Abstract
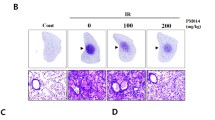
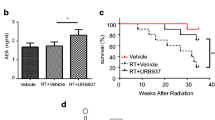
The present research assessed the effects of lettuce glycoside B (LGB), a compound separated and purified from Pterocypsela laciniata, on irradiation-induced pulmonary fibrosis and explored the mechanism involved. Animal model of irradiation exposure inducing pulmonary fibrosis was established by Co irradiator. Rats were intraperitoneally treated with LGB (100, 200 and 400 mg/kg) once per day for a month. Lung index data were analyzed. The levels of fibrosis were assessed by hydroxyproline (Hyp) of pulmonary and lung tissue sections after irradiation exposure. Alveolitis and fibrosis levels were calculated from semi-quantitative analysis of hematoxylin and eosin and Masson’s trichrome lung section staining. The serum levels of transforming growth factor β1 (TGF-β1), interleukin (IL)-6, and tumor necrosis factor-α (TNF-α) were also evaluated. Antioxidant enzymes of superoxide dismutase (SOD) were measured in serum. Moreover, we also measured serum malondialdehyde (MDA) levels, a marker of oxidative stress. Treatment with LGB significantly reduced mortality rates and lung index scores and MDA content, enhanced SOD and other antioxidant enzymes activity, and regulated serum levels of TGF-β1, IL-6, and TNF-α. These results demonstrated that LGB significantly inhibited irradiation-induced pulmonary fibrosis. Furthermore, the results suggested promising clinical effect of LGB therapies for treating irradiation-induced pulmonary fibrosis.



Similar content being viewed by others
References
You, H., Wei, L., Sun, W. L., et al. (2014). The green tea extract epigallocatechin-3-gallate inhibits irradiation-induced pulmonary fibrosis in adult rats. International Journal of Molecular Medicine, 34, 92–102.
Rhieu, B. H., Epperly, M. W., Cao, S., et al. (2014). Improved longevity of hematopoiesis in long-term bone marrow cultures and reduced irradiation-induced pulmonary fibrosis in Toll-like receptor-4 deletion recombinant-negative mice. In Vivo, 28, 441–448.
Wu, Z., Wang, X., Yang, R., et al. (2013). Effects of carbon ion beam irradiation on lung injury and pulmonary fibrosis in mice. Experimental and Therapeutic Medicine, 5, 771–776.
Kalash, R., Berhane, H., Goff, J., et al. (2013). Effects of thoracic irradiation on pulmonary endothelial compared to alveolar type-II cells in fibrosis-prone C57BL/6NTac mice. In Vivo, 27, 291–297.
Gao, F., Fish, B. L., Moulder, J. E., et al. (2013). Enalapril mitigates radiation-induced pneumonitis and pulmonary fibrosis if started 35 days after whole-thorax irradiation. Radiation Research, 180, 546–552.
Rhieu, B. H., Epperly, M. W., Cao, S., et al. (2014). Increased hematopoiesis in long-term bone marrow cultures and reduced irradiation-induced pulmonary fibrosis in von willebrand factor homologous deletion recombinant mice. In Vivo, 28, 449–456.
Zhan, H., Li, S., Sun, J., et al. (2014). Lettuce glycoside B ameliorates cerebral ischemia reperfusion injury by increasing nerve growth factor and neurotrophin-3 expression of cerebral cortex in rats. Indian Journal of Pharmacology, 46, 63–68.
Ali, E. N., & Mansour, S. Z. (2011). Boswellic acids extract attenuates pulmonary fibrosis induced by bleomycin and oxidative stress from gamma irradiation in rats. Chinese Medicine, 6, 36.
Wang, L., Feng, Y., Fu, X. L., et al. (2006). Effects of gene therapy with replication-defective adenovirus ericlosing Egr-1 promoter and Smad7 cDNA on irradiation-induced pulmonary fibrosis: experiment with mice. Zhonghua Yi Xue Za Zhi, 86, 2847–2852.
Epperly, M. W., Franicola, D., Zhang, X., et al. (2006). Reduced irradiation pulmonary fibrosis and stromal cell migration in Smad3-/- marrow chimeric mice. In Vivo, 20, 573–582.
