Abstract
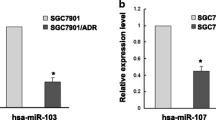
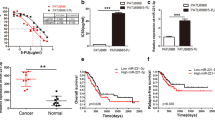
The development of drug resistance has largely limited the clinical outcome of anti-cancer treatment. Recent work has highlighted the involvement of non-coding RNAs, microRNAs (miRNAs), in cancer development. The present study aimed to investigate the role of miR-21 in the development of drug resistance to paclitaxel in gastric cancer cells. Our study found that the expression of miR-21 upregulated in the paclitaxel resistant cell line SGC7901/paclitaxel compared to its parental line SGC7901. Moreover, over-expression of miR-21 significantly decreased antiproliferative effects and apoptosis induced by paclitaxel, while knockdown of miR-21 dramatically increased antiproliferative effects and apoptosis induction by paclitaxel. Moreover, our results demonstrated that miR-21 may modulate the sensitivity to PTX, at least in part, by regulating the expression of P-glycoprotein.




Similar content being viewed by others
References
Meng, F., Henson, R., Lang, M., Wehbe, H., Maheshwari, S., Mendell, J. T., et al. (2006). Involvement of human micro-RNA in growth and response to chemotherapy in human cholangiocarcinoma cell lines. Gastroenterology, 130, 2113–2129.
Jemal, A., Bray, F., Center, M. M., Ferlay, J., Ward, E., & Forman, D. (2011). Global cancer statistics. CA: A Cancer Journal for Clinicians, 61, 69–90.
Xie, Z., Cao, L., & Zhang, J. (2013). miR-21 modulates paclitaxel sensitivity and hypoxia-inducible factor-1alpha expression in human ovarian cancer cells. Oncology Letters, 6, 795–800.
Wu, G., Qin, X. Q., Guo, J. J., Li, T. Y., & Chen, J. H. (2014). AKT/ERK activation is associated with gastric cancer cell resistance to paclitaxel. International Journal of Clinical and Experimental Pathology, 7, 1449–1458.
Huang, H., Han, Y., Gao, J., Feng, J., Zhu, L., Qu, L., et al. (2013). High level of serum AMBP is associated with poor response to paclitaxel-capecitabine chemotherapy in advanced gastric cancer patients. Medical Oncology, 30, 748.
Yuan, M., Yang, Y., Lv, W., Song, Z., & Zhong, H. (2014). Paclitaxel combined with capecitabine as first-line chemotherapy for advanced or recurrent gastric cancer. Oncology Letters, 8, 351–354.
Yoshino, S., Furuya, T., Shimizu, R., Ozasa, H., Nishimura, T., Fukuda, S., et al. (2013). Combination phase II study of weekly paclitaxel and 5′-DFUR for unresectable or recurrent gastric cancer. Anticancer Research, 33, 2629–2633.
Wang, X., Wang, M. L., Zhou, L. Y., Lu, X. Y., Yang, J. F., & Yu, H. G. (2013). Randomized phase II study comparing paclitaxel with S-1 vs. S-1 as first-line treatment in patients with advanced gastric cancer. Clinical and Translational Oncology, 15, 836–842.
Kadokura, M., Iwasa, S., Honma, Y., Kato, K., Hamaguchi, T., Yamada, Y., et al. (2013). Weekly paclitaxel as second-line chemotherapy in Japanese patients with advanced gastric cancer. Anticancer Research, 33, 4547–4552.
Ando, T., Hosokawa, A., Kajiura, S., Itaya, Y., Ueda, A., Fujinami, H., et al. (2012). Efficacy of weekly paclitaxel in patients with advanced gastric cancer refractory to docetaxel-based chemotherapy. Gastric Cancer, 15, 427–432.
Sasaki, Y., Nishina, T., Yasui, H., Goto, M., Muro, K., Tsuji, A., et al. (2014). Phase II trial of nanoparticle albumin-bound paclitaxel as second-line chemotherapy for unresectable or recurrent gastric cancer. Cancer Science, 105, 812–817.
Kong, Y. W., Ferland-McCollough, D., Jackson, T. J., & Bushell, M. (2012). microRNAs in cancer management. The Lancet Oncology, 13, e249–e258.
Flynt, A. S., & Lai, E. C. (2008). Biological principles of microRNA-mediated regulation: Shared themes amid diversity. Nature Reviews Genetics, 9, 831–842.
Carthew, R. W., & Sontheimer, E. J. (2009). Origins and mechanisms of miRNAs and siRNAs. Cell, 136, 642–655.
