Abstract
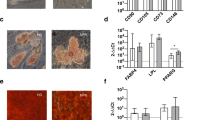
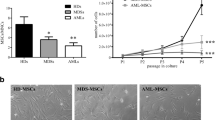
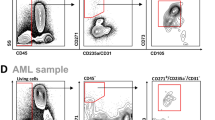
An essential component of the hematopoietic microenvironment, bone marrow mesenchymal stem cells (BM-MSCs) play an important role in the homeostasis and pathogenesis of the hematopoietic system by regulating the fate of hematopoietic stem cells (HSCs). Previous studies revealed that BM-MSCs were functionally remodeled by malignant cells in leukemia. However, the alterations in BM-MSCs in polycythemia vera (PV) and their effects on HSCs still need to be elucidated. Our results demonstrated that although BM-MSCs from PV patients shared similar surface markers with those from healthy donors, they exhibited enhanced proliferation, decreased senescence, and abnormal osteogenic differentiation capacities. The CD146+CD271+ BM-MSC subpopulation, which is considered to give rise to typical cultured BM-MSCs and form bone and the hematopoietic stroma, was then sorted. Compared with those from healthy donors, CD146+CD271+ BM-MSCs from PV patients showed an impaired mesensphere formation capacity and abnormal differentiation toward osteogenic lineages. In addition, CD146+CD271+ PV BM-MSCs showed altered hematopoietic supportive activity when cocultured with cord blood CD34+ cells. Our study suggested that remodeled CD146+CD271+ BM-MSCs might contribute to the pathogenesis of PV, a finding that will shed light on potential therapeutic strategies for PV.
Graphical abstract






Similar content being viewed by others
Data availability
The accession number for the RNA-seq data reported in this paper is NCBI Gene Expression Omnibus (GEO) GSE87806.
References
Martinaud, C., Desterke, C., Konopacki, J., Pieri, L., Torossian, F., Golub, R., Schmutz, S., Anginot, A., Guerton, B., Rochet, N., Albanese, P., Henault, E., Pierre-Louis, O., Souraud, J. B., de Revel, T., Dupriez, B., Ianotto, J. C., Bourgeade, M. F., Vannucchi, A. M., … Le Bousse-Kerdiles, M. C. (2015). Osteogenic Potential of Mesenchymal Stromal Cells Contributes to Primary Myelofibrosis. Cancer Research, 75(22), 4753–4765. https://doi.org/10.1158/0008-5472.CAN-14-3696
Spivak, J. L. (2018). Polycythemia Vera. Current Treatment Options in Oncology, 19(2), 12. https://doi.org/10.1007/s11864-018-0529-x
Le, P. M., Andreeff, M., & Battula, V. L. (2018). Osteogenic niche in the regulation of normal hematopoiesis and leukemogenesis. Haematologica, 103(12), 1945–1955. https://doi.org/10.3324/haematol.2018.197004
Schepers, K., Campbell, T. B., & Passegue, E. (2015). Normal and leukemic stem cell niches: Insights and therapeutic opportunities. Cell Stem Cell, 16(3), 254–267. https://doi.org/10.1016/j.stem.2015.02.014
Chen, Q., Shou, P., Zheng, C., Jiang, M., Cao, G., Yang, Q., Cao, J., Xie, N., Velletri, T., Zhang, X., Xu, C., Zhang, L., Yang, H., Hou, J., Wang, Y., & Shi, Y. (2016). Fate decision of mesenchymal stem cells: Adipocytes or osteoblasts? Cell Death and Differentiation, 23(7), 1128–1139. https://doi.org/10.1038/cdd.2015.168
