Abstract
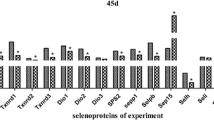
Selenoprotein U (SelU) may regulate a myriad of biological processes through its redox function. In chicks, neither the nucleotide sequence nor the amino acid sequence is known. The main objectives of this study were to clone and characterize the chicken Selu gene and investigate Selu messenger RNA (mRNA) and protein expression in chicken tissues. The coding sequence (CDS) of Selu contained 387 bases with a typical mammalian selenocysteine insertion sequence (SECIS) located in the 3′-untranslated region. The deduced amino acid sequence of chicken SelU contains 224 amino acids with UAA as the stop codon. Like all SelU genes identified in different species, chicken SelU contains one well-conserved selenocysteine (Sec) at the 85th position encoded by the UGA codon. The SECIS element was with the conserved denosine (--AAA--) rather than the motif cytidine (--CC--) motif. Moreover, the expression pattern of Selu mRNA in muscle, liver, kidney, heart, spleen, lung, testis, and brain was analyzed with real-time quantitative PCR in young male chickens fed a Se-deficient corn-soybean meal basal diet supplemented with 0.0 and 0.3 mg Se/kg in the form of sodium selenite. We found that the abundance of Selu mRNA in muscle, liver, kidney, heart, spleen, and lung was downregulated (P < 0.05) by Se deficiency. However, it was not affected by dietary Se concentrations in testis and brain. Furthermore, protein abundance of SelU in these seven tissues was consistent with the mRNA abundance. Hence, we suggest that Selu might play an important role in the biochemical function of Se in birds.




Similar content being viewed by others
Abbreviations
- BD:
-
Basal diet
- EST:
-
Expressed sequence tag
- PCR:
-
Polymerase chain reaction
- Q-PCR:
-
Real-time quantitative PCR
- Se:
-
Selenium
- SECIS:
-
Selenocysteine insertion sequence
- SelU:
-
Selenoprotein U
References
Rayman MP (2000) The importance of selenium to human health. Lancet 356:233–241
Schomburg L, Schweizer U, Köhrle J (2004) Selenium and selenoproteins in mammals: extraordinary, essential, enigmatic. Cell Mol Life Sci 61:1988–1995
Kryukov GV, Castellano S, Novoselov SV et al (2003) Characterization of mammalian selenoproteomes. Science 300:1439–1443
Castellano S, Novoselov SV, Kryukov GV et al (2004) Reconsidering the evolution of eukaryotic selenoproteins: a novel nonmammalian family with scattered phylogenetic distribution. EMBO Rep 5:71–77
Liu Y, Zhao H, Zhang Q et al (2012) Prolonged dietary selenium deficiency or excess does not globally affect selenoprotein gene expression and/or protein production in various tissues of pigs. J Nutr 142:1410–1416
Sunde RA, Raines AM (2011) Selenium regulation of the selenoprotein and nonselenoprotein transcriptomes in rodents. Adv Nutr 2:138–150
Huang JQ, Li DL, Zhao H et al (2011) The selenium deficiency disease exudative diathesis in chicks is associated with downregulation of seven common selenoprotein genes in liver and muscle. J Nutr 141:1605–1610
Mariotti M, Lobanov AV, Guigo R et al (2010) SECISearch3 and Seblastian: new tools for prediction of SECIS elements and selenoproteins. Nucleic Acids Res 41:e149–e149
Roy A, Kucukural A, Zhang Y (2010) I-TASSER: a unified platform for automated protein structure and function prediction. Nat Protoc 5:725–738
Zhang Y (2008) I-TASSER server for protein 3D structure prediction. BMC Bioinforma 9:40
Tamura K, Dudley J, Nei M et al (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Grundner-Culemann E, Martin GW, Harney JW et al (1999) Two distinct SECIS structures capable of directing selenocysteine incorporation in eukaryotes. RNA 5:625–635
Korotkov KV, Novoselov SV, Hatfield DL et al (2002) Mammalian selenoprotein in which selenocysteine (Sec) incorporation is supported by a new form of Sec insertion sequence element. Mol Cell Biol 22:1402–1411
Noguchi T, Cantor AH, Scott ML (1973) Mode of action of selenium and vitamin E in prevention of exudative diathesis in chicks. J Nutr 103:1502–1511
Thompson J, Scott M (1969) Role of selenium in the nutrition of the chick. J Nutr 97:335–342
You L, Liu C, Yang ZJ et al (2014) Prediction of Selenoprotein T Structure and Its Response to Selenium Deficiency in Chicken Immune Organs. Biol Trace Elem Res 160:222–231
Mariotti M, Ridge PG, Zhang Y et al (2012) Composition and evolution of the vertebrate and mammalian selenoproteomes. PLoS One 7:e33066
Fomenko DE, Gladyshev VN (2003) Identity and functions of CxxC-derived motifs. Biochemistry 42:11214–11225
Burk RF, Hill KE (2009) Selenoprotein P—expression, functions, and roles in mammals. BBA-Gen Subj 1790:1441–1447
Li JL, Ruan HF, Li HX et al (2011) Molecular cloning, characterization and mRNA expression analysis of a novel selenoprotein: avian selenoprotein W from chicken. Mol Biol Rep 38:4015–4022
Ou BR, Jiang MJ, Lin CH et al (2011) Characterization and expression of chicken selenoprotein W. Biometals 24:323–333
Kryukov GV, Kryukov VM, Gladyshev VN (1999) New mammalian selenocysteine-containing proteins identified with an algorithm that searches for selenocysteine insertion sequence elements. J Biol Chem 274:33888–33897
Reeves MA, Bellinger FP, Berry MJ et al (2010) The neuroprotective functions of selenoprotein M and its role in cytosolic calcium regulation. Antioxid Redox Signal 12:809–818
Dikiy A, Novoselov SV, Fomenko DE et al (2007) SelT, SelW, SelH, and Rdx12: genomics and molecular insights into the functions of selenoproteins of a novel thioredoxin-like family. Biochemistry 46:6871–6882
Schomburg L, Schweizer U (2009) Hierarchical regulation of selenoprotein expression and sex-specific effects of selenium. BBA-Gen Subj 1790:1453–1462
Yao H, Liu W, Zhao W et al (2014) Different responses of selenoproteins to the altered expression of selenoprotein W in chicken myoblasts. RSC Adv 4:64032–64042
Yao HD, Wu Q, Zhang ZW et al (2013) Selenoprotein W serves as an antioxidant in chicken myoblasts. BBA-Gen Subj 1830:3112–3120
Yao HD, Wu Q, Zhang ZW et al (2013) Gene expression of endoplasmic reticulum resident selenoproteins correlates with apoptosis in various muscles of se-deficient chicks. J Nutr 143:613–619
Zhang S, Rocourt C, Cheng WH (2010) Selenoproteins and the aging brain. Mech Ageing Dev 131:253–260
Acknowledgments
This research was supported in part by Chinese Natural Science Foundation: the Major International (Regional) Joint Research Program of China (No. 31320103920) and the Project of Creating Excellent of the Capital (Beijing) Food Safety Technology (No. Z141100002614011).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, YY., Huang, JQ., Lin, GC. et al. Characterization and Expression of Chicken Selenoprotein U. Biol Trace Elem Res 166, 216–224 (2015). https://doi.org/10.1007/s12011-015-0257-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-015-0257-z