Abstract
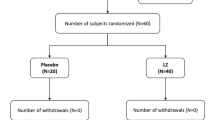
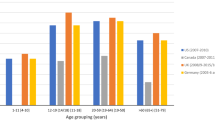
The purpose of the present study was the assessment of the serum concentration of antioxidant microelements—zinc, copper, selenium, manganese, and Cu/Zn ratio in children and adolescents with myopia. Eighty-three children were examined (mean age 14.36 ± 2.49 years) with myopia. The control group was 38 persons (mean age 12.89 ± 3.84 years). Each patient had complete eye examination. The serum concentration of zinc, copper, manganese, and selenium was determined by atomic absorption spectrometry. Cu/Zn ratio, which is the indicator of the oxidative stress, was also calculated. The average serum concentration of zinc in myopic patients was significantly lower (0.865 ± 0.221 mg L−1) in comparison to the control group (1.054 ± 0.174 mg L−1). There was significantly higher Cu/Zn ratio in myopic patients (1.196 ± 0.452) in comparison to that in the control group (0.992 ± 0.203). The average serum concentration of selenium in the study group was significantly lower (40.23 ± 12.07 μg L−1) compared with that in the control group (46.00 ± 12.25 μg L−1). There were no essential differences between serum concentration of copper and manganese in the study group and the control group. Low serum concentration of zinc and selenium in myopic children may imply an association between insufficiency of these antioxidant microelements and the development of the myopia and could be the indication for zinc and selenium supplementation in the prevention of myopia. Significantly, higher Cu/Zn ratio in the study group can suggest the relationship between myopia and oxidative stress.




Similar content being viewed by others
References
Czepita D, Żejmo M, Czepita DA, Łodygowska E (2013) Myopia-epidemiology, pathogenesis, treatment. Okulistyka 1:74–78
Morgan IG, Ohno-Matsui K, Saw SM (2012) Myopia. Lancet 379:1739–1748
Bosch-Morell F, Mérida S, Navea A (2015) Oxidative stress in myopia. Oxidative Med Cell Longev 2015: 750637. doi:10.1155/2015/750637
Jonas JB, Xu L (2014) Histological changes of high axial myopia. Eye 28:113–117
McBrien NA, Jobling AI, Gentle A (2009) Biomechanics of the sclera in myopia: extracellular and cellular factors. Optom Vis Sci 86:23–30
Gentle A, Liu Y, Martin JE, Conti GL, McBrien NA (2003) Collagen gene expression and the altered accumulation of scleral collagen during the development of high myopia. J Biol Chem 278:16587–16594
Cassagne M, Malecaze F, Soler V (2014) Pathophysiology of myopia: nature versus nurture. J Fr Ophtalmol 37:407–414
Baldwin W (1981) A review of statistical studies of relations between myopia and ethnic, behavioral, and physiological characteristics. Am J Optom Physiol Optic 58:516–527
Huibi X, Kaixun H, Qiuhua G, Yushan Z, Xiuxian H (2001) Prevention of axial elongation in myopia by the trace element zinc. Biol Trace Elem Res 79:39–47
Sukalski KA, LaBerge TP, Johnson WT (1997) In vivo oxidative modification of erythrocyte membrane proteins in copper deficiency. Free Radic Biol Med 22:835–842
Kuo S, Chesrown SE, Mellott JK, Rogers RJ, Hsu J, Nick HS (1999) In vivo architecture of the manganese superoxide dismutase promoter. J Biol Chem 274:3345–3354
Rahman MA, Rahman B, Ahmed N (2013) High blood manganese in iron-deficient children in Karachi. Public Health Nutr 16:1677–1683
Harris ED, Rayton JK, Balthrop JE, DiSilvestro RA, Garcia-de-Quevedo M (1980) Copper and the synthesis of elastin and collagen. CIBA Found Symp 79:163–182
Rucker RB, Kosonen T, Clegg MS, Mitchell AE, Rucker BR, Uriu-Hare JY, et al. (1998) Copper, lysyl oxidase, and extracellular matrix protein cross-linking. Am J Clin Nutr 67(Suppl 5):996S–1002S
Avetisov ES, Vinetskaia MI, Iomdina EN, Makhmudova FR, Boltaeva ZK, Tarutta EP (1991) Copper metabolism in scleral tissue and possibilities of its correction in myopia. Vestn oftalmol 107:31–34
Shils ME, Olson JA, Shike M, Ross AC (1998) Modern nutrition in health and disease, 2nd ed., Lippincott Williams & Wilkins, 1326–1338
Silverstone BZ, Syrkin N, Algur N, Berson D (1985) A metabolic aspect of high myopia. Ann Ophthalmol 17:546–551
Silverstone BZ, Seelenfreund MH, Berson D, Grushka E, Mazover AJ, Kristol L, et al. (1986) Copper and zinc metabolism in high myopic patients with retinal detachment evaluation of the Cu/Zn ratio. Metab Pediatr Syst Ophthalmol 9:581–583
Qiang M, Zhao R (1991) A logistic regression analysis of relations between juvenile myopia and TV-watching, trace elements, and psychological characteristics. Chung Hua Yu Fang I Hsueh Tsa Chih 25:222–224
Shiue C, Ko LS (1988) Study on serum copper and zinc levels in high myopia. Acta Ophthalmol Suppl 185:141–142
Wang L (2009) Variation analysis of six kinds of common microelements contents of blood in myopic primary school students in Dongguan district. Cent Chin Med J 1:20–21