Epperly, M. W., Guo, H., Shields, D., et al. (2004). Correlation of ionizing irradiation-induced late pulmonary fibrosis with long-term bone marrow culture fibroblast progenitor cell biology in mice homozygous deletion recombinant negative for endothelial cell adhesion molecules. In Vivo, 18, 1–14.
Epperly, M. W., Guo, H., Gretton, J. E., et al. (2003). Bone marrow origin of myofibroblasts in irradiation pulmonary fibrosis. American Journal of Respiratory Cell and Molecular Biology, 29, 213–224.
Cheresh, P., Kim, S. J., Tulasiram, S., et al. (2013). Oxidative stress and pulmonary fibrosis. Biochimica et Biophysica Acta, 1832, 1028–1040.
Brass, D. M., Spencer, J. C., Li, Z., et al. (2012). Innate immune activation by inhaled lipopolysaccharide, independent of oxidative stress, exacerbates silica-induced pulmonary fibrosis in mice. PloS One, 7, e40789.
Inghilleri, S., Morbini, P., Campo, I., et al. (2011). Erratum to “factors influencing oxidative imbalance in pulmonary fibrosis: an immunohistochemical study”. Pulmonary Medicine, 2011, 515608.
Inghilleri, S., Morbini, P., Campo, I., et al. (2011). Factors influencing oxidative imbalance in pulmonary fibrosis: an immunohistochemical study. Pulmonary Medicine, 2011, 421409.
Cui, Y., Robertson, J., Maharaj, S., et al. (2011). Oxidative stress contributes to the induction and persistence of TGF-beta1 induced pulmonary fibrosis. International Journal of Biochemistry & Cell Biology, 43, 1122–1133.
Mazur, W., Lindholm, P., Vuorinen, K., et al. (2010). Cell-specific elevation of NRF2 and sulfiredoxin-1 as markers of oxidative stress in the lungs of idiopathic pulmonary fibrosis and non-specific interstitial pneumonia. APMIS, 118, 703–712.
Kliment, C. R., & Oury, T. D. (2010). Oxidative stress, extracellular matrix targets, and idiopathic pulmonary fibrosis. Free Radical Biology and Medicine, 49, 707–717.
Kliment, C. R., Englert, J. M., Gochuico, B. R., et al. (2009). Oxidative stress alters syndecan-1 distribution in lungs with pulmonary fibrosis. Journal of Biological Chemistry, 284, 3537–3545.
Vuorinen, K., Ohlmeier, S., Lepparanta, O., et al. (2008). Peroxiredoxin II expression and its association with oxidative stress and cell proliferation in human idiopathic pulmonary fibrosis. Journal of Histochemistry and Cytochemistry, 56, 951–959.
Helal, G. K., & Helal, O. K. (2009). Metallothionein attenuates carmustine-induced oxidative stress and protects against pulmonary fibrosis in rats. Archives of Toxicology, 83, 87–94.
Walters, D. M., Cho, H. Y., & Kleeberger, S. R. (2008). Oxidative stress and antioxidants in the pathogenesis of pulmonary fibrosis: a potential role for Nrf2. Antioxidants & Redox Signaling, 10, 321–332.
Xie, H., Wang, R., Tang, X., et al. (2012). Paraquat-induced pulmonary fibrosis starts at an early stage of inflammation in rats. Immunotherapy, 4, 1809–1815.
Zhao, Y., Cui, A., Wang, F., et al. (2012). Characteristics of pulmonary inflammation in combined pulmonary fibrosis and emphysema. Chinese Medical Journal, 125, 3015–3021.
Bui, S., Boisserie-Lacroix, V., Ceccato, F., et al. (2012). Pulmonary inflammation in cystic fibrosis. Archives de Pediatrie, 19(Suppl 1), S8–12.
Becker, K. A., Henry, B., Ziobro, R., et al. (2012). Role of CD95 in pulmonary inflammation and infection in cystic fibrosis. Journal Of Molecular Medicine, 90, 1011–1023.
Yang, H. Z., Wang, J. P., Mi, S., et al. (2012). TLR4 activity is required in the resolution of pulmonary inflammation and fibrosis after acute and chronic lung injury. American Journal of Pathology, 180, 275–292.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, Y., Gao, Y., Chen, Y. et al. Effects of Lettuce Glycoside B in Ameliorating Pulmonary Fibrosis Induced by Irradiation Exposure and its Anti-Oxidative Stress Mechanism. Cell Biochem Biophys 71, 971–976 (2015). https://doi.org/10.1007/s12013-014-0295-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0295-8