Abstract
Boron is a trace element which plays important roles in immune response. The relationship between boron and splenic lymphocyte proliferation, apoptosis, secretion of cytokines, and genes potentially related to immune response in ostrich chicks were investigated in the present study. Different concentrations of boron (0, 0.01, 0.1, 0.5, 1, 5, 10, 25, 50, and 100 mmol/L) were applied to splenic lymphocytes of African ostrich, respectively. The effect of boron on lymphocyte proliferation was checked by the CCK-8 method. Flow cytometry was used to detect the effect of boron on apoptosis. The secretion levels of IL-6 and IFN-α were determined by ELISA. Splenic lymphocyte gene expression profiles of ostrich chicks treated with boron (0, 0.1, 100 mmol/L) were studied using RNA-seq technology. The results showed that cell proliferation increased with 0.01–10 mmol/L boron, when it was 25–100 mmol/L, the cell proliferation gradually decreased as the boron concentration increased. Apoptosis ratio in ostrich splenic lymphocytes was closely related to boron concentrations. 0.01- and 0.1-mmol/L boron inhibited apoptosis in splenic lymphocytes, whereas 1, 10, 50, and 100-mmol/L boron promoted apoptosis. As the concentration of boron increased, the secretion of IL-6 gradually decreased; IFN-α was initially increased and then decreased with boron concentrations increased, reaching the maximum level with 1 mmol/L boron. In terms of the RNA-Seq data, there was no differentially expressed gene between the 0- and 0.1-mmol/L boron-treated samples; 21 differentially expressed genes were found between the 0- and 100-mmol/L boron-treated samples; 43 differentially expressed genes were found between the 0.1- and 100-mmol/L boron-treated samples. Functional analysis of the differentially expressed genes by Gene Ontology verified multiple functions associated with immune response. Pathway analysis showed that systemic lupus erythematosus, alcoholism, viral carcinogenesis, and necroptosis pathway were the major enriched pathways, and BIRC2-3, FTH1, and IL-1β genes showed differential expression in necroptosis pathway. These results demonstrated that low concentrations (0.01–0.1 mmol/L) of boron may promote the proliferation and the secretion of cytokines, inhibit cell apoptosis of ostrich splenic lymphocytes by enhancing the function of the cell membrane and the activity of intracellular catalytic enzymes, whereas high-concentration (25–100 mmol/L) boron had opposite effects on cells. The necroptosis pathway might play a pivotal role in regulating the immune response of boron-treated splenic lymphocytes in ostrich chicks.








Similar content being viewed by others
References
Huchzermeyer FW (2002) Diseases of farmed crocodiles and ostriches. Rev Sci Tech 21:265–276
Fan JS (2010) A review of common diseases of ostriches (I). Contemp LS Poult Breed 12:55–57 (in Chinese)
Bonato M, Evans MR, Hasselquist D, Sherley RB, Cloete S, Cherry M (2013) Ostrich chick humoral immune responses and growth rate are predicted by parental immune responses and paternal coloration. Behav Ecol Sociobiol 67:1891–1901
Hunt CD (2006) Dietary boron: evidence for essentiality and homeostatic control in humans and animals. Adv Plant Anim Boron Nutr, p251-267.
Geyikoğglu F, Türkez H (2007) Acute toxicity of boric acid on energy metabolism of the breast muscle in broiler chickens. Biologia 62:112–117
Bozkurt M, Küçükyılmaz K, Catlı AU, Cınar M, Cabuk M, Bintaş E (2012) Effects of boron supplementation to diets deficient in calcium and phosphorus on performance with some serum, bone and fecal characteristics of broiler chickens. Asian Australas J Anim Sci 25:248–255
Couto EAA, Dias-Arieira CR, Kath J, Homiak JA, Puerari HH (2016) Boron and zinc inhibit embryonic development, hatching and reproduction of Meloidogyne incognita. Acta Agric Scand B Soil Plant Sci 66:346–352
Haseeb K, Wang J, Xiao K, Yang KL, Sun PP, Wu XT, Luo Y, Song H, Liu HZ, Zhong JM, Peng KM (2018) Effects of boron supplementation on expression of Hsp70 in the spleen of African ostrich. Biol Trace Elem Res 182:317–327
Xiao K, Ansari AR, Rehman ZU, Khaliq H, Song H, Tang J, Wang J, Wang W, Sun PP, Zhong J, Peng KM (2015) Effect of boric acid supplementation of ostrich water on the expression of Foxn1 in thymus. Histol Histopathol 30:1367–1378
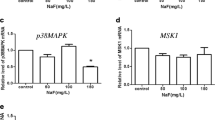
Xiao K, Yang K, Wang J, Sun P, Huang H, Khaliq H, Naeem MA, Zhong J, Peng K (2019) Transcriptional study revealed that boron supplementation may alter the immune-related genes through MAPK signaling in ostrich chick thymus. Biol Trace Elem Res 189:209–223
Huang HB, Xiao K, Lu S, Yang KL, Ansari AR, Khaliq H, Song H, Zhong J, Liu HZ, Peng KM (2015) Increased thymic cell turnover under boron stress may bypass TLR3/4 pathway in African Ostrich. PLoS One 10:e0129596
Jin EH, Gu YF, Wang J, Jin GM, Li SH (2014) Effect of supplementation of drinking water with different levels of boron on performance and immune organ parameters of broilers. Ital J Anim Sci 13:205–214