Li J, Peng Y, Li X (2005) An analysis of microelements in patients with high myopia. Journal of Guangzhou University of Traditional Chinese Medicine 3:197–199
Huo M, Liu H, Cao J (2006) The relationship between serum zinc, copper, selenium and the visions of middle school students. Chin J Sch Health 4:318–319
Xie X, He H, Wang J, et al. (2003) Clinical significance of serum trace elements in juvenile patients with myopia. J Huaihai Med 4:279–280
Neumeister B, Besenthal I, Böhm BO (2013) Klinikleitfaden. Labordiagnostik. Urban & Fisher Verlag, München
FAO, IAEA (1996) Trace elements in human nutrition and health. World Health Organization, Geneva
Fedor M, Urban B, Bakunowicz-Łazarczyk A (2015) Rola pierwiastków śladowych w patogenezie i progresji krótkowzroczności. Okul Dypl 5:12–16
Powell SR (2000) The antioxidant properties of zinc. J Nutr 130:1447S–1454S
Florence TM (1995) The role of free radicals in disease. Aust N Z J Ophthalmol 23:3–7
Markiewicz R, Socha K, Borawska MH, Gutowska A (2008) Zinc and copper in diets of people living in the social nursing home in Białystok. Rocz Panstw Zakl Hig 59(4):415–420
Charkiewicz AE, Omeljaniuk WJ, Kanicka M, Szpak A, Szpak A (2013) Average mineral content in the diet of men aged 45-54 years living in the Bialystok region. Probl Hig Epidemiol 94(4):905–909
Socha K, Borawska MH, Markiewicz R, Charkiewicz WJ (2009) An evaluation of the nutritional habits of the students of the Institute of Cosmetology and Health Care in Bialystok. Bromatol Chem Toksykol 42(3):704–708
Gapy B, Raszeja-Specht A, Bielarczyk H (2014) Role of zinc in physiological and pathological processes of the body. J Lab Diagn 50:45–52
Milne DB (1998) Copper intake and assessment of copper status. Am J Clin Nutr 67(Suppl 5):1041S–1045S
Kolia KI, M’boh GM, Bagré I, Djaman AJ (2014) Assessment of serum iron, manganese and Cu/Zn ratio in the course of falciparum malaria among ivorian patients. Int J Biochem Res Rev 4:527–535
M’boh GM, Boyvin L, Beourou S, Djaman AJ (2013) Int J Child Health Nutr 2:29–33
Bettger WJ (1993) Zinc and selenium, site-specific versus general antioxidation. Can J Physiol Pharmacol 71:721–724
Wasowicz W, Gromadzinska J, Rydzynski K, Tomczak J (2003) Selenium status of low-selenium area residents: polish experience. Toxicol Lett 137:95–101
Markiewicz R, Socha K, Borawska MH, Gutowska A (2010) Selenium in diets and serum of elderly people from the north-eastern region of Poland. Fresenius Environ Bull 19:372–377
Prabhu KS, Zamamiri-Davis F, Stewart JB, Thompson JT, Sordillo LM, Reddy CC (2002) Selenium deficiency increases the expression of inducible nitric oxide synthase in RAW 264.7 macrophages: role of nuclear factor-kappaB in up-regulation. Biochem J 366:203–209
Djujić IS, Jozanov-Stankov ON, Milovac M, Jankovic V, Djermanovic V (2000) Bioavailability and possible benefits of wheat intake naturally enriched with selenium and its products. Biol Trace Elem Res 77:273–285
Skripchenko ND, Sharafetdinov KK, Plotnikova OA, Meshcheriakova VA, Mal’tsev G (2003) Effect of selenium enriched diet on lipid peroxidation in patients with diabetes mellitus type 2. Vopr Pitan 72:14–17
Vinceti M, Mandrioli J, Borella P, Michalke B, Tsatsakis A, Finkelstein Y (2014) Selenium neurotoxicity in humans: bridging laboratory and epidemiologic studies. Toxicol Lett 230:295–303
Nagy G, Benko I, Kiraly G, Voros O, Tanczos B, Sztrik A, et al. (2015) Cellular and nephrotoxicity of selenium species. J Trace Elem Med Biol 30:160–167
Hays SM, Macey K, Nong A, Aylward LL (2014) Biomonitoring equivalents for selenium. Regul Toxicol Pharmacol 70(1):333–339
Kenfield SA, Van Blarigan EL, DuPre N, Stampfer MJ, Giovannucci LE, Chan JM (2015) er mortality. J Natl Cancer Inst 107(1):360. doi:10.1093/jnci/dju360
Acknowledgments
The study was conducted with the use of equipment purchased by the Medical University of Bialystok as part of the OP DEP 2007-2013, Priority Axis I.3, contract No. POPW.01.03.00-20-022/09. The presented paper was financially supported by the Polish Ministry of Science and Higher Education (grant no. N/ST/ZB/15/001/1169 and N/ST/ZB/15/2/1169). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interests
The authors declare that they have no conflict interests.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
This article does not contain any studies with animals performed by any of the authors.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Fedor, M., Socha, K., Urban, B. et al. Serum Concentration of Zinc, Copper, Selenium, Manganese, and Cu/Zn Ratio in Children and Adolescents with Myopia. Biol Trace Elem Res 176, 1–9 (2017). https://doi.org/10.1007/s12011-016-0805-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-016-0805-1