Ren, Y., Zhou, X., Mei, M., Yuan, X. B., Han, L., Wang, G. X., et al. (2010). MicroRNA-21 inhibitor sensitizes human glioblastoma cells U251 (PTEN-mutant) and LN229 (PTEN-wild type) to taxol. BMC Cancer, 10, 27.
Volinia, S., Calin, G. A., Liu, C. G., Ambs, S., Cimmino, A., Petrocca, F., et al. (2006). A microRNA expression signature of human solid tumors defines cancer gene targets. Proceedings of the National Academy of Sciences of the United States of America, 103, 2257–2261.
Chan, S. H., Wu, C. W., Li, A. F., Chi, C. W., & Lin, W. C. (2008). miR-21 microRNA expression in human gastric carcinomas and its clinical association. Anticancer Research, 28, 907–911.
Zhang, Z., Li, Z., Gao, C., Chen, P., Chen, J., Liu, W., et al. (2008). miR-21 plays a pivotal role in gastric cancer pathogenesis and progression. Laboratory Investigation: A Journal of Technical Methods and Pathology, 88, 1358–1366.
Li, Z., Hu, S., Wang, J., Cai, J., Xiao, L., Yu, L., & Wang, Z. (2010). MiR-27a modulates MDR1/P-glycoprotein expression by targeting HIPK2 in human ovarian cancer cells. Gynecologic Oncology, 119, 125–130.
Bao, L., Yan, Y., Xu, C., Ji, W., Shen, S., Xu, G., et al. (2013). MicroRNA-21 suppresses PTEN and hSulf-1 expression and promotes hepatocellular carcinoma progression through AKT/ERK pathways. Cancer Letters, 337, 226–236.
Chen, C., Ridzon, D. A., Broomer, A. J., Zhou, Z., Lee, D. H., Nguyen, J. T., et al. (2005). Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Research, 33, e179.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta delta C(T)) method. Methods, 25, 402–408.
Huang, S., Chen, M., Shen, Y., Shen, W., Guo, H., Gao, Q., & Zou, X. (2012). Inhibition of activated Stat3 reverses drug resistance to chemotherapeutic agents in gastric cancer cells. Cancer Letters, 315, 198–205.
Goda, K., Bacso, Z., & Szabo, G. (2009). Multidrug resistance through the spectacle of P-glycoprotein. Current Cancer Drug Targets, 9, 281–297.
Roberti, A., La Sala, D., & Cinti, C. (2006). Multiple genetic and epigenetic interacting mechanisms contribute to clonally selection of drug-resistant tumors: Current views and new therapeutic prospective. Journal of Cellular Physiology, 207, 571–581.
Selimovic, D., Hassan, M., Haikel, Y., & Hengge, U. R. (2008). Taxol-induced mitochondrial stress in melanoma cells is mediated by activation of c-Jun N-terminal kinase (JNK) and p38 pathways via uncoupling protein 2. Cellular Signalling, 20, 311–322.
Yu, J., Gao, J., Lu, Z., Li, Y., & Shen, L. (2012). Serum levels of TUBB3 correlate with clinical outcome in Chinese patients with advanced gastric cancer receiving first-line paclitaxel plus capecitabine. Medical Oncology, 29, 3029–3034.
Zhang, C., Awasthi, N., Schwarz, M. A., & Schwarz, R. E. (2013). The dual PI3K/mTOR inhibitor NVP-BEZ235 enhances nab-paclitaxel antitumor response in experimental gastric cancer. International Journal of Oncology, 43, 1627–1635.
Haruki, K., Shiba, H., Fujiwara, Y., Furukawa, K., Iwase, R., Uwagawa, T., et al. (2013). Inhibition of nuclear factor-kappaB enhances the antitumor effect of paclitaxel against gastric cancer with peritoneal dissemination in mice. Digestive Diseases and Sciences, 58, 123–131.
Le, X. F., & Bast, R. C., Jr. (2011). Src family kinases and paclitaxel sensitivity. Cancer Biology and Therapy, 12, 260–269.
Qiu, H., Yashiro, M., Zhang, X., Miwa, A., & Hirakawa, K. (2011). A FGFR2 inhibitor, Ki23057, enhances the chemosensitivity of drug-resistant gastric cancer cells. Cancer Letters, 307, 47–52.