Zhou, H. S., Carter, B. Z., & Andreeff, M. (2016). Bone marrow niche-mediated survival of leukemia stem cells in acute myeloid leukemia: Yin and Yang. Cancer Biology & Medicine, 13(2), 248–259. https://doi.org/10.20892/j.issn.2095-3941.2016.0023
Schepers, K., Pietras, E. M., Reynaud, D., Flach, J., Binnewies, M., Garg, T., Wagers, A. J., Hsiao, E. C., & Passegue, E. (2013). Myeloproliferative neoplasia remodels the endosteal bone marrow niche into a self-reinforcing leukemic niche. Cell Stem Cell, 13(3), 285–299. https://doi.org/10.1016/j.stem.2013.06.009
Sun, T., Ju, M., Dai, X., Dong, H., Gu, W., Gao, Y., Fu, R., Liu, X., Huang, Y., Liu, W., Ch, Y., Wang, W., Li, H., Zhou, Y., Shi, L., Yang, R., & Zhang, L. (2020). Multilevel defects in the hematopoietic niche in essential thrombocythemia. Haematologica, 105(3), 661–673. https://doi.org/10.3324/haematol.2018.213686
Mead, A. J., & Mullally, A. (2017). Myeloproliferative neoplasm stem cells. Blood, 129(12), 1607–1616. https://doi.org/10.1182/blood-2016-10-696005
Tormin, A., Li, O., Brune, J. C., Walsh, S., Schutz, B., Ehinger, M., Ditzel, N., Kassem, M., & Scheding, S. (2011). CD146 expression on primary nonhematopoietic bone marrow stem cells is correlated with in situ localization. Blood, 117(19), 5067–5077. https://doi.org/10.1182/blood-2010-08-304287
Shi, H., Wang, Y., Li, R., Xing, W., Yang, F. C., Bai, J., & Zhou, Y. (2018). Alteration in the Cytokine Secretion of Bone Marrow Stromal Cells from Patients with Chronic Myelomonocytic Leukemia Contribute to Impaired Hematopoietic Supportive Activity. Stem Cells Int, 2018, 5921392. https://doi.org/10.1155/2018/5921392
Ramos, T. L., Sanchez-Abarca, L. I., Roson-Burgo, B., Redondo, A., Rico, A., Preciado, S., Ortega, R., Rodriguez, C., Muntion, S., Hernandez-Hernandez, A., De Las Rivas, J., Gonzalez, M., Gonzalez Porras, J. R., Del Canizo, C., & Sanchez-Guijo, F. (2017). Mesenchymal stromal cells (MSC) from JAK2+ myeloproliferative neoplasms differ from normal MSC and contribute to the maintenance of neoplastic hematopoiesis. PLoS ONE, 12(8), e0182470. https://doi.org/10.1371/journal.pone.0182470
Li, R., Zhou, Y., Cao, Z., Liu, L., Wang, J., Chen, Z., Xing, W., Chen, S., Bai, J., Yuan, W., Cheng, T., Xu, M., Yang, F. C., & Zhao, Z. (2018). TET2 Loss Dysregulates the Behavior of Bone Marrow Mesenchymal Stromal Cells and Accelerates Tet2(-/-)-Driven Myeloid Malignancy Progression. Stem Cell Reports, 10(1), 166–179. https://doi.org/10.1016/j.stemcr.2017.11.019
Zhang, P., Chen, Z., Li, R., Guo, Y., Shi, H., Bai, J., Yang, H., Sheng, M., Li, Z., Li, Z., Li, J., Chen, S., Yuan, W., Cheng, T., Xu, M., Zhou, Y., & Yang, F. C. (2018). Loss of ASXL1 in the bone marrow niche dysregulates hematopoietic stem and progenitor cell fates. Cell Discov, 4, 4. https://doi.org/10.1038/s41421-017-0004-z
Till, J. E., & Mc, C. E. (1961). A direct measurement of the radiation sensitivity of normal mouse bone marrow cells. Radiat Res, 14, 213–22.
