Abstract
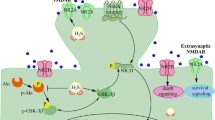
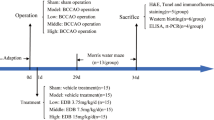
Chronic cerebral hypoperfusion (CCH) is associated with various neurodegenerative diseases characterized by cognitive impairment. Dozens of studies including ours have indicated that exogenous administration of vascular endothelial growth factor (VEGF) could exert effective cognitive protection during ischemia. Nevertheless, the underlying mechanism has not been well clarified. To address this issue, we explored the synaptic mechanisms in vivo since hippocampal synaptic function is essential to the learning and memory process. Besides, the role of autophagy in cognitive dysfunction under conditions of CCH is still controversial. And abnormal autophagy could threaten normal neurotransmission at synapse where a large amount of protein synthesis and degradation take place. Hence, we further examined whether the altered synaptic function was associated with autophagy. The results showed that CCH impaired spatial cognition as evidenced in Morris water maze. We further found that VEGF mitigated impaired hippocampal synaptic function including basal synaptic transmission, paired-pulse facilitation, short-term, long-term plasticity, depotentiation, and the level of synaptic proteins as assessed by electrophysiological examination and western blot assay. Furthermore, our results demonstrated that CCH could induce excessive autophagy which could be inhibited by VEGF. Thus, we speculated that VEGF could ameliorate impaired synaptic function induced by CCH because of its ability to inhibit excessive autophagy, and eventually improve spatial learning and memory function. Importantly, our findings shed light on potential therapeutic strategies to be exploited in the usage of VEGF.







Similar content being viewed by others
References
An, L., Yang, Z., & Zhang, T. (2013). Imbalanced synaptic plasticity induced spatial cognition impairment in male offspring rats treated with chronic prenatal ethanol exposure. Alcoholism, Clinical and Experimental Research, 37(5), 763–770. doi:10.1111/acer.12040.
Balduini, W., De Angelis, V., Mazzoni, E., & Cimino, M. (2001). Simvastatin protects against long-lasting behavioral and morphological consequences of neonatal hypoxic/ischemic brain injury. Stroke, 32(9), 2185–2191.
Carloni, S., Buonocore, G., & Balduini, W. (2008). Protective role of autophagy in neonatal hypoxia-ischemia induced brain injury. Neurobiology of Disease, 32(3), 329–339. doi:10.1016/j.nbd.2008.07.022.
Clarke, P. G. (1990). Developmental cell death: Morphological diversity and multiple mechanisms. Anatomy and Embryology, 181(3), 195–213.
Codogno, P., & Meijer, A. J. (2005). Autophagy and signaling: Their role in cell survival and cell death. Cell Death and Differentiation, 12(Suppl 2), 1509–1518. doi:10.1038/sj.cdd.4401751.
Erickson, M. A., Maramara, L. A., & Lisman, J. (2010). A single brief burst induces GluR1-dependent associative short-term potentiation: a potential mechanism for short-term memory. Journal of Cognitive Neuroscience, 22(11), 2530–2540. doi:10.1162/jocn.2009.21375.
Erickson, B. A., Meeks, J. J., Roehl, K. A., Gonzalez, C. M., & Catalona, W. J. (2009). Bladder neck contracture after retropubic radical prostatectomy: Incidence and risk factors from a large single-surgeon experience. BJU International, 104(11), 1615–1619. doi:10.1111/j.1464-410X.2009.08700.x.
Eshkoor, S. A., Hamid, T. A., Mun, C. Y., & Ng, C. K. (2015). Mild cognitive impairment and its management in older people. Clinical Interventions in Aging, 10, 687–693. doi:10.2147/CIA.S73922.
Ginet, V., Spiehlmann, A., Rummel, C., Rudinskiy, N., Grishchuk, Y., Luthi-Carter, R., et al. (2014). Involvement of autophagy in hypoxic-excitotoxic neuronal death. Autophagy, 10(5), 846–860. doi:10.4161/auto.28264.
Gruneberg, D., Montellano, F. A., Plaschke, K., Li, L., Marti, H. H., & Kunze, R. (2016). Neuronal prolyl-4-hydroxylase 2 deficiency improves cognitive abilities in a murine model of cerebral hypoperfusion. Experimental Neurology, 286, 93–106. doi:10.1016/j.expneurol.2016.10.001.
Heiss, W. D., Kracht, L. W., Thiel, A., Grond, M., & Pawlik, G. (2001). Penumbral probability thresholds of cortical flumazenil binding and blood flow predicting tissue outcome in patients with cerebral ischaemia. Brain, 124(Pt 1), 20–29.