Mi, Y. J., Liang, Y. J., Huang, H. B., Zhao, H. Y., Wu, C. P., Wang, F., et al. (2010). Apatinib (YN968D1) reverses multidrug resistance by inhibiting the efflux function of multiple ATP-binding cassette transporters. Cancer Research, 70, 7981–7991.
Sanui, A., Yotsumoto, F., Tsujioka, H., Fukami, T., Horiuchi, S., Shirota, K., et al. (2010). HB-EGF inhibition in combination with various anticancer agents enhances its antitumor effects in gastric cancer. Anticancer Research, 30, 3143–3149.
Gutierrez-Gonzalez, A., Belda-Iniesta, C., Bargiela-Iparraguirre, J., Dominguez, G., Garcia Alfonso, P., Perona, R., & Sanchez-Perez, I. (2013). Targeting Chk2 improves gastric cancer chemotherapy by impairing DNA damage repair. Apoptosis: An International Journal on Programmed Cell Death, 18, 347–360.
Huang, D., Wang, H., Liu, R., Li, H., Ge, S., Bai, M., et al. (2014). miRNA27a is a biomarker for predicting chemosensitivity and prognosis in metastatic or recurrent gastric cancer. Journal of Cellular Biochemistry, 115, 549–556.
Liu, X., Ru, J., Zhang, J., Zhu, L. H., Liu, M., Li, X., & Tang, H. (2013). miR-23a targets interferon regulatory factor 1 and modulates cellular proliferation and paclitaxel-induced apoptosis in gastric adenocarcinoma cells. PLoS One, 8, e64707.
Wu, H., Huang, M., Lu, M., Zhu, W., Shu, Y., Cao, P., & Liu, P. (2013). Regulation of microtubule-associated protein tau (MAPT) by miR-34c-5p determines the chemosensitivity of gastric cancer to paclitaxel. Cancer Chemotherapy and Pharmacology, 71, 1159–1171.
Kovalchuk, O., Filkowski, J., Meservy, J., Ilnytskyy, Y., Tryndyak, V. P., Chekhun, V. F., & Pogribny, I. P. (2008). Involvement of microRNA-451 in resistance of the MCF-7 breast cancer cells to chemotherapeutic drug doxorubicin. Molecular Cancer Therapeutics, 7, 2152–2159.
Pan, Y. Z., Morris, M. E., & Yu, A. M. (2009). MicroRNA-328 negatively regulates the expression of breast cancer resistance protein (BCRP/ABCG2) in human cancer cells. Molecular Pharmacology, 75, 1374–1379.
Miller, T. E., Ghoshal, K., Ramaswamy, B., Roy, S., Datta, J., Shapiro, C. L., et al. (2008). MicroRNA-221/222 confers tamoxifen resistance in breast cancer by targeting p27Kip1. The Journal of Biological Chemistry, 283, 29897–29903.
Yang, S. M., Huang, C., Li, X. F., Yu, M. Z., He, Y., & Li, J. (2013). miR-21 confers cisplatin resistance in gastric cancer cells by regulating PTEN. Toxicology, 306, 162–168.
Breier, A., Barancik, M., Sulova, Z., & Uhrik, B. (2005). P-glycoprotein—implications of metabolism of neoplastic cells and cancer therapy. Current Cancer Drug Targets, 5, 457–468.
Kim, H. G., Hien, T. T., Han, E. H., Hwang, Y. P., Choi, J. H., Kang, K. W., et al. (2011). Metformin inhibits P-glycoprotein expression via the NF-kappaB pathway and CRE transcriptional activity through AMPK activation. British Journal of Pharmacology, 162, 1096–1108.
Bourguignon, L. Y., Spevak, C. C., Wong, G., Xia, W., & Gilad, E. (2009). Hyaluronan-CD44 interaction with protein kinase C(epsilon) promotes oncogenic signaling by the stem cell marker Nanog and the production of microRNA-21, leading to down-regulation of the tumor suppressor protein PDCD4, anti-apoptosis, and chemotherapy resistance in breast tumor cells. The Journal of Biological Chemistry, 284, 26533–26546.
Acknowledgments
The research is supported by Innovation Fund of Xinjiang Medical University (Fund No. XJC201264) and Innovation Fund of The Third Affiliated Hospital (Xinjiang Tumor Hospital), Xinjiang Medical University (Fund No. SLCX0304).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Jin, B., Liu, Y. & Wang, H. Antagonism of miRNA-21 Sensitizes Human Gastric Cancer Cells to Paclitaxel. Cell Biochem Biophys 72, 275–282 (2015). https://doi.org/10.1007/s12013-014-0450-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0450-2