, B., & Uzan, M. (2004). Acute ethanol intoxication in a model of traumatic brain injury: The protective role of moderate doses demonstrated by immunoreactivity of synaptophysin in hippocampal neurons. Neurological Research, 26(1), 108–112. https://doi.org/10.1179/016164104773026633.
Wang, T., Chou, D. Y.-T., Ding, J. Y., Fredrickson, V., Peng, C., Schafer, S., et al. (2012). Reduction of brain edema and expression of aquaporins with acute ethanol treatment after traumatic brain injury. Journal of Neurosurgery, 118(2), 390–396. https://doi.org/10.3171/2012.8.JNS12736.
Xiong, X., Barreto, G. E., Xu, L., Ouyang, Y. B., Xie, X., & Giffard, R. G. (2011). Increased brain injury and worsened neurological outcome in interleukin-4 knockout mice after transient focal cerebral ischemia. Stroke, 42(7), 2026–2032. https://doi.org/10.1161/strokeaha.110.593772.
Xu, L.-J., Ouyang, Y.-B., Xiong, X., Stary, C. M., & Giffard, R. G. (2015). Post-stroke treatment with miR-181 antagomir reduces injury and improves long-term behavioral recovery in mice after focal cerebral ischemia. Exp Neurol, 264, 1–7.
Xu, X.-F., Jing, X., Ma, H.-X., Yuan, R.-R., Dong, Q., Dong, J.-L., et al. (2018). miR-181a participates in contextual fear memory formation Via activating mTOR signaling pathway. Cerebral Cortex, 28(9), 3309–3321. https://doi.org/10.1093/cercor/bhx201.
Yang, K., Mu, X. S., Xue, J. J., Whitson, J., Salminen, A., Dixon, C. E., et al. (1994). Increased expression of c-fos mRNA and AP-1 transcription factors after cortical impact injury in rats. Brain Research, 664(1), 141–147. https://doi.org/10.1016/0006-8993(94)91964-X.
Zhang, S., Chen, J., Zhang, J., & Xu, J. (2017). miR-181a involves in the hippocampus-dependent memory formation via targeting PRKAA1. Scientific Reports, 7(1), 8480. https://doi.org/10.1038/s41598-017-09095-3.
Zhang, X.-D., Wang, H., Antaris, A. L., Li, L., Diao, S., Ma, R., et al. (2016). Traumatic brain injury imaging in the second near-infrared window with a molecular fluorophore. Advanced Materials, 28(32), 6872–6879. https://doi.org/10.1002/adma.201600706.
Zhang, Z. J., & Reynolds, G. P. (2002). A selective decrease in the relative density of parvalbumin-immunoreactive neurons in the hippocampus in schizophrenia. Schizophrenia Research, 55(1), 1–10. https://doi.org/10.1016/S0920-9964(01)00188-8.
Zhao, J., Gong, A.-Y., Zhou, R., Liu, J., Eischeid, A. N., & Chen, X.-M. (2012). Downregulation of PCAF by miR-181a/b provides feedback regulation to TNF-α–induced transcription of proinflammatory genes in liver epithelial cells. The Journal of Immunology, 188(3), 1266–1274. https://doi.org/10.4049/jimmunol.1101976.
Zou, Y., Corniola, R., Leu, D., Khan, A., Sahbaie, P., Chakraborti, A., et al. (2012). Extracellular superoxide dismutase is important for hippocampal neurogenesis and preservation of cognitive functions after irradiation. Proceedings of the National Academy of Sciences, 109(52), 21522–21527. https://doi.org/10.1073/pnas.1216913110.
Funding
This study was funded in part by American Heart Association Grants FTF-19970029 to CMS and #18POST33990395 to BBG, the Finnish Cultural Foundation grant #00171200 to OA, NIH #5R01NS053898 to RG, and VA Merit Review Grant #RX001776-01 to DJC.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Griffiths, B.B., Sahbaie, P., Rao, A. et al. Pre-treatment with microRNA-181a Antagomir Prevents Loss of Parvalbumin Expression and Preserves Novel Object Recognition Following Mild Traumatic Brain Injury. Neuromol Med 21, 170–181 (2019). https://doi.org/10.1007/s12017-019-08532-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-019-08532-y