Jia, Y., Jin, W., Xiao, Y., Dong, Y., Wang, T., Fan, M., et al. (2015). Lipoxin A4 methyl ester alleviates vascular cognition impairment by regulating the expression of proteins related to autophagy and ER stress in the rat hippocampus. Cellular & Molecular Biology Letters, 20(3), 475–487. doi:10.1515/cmble-2015-0027.
Koike, M., Shibata, M., Tadakoshi, M., Gotoh, K., Komatsu, M., Waguri, S., et al. (2008). Inhibition of autophagy prevents hippocampal pyramidal neuron death after hypoxic-ischemic injury. The American Journal of Pathology, 172(2), 454–469. doi:10.2353/ajpath.2008.070876.
Komatsu, M., Waguri, S., Chiba, T., Murata, S., Iwata, J., Tanida, I., et al. (2006). Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature, 441(7095), 880–884. doi:10.1038/nature04723.
Lee, K. M., Hwang, S. K., & Lee, J. A. (2013). Neuronal autophagy and neurodevelopmental disorders. Experimental Neurobiology, 22(3), 133–142. doi:10.5607/en.2013.22.3.133.
Li, H., Wang, J., Wang, P., Rao, Y., & Chen, L. (2016). Resveratrol reverses the synaptic plasticity deficits in a chronic cerebral hypoperfusion rat model. Journal of Stroke and Cerebrovascular Diseases, 25(1), 122–128. doi:10.1016/j.jstrokecerebrovasdis.2015.09.004.
Liang, X. H., Yu, J., Brown, K., & Levine, B. (2001). Beclin 1 contains a leucine-rich nuclear export signal that is required for its autophagy and tumor suppressor function. Cancer Research, 61(8), 3443–3449.
Liu, Z., Hu, M., Lu, P., Wang, H., Qi, Q., Xu, J., et al. (2017). Cerebrolysin alleviates cognitive deficits induced by chronic cerebral hypoperfusion by increasing the levels of plasticity-related proteins and decreasing the levels of apoptosis-related proteins in the rat hippocampus. Neuroscience Letters, 651, 72–78. doi:10.1016/j.neulet.2017.04.022.
Liu, C., Xu, X., Gao, J., Zhang, T., & Yang, Z. (2016). Hydrogen sulfide prevents synaptic plasticity from VD-induced damage via Akt/GSK-3beta pathway and notch signaling pathway in rats. Molecular Neurobiology, 53(6), 4159–4172. doi:10.1007/s12035-015-9324-x.
Luo, T., Liu, G., Ma, H., Lu, B., Xu, H., Wang, Y., et al. (2014). Inhibition of autophagy via activation of PI3 K/Akt pathway contributes to the protection of ginsenoside Rb1 against neuronal death caused by ischemic insults. International Journal of Molecular Sciences, 15(9), 15426–15442. doi:10.3390/ijms150915426.
Maiuri, M. C., Zalckvar, E., Kimchi, A., & Kroemer, G. (2007). Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nature Reviews Molecular Cell Biology, 8(9), 741–752. doi:10.1038/nrm2239.
Miyamoto, E. (2006). Molecular mechanism of neuronal plasticity: induction and maintenance of long-term potentiation in the hippocampus. Journal of Pharmacological Sciences, 100(5), 433–442.
Mizushima, N., Levine, B., Cuervo, A. M., & Klionsky, D. J. (2008). Autophagy fights disease through cellular self-digestion. Nature, 451(7182), 1069–1075. doi:10.1038/nature06639.
Mufson, E. J., Binder, L., Counts, S. E., DeKosky, S. T., de Toledo-Morrell, L., Ginsberg, S. D., et al. (2012). Mild cognitive impairment: Pathology and mechanisms. Acta Neuropathologica, 123(1), 13–30. doi:10.1007/s00401-011-0884-1.
Park, J. M., Seong, H. H., Jin, H. B., & Kim, Y. J. (2017). The effect of long-term environmental enrichment in chronic cerebral hypoperfusion-induced memory impairment in rats. Biological Research for Nursing, 19(3), 278–286. doi:10.1177/1099800416686179.
Petersen, R. C. (2011). Clinical practice. Mild cognitive impairment. The New England Journal of Medicine, 364(23), 2227–2234. doi:10.1056/NEJMcp0910237.
Qi, Y., Hu, N. W., & Rowan, M. J. (2013). Switching off LTP: mGlu and NMDA receptor-dependent novelty exploration-induced depotentiation in the rat hippocampus. Cerebral Cortex, 23(4), 932–939. doi:10.1093/cercor/bhs086.