Jin E, Li SH, Ren M, Hu Q, Gu Y, Li K (2017) Boron affects immune function through modulation of splenic T lymphocyte subsets, cytokine secretion, and lymphocyte proliferation and apoptosis in rats. Biol Trace Elem Res 178:261–275
Jin E, Ren M, Liu W, Liang S, Hu Q, Gu Y, Li S (2017) Effect of boron on thymic cytokine expression, hormone secretion, antioxidant functions, cell proliferation, and apoptosis potential via the ERK1/2 signaling pathway. J Agric Food Chem 65:11280–11291
Yang KL, Zheng XT Zhu DY, Lu S, Huang HB, Sun PP, Wang J, Tang J, Wang W, Zheng XT, Zhu DY (2014) Effects of boron on the structure of bursa of African ostrich chicks. Symposium of the Society of Animal Anatomy and Histoembryology, Chinese Society of Animal Husbandry and Veterinary Medicine p398-404.(in Chinese)
Brand TS (2010) Elsenburg ostrich feed databases. Elsenburg Agricultural Research Centre, Elsenburg, South Africa
Gong BS, Ren M, Jin EH, Hu QQ, Zhou JX, Li SH (2017) Effects of boron on morphology, proliferation and apoptosis of granulosa cells in porcine follicles. Health Res 46:164–167+172 (in Chinese)
Nielsen FH (2004) Dietary fat composition modifies the effect of boron on bone characteristics and plasma lipids in rats. BioFactors 20:161–171
Howe PD (1998) A review of boron effects in the environment. Biol Trace Elem Res 66(1-3):153–166
Canturk Z, Tunali Y, Korkmaz S, Gulbaş Z (2016) Cytotoxic and apoptotic effects of boron compounds on leukemia cell line. Cytotechnology 68:87–93
Pei YQ, Zhang Q, Hu QQ, Li SH, Jin GM, Jin EH (2018) Effects of boron on proliferation and apoptosis of splenic lymphocytes in rats. J Anhui Univ Sci Technol 2:7–13 (in Chinese)
Meager A (2002) Biological assays for interferons. J Immunol Methods 261:21–36
Han HF, Li YJ, Cheng XC, Zhang CJ, Yu ZH, Yang H, Yang RE, Fu W (2016) Study on the Anti-IBDV activity of chicken IFN-α and its effects on signaling molecule PI3K and NF-κB. Chinese J Anim Hubs Vet Med 43:2164–2169 (in Chinese)
Hunt CD (2003) Dietary boron: an overview of the evidence for its role in immune function. J Trace Elem Exp Med 16:291–306
Bai Y, Hunt CD (1995) Dietary boron alleviates adjuvant-induced arthritis (AIA) in rats. FASEB J 9:A576
Armstrong TA, Spears JW, Lloyd KE (2001) Inflammatory response, growth, and thyroid hormone concentrations are affected by long term Boron supplementation in gilts. J Anim Sci 79(6):1549–1556
Armstrong TA, Spears JW (2003) Effects of boron supplementation of pig diets on the production of tumor necrosis factor-α and interferon-γ. J Anim Sci 81(10):2552–2561
Cho YS, Challa S, Moquin D, Genga R, Ray TD, Guildford M, Chan FK (2009) Phosphorylation-driven assembly of the RIP1–RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell 137:1112–1123
Kovalenko A, Kim JC, Kang TB, Rajput A, Bogdanov K, Dittrich-Breiholz O, Kracht M, Brenner O, Wallach D (2009) Caspase-8 deficiency in epidermal keratinocytes triggers an inflammatory skin disease. J Exp Med 206:2161–2177
Schneider-Brachert W, Tchikov V, Merkel O, Jakob M, Hallas C, Kruse ML, Groitl P, Lehn A, Hildt E, Held-Feindt J, Dobner T, Kabelitz D, Krönke M, Schütze S (2006) Inhibition of TNF receptor 1 internalization by adenovirus 14.7K as a novel immune escape mechanism. J Clin Invest 116:2901–2913
Gozzelino R, Soares MP (2013) Coupling heme and iron metabolism via Ferritin H chain. Antioxid Redox Signal 20:1754–1769
Frossi B, De Carli M, Piemonte M, Pucillo C (2008) Oxidative microenvironment exerts an opposite regulatory effect on cytokine production by Th1 and Th2 cells. Mol Immunol 45:58–64
Kaminski MM, Sauer SW, Klemke CD, Süss D, Okun JG, Krammer PH, Gülow K (2010) Mitochondrial reactive oxygen species control T cell activation by regulating IL-2 and IL-4 expression: mechanism of ciprofloxacin-mediated immunosuppression. J Immunol 184:4827–4841
Belikov AV, Schraven B, Simeoni L (2015) T cells and reactive oxygen species. J Biomed Sci 22:85
Abimannan T, Peroumal D, Parida JR, Barik PK, Padhan P, Devadas S (2016) Oxidative stress modulates the cytokine response of differentiated Th17 and Th1 cells. Free Radic Biol Med 99:352–363
Guang B, Zhang SJ, Yao J, Kong SS, Cheng BL (2017) Advances in research on the mechanism of necroptosis. Prog Mod Biomed 35:6992–6996 (in Chinese)
Funding
The authors received financial support from the National Natural Science Foundation of China (No. 31672504).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures were approved by the Animal Care and Welfare Committee of our Institute.
Conflict of Interest
The authors declare that they have no conflict of interest.
Disclaimer
The authors alone are responsible for the content and writing of this article.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, X., Xiao, K., Qiu, W. et al. The Immune Regulatory Effect of Boron on Ostrich Chick Splenic Lymphocytes. Biol Trace Elem Res 199, 2695–2706 (2021). https://doi.org/10.1007/s12011-020-02392-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02392-5