Friedenstein, A. J., Chailakhjan, R. K., & Lalykina, K. S. (1970). The development of fibroblast colonies in monolayer cultures of guinea-pig bone marrow and spleen cells. Cell and Tissue Kinetics, 3(4), 393–403. https://doi.org/10.1111/j.1365-2184.1970.tb00347.x
Mo, M., Wang, S., Zhou, Y., Li, H., & Wu, Y. (2016). Mesenchymal stem cell subpopulations: Phenotype, property and therapeutic potential. Cellular and Molecular Life Sciences, 73(17), 3311–3321. https://doi.org/10.1007/s00018-016-2229-7
Busser, H., Najar, M., Raicevic, G., Pieters, K., Velez Pombo, R., Philippart, P., Meuleman, N., Bron, D., & Lagneaux, L. (2015). Isolation and Characterization of Human Mesenchymal Stromal Cell Subpopulations: Comparison of Bone Marrow and Adipose Tissue. Stem Cells Dev, 24(18), 2142–2157. https://doi.org/10.1089/scd.2015.0172
Flores-Figueroa, E., Varma, S., Montgomery, K., Greenberg, P. L., & Gratzinger, D. (2012). Distinctive contact between CD34+ hematopoietic progenitors and CXCL12+ CD271+ mesenchymal stromal cells in benign and myelodysplastic bone marrow. Laboratory Investigation, 92(9), 1330–1341. https://doi.org/10.1038/labinvest.2012.93
Kuci, S., Kuci, Z., Kreyenberg, H., Deak, E., Putsch, K., Huenecke, S., Amara, C., Koller, S., Rettinger, E., Grez, M., Koehl, U., Latifi-Pupovci, H., Henschler, R., Tonn, T., von Laer, D., Klingebiel, T., & Bader, P. (2010). CD271 antigen defines a subset of multipotent stromal cells with immunosuppressive and lymphohematopoietic engraftment-promoting properties. Haematologica, 95(4), 651–659. https://doi.org/10.3324/haematol.2009.015065
Martin-Rendon, E., Sweeney, D., Lu, F., Girdlestone, J., Navarrete, C., & Watt, S. M. (2008). 5-Azacytidine-treated human mesenchymal stem/progenitor cells derived from umbilical cord, cord blood and bone marrow do not generate cardiomyocytes in vitro at high frequencies. Vox Sanguinis, 95(2), 137–148. https://doi.org/10.1111/j.1423-0410.2008.01076.x
Sorrentino, A., Ferracin, M., Castelli, G., Biffoni, M., Tomaselli, G., Baiocchi, M., Fatica, A., Negrini, M., Peschle, C., & Valtieri, M. (2008). Isolation and characterization of CD146+ multipotent mesenchymal stromal cells. Experimental Hematology, 36(8), 1035–1046. https://doi.org/10.1016/j.exphem.2008.03.004
Funding
This work was supported by National Natural Science Foundation of China (81970120, 81890990), National Key Research and Development Program of China (2020YFE0203000) and CAMS Innovation Fund for Medical Sciences (2017-I2M-3–018).
Author information
Authors and Affiliations
Contributions
Chao Chen, Rong Li and Yuan Zhou designed the study. Chao Chen, Mingying Zhang and Rong Li performed the experiments. Chao Chen and Mingying Zhang performed RNA-Seq analysis. Jiajia Yuan, Jinqiang Yan, Yuhui Zhang, Wen Xing, Jie Bai and Yuan Zhou discussed and analyzed the data. Chao Chen, Mingying Zhang and Yuan Zhou wrote the manuscript. All authors reviewed, edited, and approved the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
This study was approved by the Ethics Committee of Institute of Hematology, and Blood Diseases Hospital, Chinese Academy of Medical Sciences, according to the 1975 Declaration of Helsinki and its later ethical standards. Written informed consent was obtained from all participants included in this study.
Conflicts of interest
The authors declare no conflict of interest, financial or otherwise.
Code Availability
Nothing to declare.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
All authors reviewed and approved the final version of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, C., Zhang, M., Li, R. et al. Remodeled CD146+CD271+ Bone Marrow Mesenchymal Stem Cells from Patients with Polycythemia Vera Exhibit Altered Hematopoietic Supportive Activity. Stem Cell Rev and Rep 19, 406–416 (2023). https://doi.org/10.1007/s12015-022-10427-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-022-10427-8