Silva, A. J. (2003). Molecular and cellular cognitive studies of the role of synaptic plasticity in memory. Journal of Neurobiology, 54(1), 224–237. doi:10.1002/neu.10169.
Steiger-Barraissoul, S., & Rami, A. (2009). Serum deprivation induced autophagy and predominantly an AIF-dependent apoptosis in hippocampal HT22 neurons. Apoptosis, 14(11), 1274–1288. doi:10.1007/s10495-009-0396-9.
Sun, Y., Jin, K., Xie, L., Childs, J., Mao, X. O., Logvinova, A., et al. (2003). VEGF-induced neuroprotection, neurogenesis, and angiogenesis after focal cerebral ischemia. Journal of Clinical Investigation, 111(12), 1843–1851. doi:10.1172/JCI17977.
Tramutola, A., Triplett, J. C., Di Domenico, F., Niedowicz, D. M., Murphy, M. P., Coccia, R., et al. (2015). Alteration of mTOR signaling occurs early in the progression of Alzheimer disease (AD): Analysis of brain from subjects with pre-clinical AD, amnestic mild cognitive impairment and late-stage AD. Journal of Neurochemistry, 133(5), 739–749. doi:10.1111/jnc.13037.
Wang, Y., Galvan, V., Gorostiza, O., Ataie, M., Jin, K., & Greenberg, D. A. (2006). Vascular endothelial growth factor improves recovery of sensorimotor and cognitive deficits after focal cerebral ischemia in the rat. Brain Research, 1115(1), 186–193. doi:10.1016/j.brainres.2006.07.060.
Wang, P., Guan, Y. F., Du, H., Zhai, Q. W., Su, D. F., & Miao, C. Y. (2012). Induction of autophagy contributes to the neuroprotection of nicotinamide phosphoribosyltransferase in cerebral ischemia. Autophagy, 8(1), 77–87. doi:10.4161/auto.8.1.18274.
Wen, Y. D., Sheng, R., Zhang, L. S., Han, R., Zhang, X., Zhang, X. D., et al. (2008). Neuronal injury in rat model of permanent focal cerebral ischemia is associated with activation of autophagic and lysosomal pathways. Autophagy, 4(6), 762–769.
Xing, S., Zhang, Y., Li, J., Zhang, J., Li, Y., Dang, C., et al. (2012). Beclin 1 knockdown inhibits autophagic activation and prevents the secondary neurodegenerative damage in the ipsilateral thalamus following focal cerebral infarction. Autophagy, 8(1), 63–76. doi:10.4161/auto.8.1.18217.
Yang, J., Yao, Y., Chen, T., & Zhang, T. (2014). VEGF ameliorates cognitive impairment in in vivo and in vitro ischemia via improving neuronal viability and function. NeuroMolecular Medicine, 16(2), 376–388. doi:10.1007/s12017-013-8284-4.
Yang, J., Yao, Y., Wang, L., Yang, C., Wang, F., Guo, J., et al. (2017). Gastrin-releasing peptide facilitates glutamatergic transmission in the hippocampus and effectively prevents vascular dementia induced cognitive and synaptic plasticity deficits. Experimental Neurology, 287(Pt 1), 75–83. doi:10.1016/j.expneurol.2016.08.008.
Zhu, Y., Zhang, Q., Zhang, W., Li, N., Dai, Y., Tu, J., et al. (2017). Protective effect of 17beta-estradiol upon hippocampal spine density and cognitive function in an animal model of vascular dementia. Scientific Reports, 7, 42660. doi:10.1038/srep42660.
Zou, W., Song, Y., Li, Y., Du, Y., Zhang, X., & Fu, J. (2017). The Role of Autophagy in the Correlation Between Neuron Damage and Cognitive Impairment in Rat Chronic Cerebral Hypoperfusion. Molecular Neurobiology. doi:10.1007/s12035-016-0351-z.
Zucker, R. S., & Regehr, W. G. (2002). Short-term synaptic plasticity. Annual Review of Physiology, 64, 355–405. doi:10.1146/annurev.physiol.64.092501.114547.
Acknowledgements
The study was supported by grants from the National Natural Science Foundation of China (31500865, 91520205) and Tianjin Municipal Natural Science Foundation (17JCQNJC10500). We are grateful to Professor Zhuo Yang and Tao Zhang from Nankai University for providing the technical assistance.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Wang, L., Wang, J., Wang, F. et al. VEGF-Mediated Cognitive and Synaptic Improvement in Chronic Cerebral Hypoperfusion Rats Involves Autophagy Process. Neuromol Med 19, 423–435 (2017). https://doi.org/10.1007/s12017-017-8458-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-017-8458-6